Abstract
Background: Based on the results of a phase I/II study in Japan (Trial registration: JapicCTI-173646), tirabrutinib (TIR), a second-generation inhibitor of Bruton's tyrosine kinase, was approved in March 2020 for the treatment of relapsed or refractory primary central nervous system lymphoma (r/r PCNSL). We have previously reported overall response rate of 63.6% and manageable safety profile results of this study (Narita et al. Neuro Oncol. 2021;23(1):122-133). Further, one-year follow-up data after the last patient had enrolled showed that the effects of TIR persisted in r/r PCNSL patients (Mishima et al. Poster presented at the Society for Neuro-Oncology virtual conference; November 19-21, 2020). Here, based on this one-year follow-up data, we describe the Quality of Life (QoL) and Karnofsky Performance Status (KPS) in r/r PCNSL patients treated with TIR.
Methods: Patients with r/r PCNSL, age ≥20 years, and KPS ≥70 were treated with TIR once daily (QD) at a dose of 320 mg, 480 mg, or 480 mg upon fasting (480 mg fasted QD). TIR was administered in 28-day cycles, and treatment was continued until disease progression or clinically unacceptable toxicity was observed. QoL was assessed using questionnaires issued by the European Organization for Research and Treatment of Cancer (EORTC), namely QLQ-C30 (EORTC QLQ-C30), EORTC QLQ-BN20, and EuroQol 5 dimensions 3-level (EQ-5D-3L). The QoL survey was conducted during the screening period, on Day 28 of Cycle 1, after every 2 cycles (i.e., after Day 1 of Cycle 3), after every 4 cycles (i.e., after Day 1 of Cycle 25), and at the end of treatment. KPS was measured during the screening period, on days 1, 8, 15, and 28 of Cycle 1, on day 1 of every cycle after Cycle 3, and at the end of treatment.
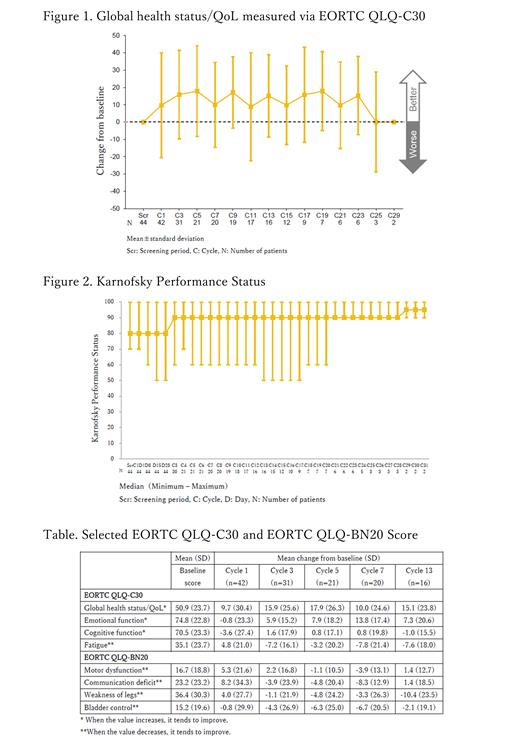
Results: Forty-four patients were prospectively enrolled, and 20, 7, and 17 patients were treated with TIR at 320 mg QD, 480 mg QD, and 480 mg fasted QD, respectively. Median patient age was 60 years (range 29-86). Median follow-up period was 14.9 months (range, 1.4-27.7) for the entire cohort but was 19.1 months, 23.5 months, and 12.0 months for the 320 mg QD, 480 mg QD, and 480 mg fasted QD groups, respectively. At the time of data cutoff (February 25, 2020), 11 patients (25%) remained on treatment. Mean (SD) score for the global health status/QoL section of the EORTC QLQ-C30 was 50.9 (23.7) at baseline and remained relatively constant during treatment (Figure 1). This trend was also observed for emotional function, cognitive function, and fatigue sections of the EORTC QLQ-C30, for motor dysfunction, communication deficit, weakness of legs, and bladder control sections of the EORTC QLQ-BN20, and in items pertaining to self-care, usual activities, and anxiety/depression in the EQ-5D-3L. Median KPS was 80.0 (range, 70-100) at baseline, which remained unchanged during TIR treatment (Figure 2).
Conclusion: QoL and KPS scores in r/r PCNSL patients were maintained during TIR administration, a new treatment option for r/r PCNSL, which does not lead to the deterioration of QoL and KPS.
Terui: Ono Pharmaceutical: Speakers Bureau; MSD: Speakers Bureau; Janssen: Speakers Bureau; Esai: Speakers Bureau; Chugai Pharmaceutical: Speakers Bureau; Celgene: Speakers Bureau; AbbVie: Speakers Bureau; Takeda Pharmaceutical: Speakers Bureau. Narita: Ono Pharmaceutical co.: Honoraria, Research Funding; Dainippon-Sumitomo: Membership on an entity's Board of Directors or advisory committees, Research Funding; Eisai: Honoraria, Research Funding; Stella-pharma: Research Funding; Daiichi-Sankyo: Honoraria, Research Funding; Bayer: Research Funding; Ohara: Research Funding; Chugai Pharmaceutical co.: Honoraria; Novocure: Honoraria. Nagane: AbbVie: Membership on an entity's Board of Directors or advisory committees, Research Funding; Chugai: Honoraria, Research Funding; Daiichi-Sankyo: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Eisai: Honoraria, Research Funding; Pfizer: Research Funding; MSD: Research Funding; Astellas: Research Funding; Nippon-Kayaku: Honoraria, Research Funding; Bayer: Honoraria, Research Funding; Shionogi: Research Funding; Otsuka: Research Funding; Ono Pharma: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Novocure: Honoraria; Sumitomo Dainippon Pharma: Honoraria; RIEMSER: Membership on an entity's Board of Directors or advisory committees. Mishima: Ono Pharmaceutical Co: Research Funding; Astellas: Research Funding; HOYA Technosurgical Co.: Research Funding; Daiichi-Sankyo: Research Funding; AbbVie: Research Funding; Medical U and A: Research Funding; Teijin Pharma: Research Funding; Eisai: Research Funding; MSD: Research Funding; Chugai: Research Funding. Arakawa: Sanofi: Research Funding; Carl Zeiss: Honoraria, Research Funding; Brainlab: Honoraria, Research Funding; Nihon Medi-Physics: Research Funding; Ono Pharmaceutical: Honoraria, Research Funding; Philips: Research Funding; Siemens: Research Funding; Tanabe Mitsubishi: Research Funding; Chugai: Honoraria, Research Funding; Eisai: Honoraria, Research Funding; Merck: Honoraria, Research Funding; Meiji Seika: Honoraria, Research Funding; Daiichi Sankyo: Honoraria, Research Funding; CSL Behring: Honoraria, Research Funding; Takeda: Research Funding; Pfizer: Research Funding; Stryker: Research Funding; Astellas Pharma: Research Funding; Taiho Pharma: Research Funding; Nippon Kayaku: Honoraria; Novocure: Honoraria; UCB Japan: Honoraria; Integra Japan: Honoraria; Otsuka: Honoraria; Abbvie: Honoraria. Yonezawa: Eisai: Speakers Bureau; Ono Pharmaceutical co.: Speakers Bureau; Chugai Pharmaceutical co.: Speakers Bureau. Fukuhara: Celgene: Honoraria, Research Funding; Chugai Pharmaceutical: Honoraria, Research Funding; Eisai: Honoraria; HUYA Bioscience International: Honoraria; Incyte: Research Funding; Janssen: Honoraria; Kyowa Kirin: Honoraria; Nippon Shinyaku: Honoraria; Novartis: Honoraria; Ono Pharmaceutical: Honoraria, Research Funding; Takeda Pharmaceutical: Honoraria; Zenyaku Kogyo: Honoraria; Bayer: Research Funding; AbbVie: Honoraria. Sugiyama: Daichi Sankyo Inc.: Consultancy; Ono Pharmaceutical Inc: Honoraria. Aoi: Ono Pharma USA, Inc.: Current Employment. Nishikawa: Novocure: Consultancy; Chugai: Honoraria, Research Funding; MSD: Research Funding; Daiichi-Sankyo: Honoraria, Research Funding; Eisai: Honoraria, Research Funding; Ono: Honoraria; Nihon-Kayaku: Honoraria.
Tirabrutinib. Clinical trial for PCNSL.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal