In this issue of Blood, Mittelman et al1 and Pagano et al2 show that patients with hematologic malignancies who are fully or partially vaccinated are at higher risk of poor clinical outcomes after COVID-19 infection compared with a matched control group or the general population.
Although it had already been established that the serologic response to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) can be impaired in ∼25% of patients with hematologic malignancies,3 the clinical implications of this were unknown before the studies by Mittelman et al and Pagano et al. Nevertheless, a harbinger of trouble had already appeared based on the remarkable 34% risk of death among adult patients with blood cancer infected with COVID-19 observed shortly after the pandemic began but before vaccines became available in December 2020.4
Both studies make inroads into the missing link between immune response and clinical outcomes by taking advantage of data from a coordinated health care system in Israel1 or across 42 hematology departments in European and non-European countries2 where most patients received two (full) doses of an mRNA vaccine. Clinical outcomes were followed either up to 63 days after administration of the first dose of the vaccine1 or a median of 64 days after the last dose of the vaccine and COVID-19 diagnosis.2
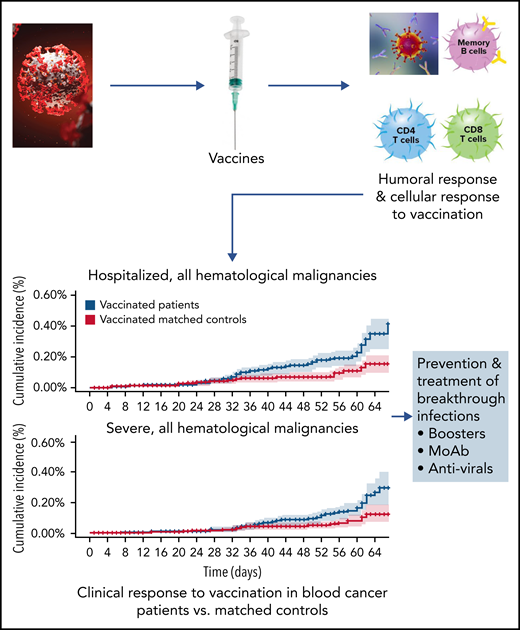
Mittelman et al compared vaccine efficacy in those with blood cancer with that in matched controls (without blood cancer); the analysis focused on COVID-19 outcomes from days 7 to 43 after the second vaccine dose (see figure). They examined outcomes in 32 516 vaccinated patients with blood cancer compared with outcomes in an equal number of control participants. Among all patients with hematologic malignancies, the relative risk (RR), compared with that in matched controls, for infection was 1.60 (95% confidence interval [CI], 1.12-2.37); for symptomatic COVID-19, 1.72l (95% CI, 1.05-2.85); for COVID-19 hospitalization, 3.13 (95% CI, 1.68-7.08); for severe COVID-19, 2.27 (95% CI, 1.18-5.19); and for COVID-19–related death, 1.66 (95% CI, 0.72-4.47). The RR increased when 5107 patients who were receiving active treatment for their disease were compared.
Progression of SARS-CoV-2 vaccination investigation in patients with hematologic malignancies compared with control-matched population. Moab, monoclonal antibody. The figure has been adapted from Figure 2 in the article by Mittelman et al that begins on page 1439.
Progression of SARS-CoV-2 vaccination investigation in patients with hematologic malignancies compared with control-matched population. Moab, monoclonal antibody. The figure has been adapted from Figure 2 in the article by Mittelman et al that begins on page 1439.
Pagano et al reported 113 COVID-19 infections from January to August 2021 among partially (23%) or fully (77%) vaccinated patients. Approximately 70% of these patients were undergoing active treatment for underlying hematologic malignancies. Sixty percent of the patients infected with COVID-19 had severe or critical infections. After follow-up of 30 days post–COVID-19 diagnosis, the overall mortality rate was 12.4% (n = 14). Further analysis was performed in 40 of the 113 patients to examine the correlation between serologic response and breakthrough infection. Approximately 70% of the patients did not generate an antibody response to the vaccine. The authors concluded that low serologic response rates to SARS-CoV-2 vaccines may be correlated with higher rates of infection in patients with hematologic malignancies, although there was no correlation with mortality resulting from COVID-19 breakthrough infection.
These studies indicate that there is a clear increased risk of significant breakthrough infection immediately after vaccination in patients with hematologic malignancies that is higher than the risk in the general population. This outcome requires public health guidance for patients with blood cancer.
These studies do have important limitations. First, the data were generated before the delta or omicron variants of COVID-19 became the prevalent viral strains. Second, the incidence of infection was too small to statistically examine the outcomes of patients with specific types of blood cancer. Finally, although clinical outcomes of patients with hematologic malignancies can be worse than those of the normal population, the causative basis for impaired immune response remains an open question for future investigations.
What do we know about the immune response to SARS-CoV-2 vaccination in patients with blood cancer? Antispike antibody assessment can be easily performed in the clinic with commercial assays; however, the functional assessment of neutralizing antibodies is more meaningful, although there is a moderate correlation between anti-S antibodies and neutralizing antibody production.5 Collectively, these studies show that the highest percentage of seronegative patients have B cell–derived malignancies, such as chronic lymphocytic leukemia or non-Hodgkin lymphoma.3 This can be due to the disease itself, as well as to B cell–suppressive therapies, including anti-CD20 antibodies, Bruton tyrosine kinase inhibitors, and CD19 chimeric antigen receptor T-cell therapy. The adequate serologic response associated with protection from COVID-19 infection remains to be defined.
In contrast, we know less about the cellular response to COVID-19 infection or vaccination mediated by T cells. We know that the production of a higher number of CD8+ cells is associated with improved survival in COVID-19–infected patients who have hematologic malignancies.6 Emerging data suggest that although the T-cell response to vaccination can be muted in patients with blood cancer compared with in matched controls, many patients produce COVID-19–killing T cells in response to vaccination.7,8 Those patients who have no antispike antibodies or neutralizing antibodies, as well as no functional T cells, in response to vaccination are expected to be at the highest risk of future infection.
Even if we do not fully understand the mechanistic link between immune response and patient outcome after vaccination, therapies are needed for patients at high risk of COVID-19 infection and/or poor clinical outcomes as well as patients who develop infections after vaccination. One simple solution is to make sure all patients with blood cancer get third vaccinations, because in a preliminary study, ∼50% of the patients who were seronegative after initial vaccination seroconverted after booster vaccination.8 In addition, monoclonal antibody cocktails have already received emergency use authorization (EUA) by the US Food and Drug Administration (FDA) for the treatment of patients with COVID-19 infection9 or for patients treated postexposure to COVID-19 (FDA EUA, 16 September 2021). Evusheld has received an EUA to treat high-risk patients prophylactically preexposure. The likely candidates for monoclonal antibody therapy include immunocompromised patients with hematologic malignancies, especially those receiving active treatment or at high risk of COVID-19 infection.
In summary, these studies are a clear warning sign that patients with hematologic diseases may be vulnerable to breakthrough infection that results in poor outcomes. Patients need to get vaccinated but act unvaccinated, and they need to make certain everyone around them is vaccinated.
Conflict-of-interest disclosure: The authors declare no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal