Abstract
Background:
Progression of disease (POD24) within 24 months after front-line therapy in patients with Follicular lymphoma (FL) is associated with poor prognosis compared to delayed progression. There is a scarcity of data regarding the best treatment approach for these patients. We performed this systematic review to assess the efficacy of various therapeutic regimens used for the treatment of FL with POD24.
Methodology:
Three databases (PubMed, Cochrane Library, ClinicalTrials.gov) were searched in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines using the MeSH terms and keywords for: "Follicular Lymphoma" AND "recurrence" AND "therapy". The literature search was performed from Jan 01, 2010, to May 8, 2021. A total of 1832 studies were screened, and duplicates, reviews, and irrelevant articles were excluded. We included prospective and retrospective clinical trials presenting separate treatment outcomes of FL with POD24. A total of 3 studies were selected for inclusion after the application of inclusion and exclusion criteria.
Results:
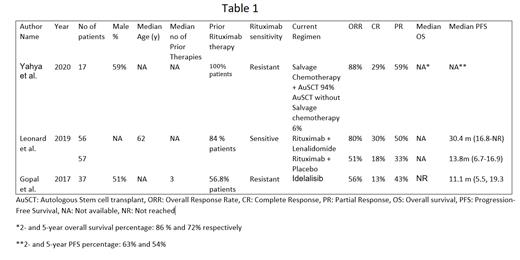
A total of 167 patients from 3 studies were included for this review. In a retrospective study, High dose chemotherapy with autologous stem cell transplantation (HDC/ AutoSCT) for patients (n=17) with rituximab resistant FL with POD24 showed an objective response rate (ORR) of 88%, complete response (CR) of 29%, and partial response (PR) of 59%. In a phase III study of rituximab sensitive patients, 12 cycles of a combination of lenalidomide and rituximab (n=56) showed ORR of 80 %, CR of 30%, and PR of 50% whereas 12 cycles of Rituximab and placebo showed ORR, CR, and PR of 51%, 18%, and 33% respectively. In a retrospective post hoc analysis of subgroups of phase II clinical trial (n=37), Idelalisib showed an ORR of 56%, CR of 13%, and PR of 43% in rituximab-resistant patients. (Table 1)
Conclusion:
FL with POD24 is a poor prognostic factor for which therapeutic data is limited and uniform treatment recommendations are not available. Further prospective clinical trials with randomization and blinding are needed.
Khan: Abbvie: Research Funding, Speakers Bureau; Astrazeneca: Research Funding, Speakers Bureau; Beigene: Research Funding, Speakers Bureau; Epizyme: Research Funding, Speakers Bureau; Genentech: Research Funding, Speakers Bureau; GSK: Research Funding, Speakers Bureau; Karyopharm: Speakers Bureau; Kite: Speakers Bureau; Morphosys: Speakers Bureau; Sanofi: Speakers Bureau; SeaGen: Speakers Bureau. Anwer: GlaxoSmithKline: Research Funding; Janssen pharmaceutical: Honoraria, Research Funding; BMS / Celgene: Honoraria, Research Funding; Allogene Therapeutics: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal