Abstract
Background
Patients (pts) with RRMM previously exposed to immunomodulatory agents, proteasome inhibitors (PIs), and anti-CD38 antibodies were reported to have poor outcomes with subsequent therapy, and deep and durable responses were uncommon (Chari et al. N Engl J Med 2019, Lonial et al. Lancet Oncol 2019, Richardson et al. J Clin Oncol 2020). This highlighted the need for treatments capable of improving responses for such pts.
Ide-cel, a BCMA-directed CAR T cell therapy, showed a favorable safety profile and frequent, deep, durable responses in heavily pretreated pts with RRMM (Raje et al. N Engl J Med 2019, Munshi et al. N Engl J Med 2021), and was approved in the US for pts with ≥ 4 prior lines of therapy including an immunomodulatory agent, a PI, and an anti-CD38 antibody. Long-term results from the pivotal phase 2 KarMMa study showed ide-cel induced an overall response rate (ORR) of 73% (33% complete response [CR] rate) with a median duration of response (DOR) of 10.9 months; median overall survival (OS) was 24.8 months (Anderson et al. ASCO 2021 Poster 8016).
Combining ide-cel with other agents may enhance efficacy, resulting in long-term clinical benefits for pts. Agents in development include iberdomide (CC-220), an oral, potent novel cereblon E3 ligase modulator (CELMoD ®) compound with direct tumoricidal and immune-stimulatory effects that has shown promising activity in heavily pretreated pts (van de Donk et al. ASH 2020 Abstract 724), and BMS-986405, a gamma secretase inhibitor that blocks shedding of surface BCMA to enhance antitumor activity of BCMA-directed CAR+ T cells (Cowan et al. ASH 2019 Abstract 204). Standard treatments for RRMM include daratumumab + pomalidomide + dexamethasone (DPd)andpomalidomide + bortezomib + low-dose dexamethasone (PVd). This dose-finding safety and efficacy study will assess ide-cel in combination with these agents in pts with RRMM.
Methods
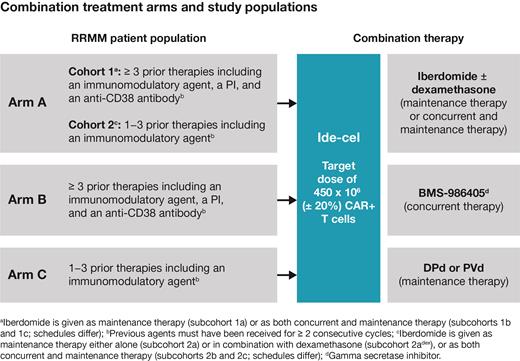
KarMMa-7 (NCT04855136) is an exploratory, open-label, multicenter, phase 1/2 study. Eligible pts are ≥ 18 years with measurable MM, who have received either ≥ 3 or 1-3 prior regimens depending on treatment arm/cohort (Figure). Pts must have achieved at least a minimal response to a prior treatment, have disease progression during, or within 6 months of completing, their last treatment, and have an ECOG performance status of 0 or 1. Enrollment is up to approximately 415 pts.
The study consists of two parts: dose finding for combination agents (phase 1) and dose expansion at the recommended phase 2 dose (RP2D) determined in phase 1 (phase 2). Pts are enrolled into 3 treatment arms (non-randomized allocation; Figure): arm A, ide-cel + iberdomide (± dexamethasone), in cohorts 1 and 2 (per pt population), as maintenance therapy (subcohort a), or as both concurrent and maintenance therapy (subcohorts b and c; schedules differ); arm B, ide-cel + BMS-986405 as concurrent therapy; arm C, ide-cel + DPdor PVd as maintenance therapy. Following leukapheresis, bridging therapy may be administered subject to restrictions. Lymphodepletion with fludarabine (30 mg/m 2/day) and cyclosphosphamide (300 mg/m 2/day) occurs for 3 consecutive days followed by 2 rest days before ide-cel infusion. Ide-cel target dose is 450 x 10 6 (± 20%) CAR+ T cells for all study arms. Combination agents are prespecified to be administered before, concurrently with, and/or following ide-cel infusion.
Primary objectives include evaluation of safety (dose-limiting toxicity rate), RP2D determination, schedule optimization for combination agents administered with ide-cel (phase 1), and assessment of efficacy (CR rate) of combination regimens (phase 2). Secondary objectives are additional safety (adverse events) and efficacy (ORR, progression-free survival [PFS], OS, time to response, DOR, time to next treatment, PFS after next therapy) parameters, feasibility of maintenance therapy, and characterizing pharmacokinetics of ide-cel.
At the end of phase 1, interim analysis will be performed to determine the RP2D. The primary analysis for efficacy and safety will be in phase 2 when all pts have a minimum follow-up of 15 months. Pts will be followed for a maximum of 2 years after the last pt receives first combination agent, or after the last pt receives any study treatment, in the respective cohorts, whichever occurs later. Recruitment began in June 2021 and is ongoing in the United States and Spain.
Study support
bluebird bio and Celgene, a Bristol-Myers Squibb Company
Raje: Caribou: Other; Janssen: Other; bluebird bio: Other; Amgen: Other; Celgene: Other; BMS: Other. Berdeja: Poseida, Sanofi, Teva: Research Funding; Bluebird bio, BMS, Celgene, CRISPR Therapeutics, Janssen, Kite Pharma, Legend Biotech, SecuraBio, Takeda: Consultancy; Lilly, Novartis: Research Funding; Celularity, CRISPR Therapeutics: Research Funding; Abbvie, Acetylon, Amgen: Research Funding; EMD Sorono, Genentech: Research Funding; GSK, Ichnos Sciences, Incyte: Research Funding. Rodriguez-Otero: Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Clínica Universidad de Navarra: Current Employment; Celgene-BMS: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; GSK: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Pfizer: Consultancy; Sanofi: Honoraria, Membership on an entity's Board of Directors or advisory committees; Kite: Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Honoraria; Regeneron: Honoraria. Green: Seagen Inc.: Research Funding; bristol myers squibb: Membership on an entity's Board of Directors or advisory committees, Patents & Royalties, Research Funding; Cellectar Biosciences: Research Funding; GSK: Membership on an entity's Board of Directors or advisory committees; Janssen Biotech: Membership on an entity's Board of Directors or advisory committees, Research Funding; Juno Therapeutics: Patents & Royalties, Research Funding; Legend Biotech: Consultancy; Neoleukin Therapeutics: Membership on an entity's Board of Directors or advisory committees; Seattle Genetics: Membership on an entity's Board of Directors or advisory committees, Research Funding; SpringWorks Therapeutics: Research Funding. Jagannath: Bristol Myers Squibb: Consultancy; Janssen Pharmaceuticals: Consultancy; Legend Biotech: Consultancy; Karyopharm Therapeutics: Consultancy; Sanofi: Consultancy; Takeda: Consultancy. Lonial: Takeda: Consultancy, Honoraria, Research Funding; Merck: Honoraria; Abbvie: Consultancy, Honoraria; BMS/Celgene: Consultancy, Honoraria, Research Funding; AMGEN: Consultancy, Honoraria; Janssen: Consultancy, Honoraria, Research Funding; GlaxoSmithKline: Consultancy, Honoraria, Research Funding; TG Therapeutics: Membership on an entity's Board of Directors or advisory committees. Gipson: Bristol Myers Squibb: Current Employment. Caia: Bristol Myers Squibb: Current Employment. Martin: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Yang: Bristol Myers Squibb: Current Employment, Current holder of individual stocks in a privately-held company. Pittari: Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Mateos: Sea-Gen: Honoraria, Membership on an entity's Board of Directors or advisory committees; Roche: Honoraria, Membership on an entity's Board of Directors or advisory committees; Oncopeptides: Honoraria, Membership on an entity's Board of Directors or advisory committees; Pfizer: Honoraria, Membership on an entity's Board of Directors or advisory committees; GSK: Honoraria; Regeneron: Honoraria, Membership on an entity's Board of Directors or advisory committees; Sanofi: Honoraria, Membership on an entity's Board of Directors or advisory committees; Bluebird bio: Honoraria; Oncopeptides: Honoraria; AbbVie: Honoraria; Celgene - Bristol Myers Squibb: Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees; Adaptive Biotechnologies: Honoraria, Membership on an entity's Board of Directors or advisory committees; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees.
Neither iberdomide nor BMS-986405 are approved, and all combinations of agents with ide-cel are off-label. These drugs and combinations of drugs are being investigated for treatment of relapsed/refractory multiple myeloma.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal