Key Points
This is the largest cohort of transplanted adolescent and adult IEI patients studied to date, detailing outcomes for 329 patients.
This study demonstrates OS and EFS and identifies adverse risk factors to help identify patients most likely to benefit from HSCT.
Abstract
Allogeneic hematopoietic stem cell transplantation (HSCT) is the gold standard curative therapy for infants and children with many inborn errors of immunity (IEI), but adolescents and adults with IEI are rarely referred for transplant. Lack of published HSCT outcome data outside small, single-center studies and perceived high risk of transplant-related mortality have delayed the adoption of HSCT for IEI patients presenting or developing significant organ damage later in life. This large retrospective, multicenter HSCT outcome study reports on 329 IEI patients (age range, 15-62.5 years at HSCT). Patients underwent first HSCT between 2000 and 2019. Primary endpoints were overall survival (OS) and event-free survival (EFS). We also evaluated the influence of IEI-subgroup and IEI-specific risk factors at HSCT, including infections, bronchiectasis, colitis, malignancy, inflammatory lung disease, splenectomy, hepatic dysfunction, and systemic immunosuppression. At a median follow-up of 44.3 months, the estimated OS at 1 and 5 years post-HSCT for all patients was 78% and 71%, and EFS was 65% and 62%, respectively, with low rates of severe acute (8%) or extensive chronic (7%) graft-versus-host disease. On univariate analysis, OS and EFS were inferior in patients with primary antibody deficiency, bronchiectasis, prior splenectomy, hepatic comorbidity, and higher hematopoietic cell transplant comorbidity index scores. On multivariable analysis, EFS was inferior in those with a higher number of IEI-associated complications. Neither age nor donor had a significant effect on OS or EFS. We have identified age-independent risk factors for adverse outcome, providing much needed evidence to identify which patients are most likely to benefit from HSCT.
Introduction
Inborn errors of immunity (IEI) are a wide group of genetically determined, rare, and complex lifelong diseases.1 The majority are characterized by severe infection and immune dysregulation including autoimmunity, autoinflammation, and an increased risk of malignancy. In children with IEI, survival and cure rates after allogeneic hematopoietic stem cell transplantation (HSCT) are now excellent, particularly in the absence of organ damage and/or infectious burden at transplant.2-5 Consequently, there is a steady increase in HSCT activity for these patients.6
HSCT for older adolescents and adults with IEI was uncommon until recently, mainly due to perceived high risks of transplant-related mortality, and was reserved as a last resort for patients with excessive morbidity.7-9 Indeed, initial publications on HSCT for adults with common variable immunodeficiency (CVID) reported disappointing survival rates of ∼50% in patients who had acquired significant organ damage pre-HSCT.10 In contrast, adolescents and very young adults with limited IEI-related morbidity can expect excellent outcomes very similar to their pediatric counterparts.11,12 Moreover, when using reduced toxicity conditioning regimens in specialized centers, even older adult IEI patients with advanced disease can expect good survival probabilities and improvement of quality of life after HSCT.12,13 Consequently, adults with IEI have recently been added to the list of indications for HSCT by the European Society for Blood and Marrow Transplantation (EBMT) and American Society for Transplantation and Cellular Therapy (ASTCT).14-16 To date, all published retrospective4,10-12 and prospective13,17 studies on HSCT outcomes in adults with IEI taken together report on <150 patients.
In children with genetically confirmed IEI known to have a poor prognosis with conservative therapy alone, the role of HSCT is usually well defined and supported by clear evidence for transplant efficacy.4 However, the decision to proceed to HSCT is challenging in older patients who may have a milder disease course during childhood or late onset presentation, in diseases with variable phenotypic severity/penetrance, or poorly documented natural history and in the absence of a genetic diagnosis.18 Further complexity is added by issues including fertility and family planning, psychosocial wellbeing, access to targeted therapies, and the need for careful donor and conditioning regimen selection, necessitating specialized interdisciplinary care.9,19
The hematopoietic cell transplant comorbidity index (HCT-CI) score has been validated as a predictive marker for survival in patients with nonmalignant diseases,20 but IEI-specific risk factors still need to be identified for this group. We undertook a multicenter retrospective analysis of the EBMT registry to report HSCT outcomes for adolescents and adults with IEI in the last 2 decades. Overall survival (OS) and event-free survival (EFS) were reported, and IEI-specific risk factors for poor outcomes were identified to aid patient selection for HSCT.
Patients and methods
Data source
This international, multicenter retrospective analysis (study number 8427018) was performed on behalf of the EBMT and European Society for Immunodeficiencies Inborn Errors Working Party. EBMT centers commit to obtaining informed consent according to the local regulations when reporting pseudonymized data to the EBMT. The EBMT registry provided HSCT data, and a study-specific questionnaire was completed by the study centers.
Study participants and transplant procedures
Eligibility criteria included age ≥15 years at HSCT, confirmed clinical and/or genetic diagnosis of IEI, and first HSCT between 2000 and 2019. The study cohort included 329 patients from 51 centers. Basic demographic data included age, sex, IEI subgroup, genetic diagnosis, age at diagnosis, age at HSCT, time from diagnosis to HSCT, HCT-CI score,21 and year of HSCT. Transplant-specific demographics included donor type, cell source, conditioning intensity, use of serotherapy, and donor/recipient CMV serostatus.
Chimerism analysis
Chimerism samples were processed as per local center protocols and included either whole blood (WB) or lineage-specific analysis.
Definitions
Neutrophil and platelet recovery were defined as the first of 3 consecutive days with an absolute neutrophil count > 0.5 × 109/L, or an unsupported platelet count > 20 × 109/L. Graft failure was defined as the absence of neutrophil recovery on day +28 or donor chimerism in peripheral blood <5% beyond day +28.22
Grading of acute and chronic graft-versus-host disease (GVHD) was performed according to modified Seattle criteria and National Institutes of Health (NIH) consensus standards where available.17 Hepatic dysfunction at HSCT was graded as either mild or moderate/severe by the reporting physician.
In this study, conditioning intensity was defined as follows: (1) myeloablative (MA): regimens containing busulfan doses >12 mg/kg and/or total area under the curve (AUC) >80 000 ng times h/mL, fludarabine plus melphalan plus either treosulfan or carmustine (BCNU); (2) reduced toxicity conditioning (RTC): busulfan ≤12 mg/kg or AUC ≤ 80 000 ng times h/mL, treosulfan-based unless combined with melphalan, fludarabine and melphalan, and (3) nonmyeloablative (NMA): fludarabine plus cyclophosphamide, TBI 2Gy plus fludarabine.
To facilitate outcome analysis based on IEI subgroups, 3 groups were established based on the most recent International Union of Immunological Societies (IUIS) classification of IEI1: phagocyte disorders/innate (PD), combined immunodeficiencies (CID), and predominantly antibody defects (PAD). For detailed information of underlying diagnoses, see supplemental Table 1.
Statistical analysis
Patient and transplant characteristics were expressed as the number and percentage of the group for categorical variables and median with ranges for continuous variables. The time origin for time-to-event analysis was first HSCT, and patients alive without an event after transplant were censored at last follow-up or time of data extraction.
Primary endpoints were OS, where an event was defined as death of any cause, and EFS, where an event was defined as graft failure (GF), moderate to severe chronic GVHD (cGVHD), or death, whichever happened first.
Secondary endpoints evaluated the influence on OS and EFS of the IEI subgroup, HCT-CI score, age at HSCT, and additional IEI-specific risk factors at the time of HSCT, including infections, low body mass index (BMI) (<18), bronchiectasis, colitis, malignancy, granulomatous lymphocytic inflammatory lung disease (GLILD), splenectomy, hepatic dysfunction, autoimmunity and systemic immunosuppressive therapy. Peripheral blood chimerism, requirement for immunoglobulin replacement therapy (IgRT), performance status, attendance of work or school, and conception at last follow-up were also documented.
OS and EFS were estimated using the Kaplan-Meier product limit estimation method, and differences in subgroups were assessed by the log-rank test. Median follow-up was determined using the reverse Kaplan-Meier method. Competing risks analyses were applied to estimate the incidences of acute grade 2 to 4 GVHD and limited and extensive cGVHD by day 100 and 1 and 2 years, respectively. The competing events were second transplant, graft failure, or nonengraftment and death. Subgroup differences in cumulative incidences were assessed using Gray’s test. Competing risks analyses were also applied to estimate incidences of neutrophil engraftment and platelet recovery, each with death as the competing event. Multivariable Cox regression was applied to investigate the simultaneous impact of multiple covariates on outcomes when a sufficient number of patients and subsequent events were available. For OS and EFS, hazard ratios (HRs) are provided. Both models use the same covariate structure: IEI subgroup (PAD, PD vs CID), number of pre-HSCT IEI complications (1 complication, ≥2 complications vs no complications), age at HSCT (HR per decade increase in age), Sorror score (HCT-CI 1-2, HCT-CI > 2 vs HCT-CI 0), and conditioning intensity (MA, RTC vs NMA). Significance of individual HR was determined by means of the Wald test. Likelihood ratio tests were performed to determine overall significance of covariates. Based on the number of events, there were a limited number of covariates which could be tested; these were selected based on clinical relevance.
All survival estimates and HRs are reported with corresponding 95% confidence intervals (CIs) in parentheses. All P values were 2-sided, and P < .05 was considered significant. Statistical analyses were performed in R version 3.6.0 (R Development Core Team, Vienna, Austria) using packages “survival,” “prodlim,” and “cmprsk.”
Results
Patient and HSCT characteristics
Three hundred twenty-nine patients (223 males and 106 females) were included in this study, and detailed patient demographics are shown in Table 1.
Patient demographics
| . | Total (N = 329) . | |
|---|---|---|
| Missing (%) . | N (%)∗ . | |
| Patient sex | ||
| Male | 0 | 223 (67.8) |
| Female | 0 | 106 (32.2) |
| IEI subgroup | ||
| CID | 0 | 163 (49.5) |
| PD | 0 | 127 (38.6) |
| PAD | 0 | 39 (11.9) |
| Age at HSCT (y) | ||
| Median (IQR) | 0 | 18.4 (16.6-22.8) |
| Time from clinical diagnosis to HSCT (mo) | ||
| Median (IQR) | 1 (0.3) | 63.1 (13-189.1) |
| Age at clinical diagnosis (y) | ||
| Median (IQR) | 3 (0.9) | 13 (3.6-17.2) |
| BMI at HSCT | ||
| Low BMI (<18) | 34 (10.3) | 74 (25.1) |
| Normal/high BMI (≥18) | 221 (74.9) | |
| Infection immediately prior to HSCT | ||
| No | 45 (13.7) | 136 (47.9) |
| Yes | 148 (52.1) | |
| Bronchiectasis at HSCT | ||
| Absent | 5 (1.5) | 238 (73.5) |
| Present | 86 (26.5) | |
| Colitis or protracted diarrhea at HSCT | ||
| Absent | 4 (1.2) | 255 (78.5) |
| Present | 70 (21.5) | |
| Malignancy prior to HSCT† | ||
| No | 5 (1.5) | 263 (80.9) |
| Yes | 62 (19.1) | |
| Remission status at HSCT (if prior malignancy) | ||
| No remission | 4 (5.6) | 23 (39.7) |
| Remission | 33 (56.9) | |
| Not evaluated | 2 (3.4) | |
| GLILD at HSCT | ||
| No | 18 (5.5) | 268 (86.2) |
| Yes | 43 (13.8) | |
| Splenectomy prior to HSCT | ||
| No | 14 (4.3) | 285 (90.5) |
| Yes | 30 (9.5) | |
| Hepatic comorbidity | ||
| No | 54 (16.4) | 239 (86.9) |
| Mild | 22 (8) | |
| Moderate/severe | 14 (5.1) | |
| IEI-associated complications (bronchiectasis, colitis, malignancy, GLILD, hepatic comorbidity, and splenectomy) | ||
| None | 56 (17) | 90 (33) |
| One complication | 99 (36.3) | |
| Two or more complications | 84 (30.8) | |
| HCT-CI (Sorror score) | ||
| 0 | 7 (2.1) | 89 (27.6) |
| 1-2 | 128 (39.8) | |
| 3-4 | 59 (18.3) | |
| >4 | 46 (14.3) | |
| . | Total (N = 329) . | |
|---|---|---|
| Missing (%) . | N (%)∗ . | |
| Patient sex | ||
| Male | 0 | 223 (67.8) |
| Female | 0 | 106 (32.2) |
| IEI subgroup | ||
| CID | 0 | 163 (49.5) |
| PD | 0 | 127 (38.6) |
| PAD | 0 | 39 (11.9) |
| Age at HSCT (y) | ||
| Median (IQR) | 0 | 18.4 (16.6-22.8) |
| Time from clinical diagnosis to HSCT (mo) | ||
| Median (IQR) | 1 (0.3) | 63.1 (13-189.1) |
| Age at clinical diagnosis (y) | ||
| Median (IQR) | 3 (0.9) | 13 (3.6-17.2) |
| BMI at HSCT | ||
| Low BMI (<18) | 34 (10.3) | 74 (25.1) |
| Normal/high BMI (≥18) | 221 (74.9) | |
| Infection immediately prior to HSCT | ||
| No | 45 (13.7) | 136 (47.9) |
| Yes | 148 (52.1) | |
| Bronchiectasis at HSCT | ||
| Absent | 5 (1.5) | 238 (73.5) |
| Present | 86 (26.5) | |
| Colitis or protracted diarrhea at HSCT | ||
| Absent | 4 (1.2) | 255 (78.5) |
| Present | 70 (21.5) | |
| Malignancy prior to HSCT† | ||
| No | 5 (1.5) | 263 (80.9) |
| Yes | 62 (19.1) | |
| Remission status at HSCT (if prior malignancy) | ||
| No remission | 4 (5.6) | 23 (39.7) |
| Remission | 33 (56.9) | |
| Not evaluated | 2 (3.4) | |
| GLILD at HSCT | ||
| No | 18 (5.5) | 268 (86.2) |
| Yes | 43 (13.8) | |
| Splenectomy prior to HSCT | ||
| No | 14 (4.3) | 285 (90.5) |
| Yes | 30 (9.5) | |
| Hepatic comorbidity | ||
| No | 54 (16.4) | 239 (86.9) |
| Mild | 22 (8) | |
| Moderate/severe | 14 (5.1) | |
| IEI-associated complications (bronchiectasis, colitis, malignancy, GLILD, hepatic comorbidity, and splenectomy) | ||
| None | 56 (17) | 90 (33) |
| One complication | 99 (36.3) | |
| Two or more complications | 84 (30.8) | |
| HCT-CI (Sorror score) | ||
| 0 | 7 (2.1) | 89 (27.6) |
| 1-2 | 128 (39.8) | |
| 3-4 | 59 (18.3) | |
| >4 | 46 (14.3) | |
IQR, interquartile ranges.
Percentages in this column are calculated from the nonmissing.
All patients had established diagnosis of IEI prior to development of malignancy.
The median age at HSCT was 18.4 years (IQR, 16.6-22.8; range, 15-62.5), with a median age at clinical diagnosis of 13 years (3.6-17.2; 0-62.1). The median time from clinical diagnosis to HSCT was 5.3 years months (1.1-15.8; 0-39.3). Median age at last follow-up for the whole cohort was 23.2 years (19.5-28.3; 15.2-62.6).
Underlying IEI diagnoses (genetically and/or clinically confirmed) were grouped into CID (49.5%), PD (38.6%), and PAD (11.9%), with further details shown in supplemental Table 1. Within the CID subgroup (n = 163), the most frequent diagnoses were genetically undefined CID (n = 37), hemophagocytic lymphohistiocytosis (HLH) (n = 23), CD40L deficiency (n = 14), Wiskott-Aldrich syndrome (WAS) (n = 13), and DOCK8 deficiency (n = 12). Within the PD subgroup, the most frequent diagnoses were chronic granulomatous disease (CGD) (n = 83), GATA2 deficiency (n = 25), and SCN (n = 12). The PAD subgroup comprised 27 patients with CVID, 9 with activated PI3K delta syndrome (APDS), and 3 with X-linked agammaglobulinaemia (XLA). Overall, the underlying disease remained genetically undefined in a total of 63 patients (19.1%, 37 CID and 26 PAD) at the time of writing.
IEI-related complications present at or immediately prior to HSCT are described in Table 1. Most common were: infection (52% of whole cohort); bronchiectasis (26.5%); colitis or protracted diarrhea (27.0%), malignancy (19.1%), GLILD (13.8%), hepatic comorbidity (13.1%), and prior splenectomy (9.5%).
Approximately one third of the cohort (35.2%) were on systemic immunomodulatory therapies immediately prior to HSCT for autoimmunity or inflammatory IEI–related complications. These included 32.5% of the CID subgroup, 48.7% of the PAD subgroup, and 31.5% of the PD subgroup. Details of immunomodulatory therapies are given in supplemental Table 2. For those patients (19.1% of whole cohort) with a documented malignancy prior to HSCT, 56.9% were in remission at the time of HSCT. Details of malignant diagnoses are given in supplemental Table 3.
One-third (33%) of the total cohort had no active IEI-related complications at the time of HSCT, 36.3% had 1 complication, and 30.8% of patients had 2 or more complications. Reflecting the comorbidities associated with IEI, patients in our cohort had HCT-CI scores of 0, 1 to 2, 3 to 4, or ≥4 in 27.6%, 39.8%, 18.3%, and 14.3%, respectively (Table 1). Some important differences in IEI-related complications were observed between IEI subgroups. Hepatic comorbidity was observed more frequently in patients with PAD (19.4% mild, 5.6% moderate/severe) than in patients with CID (8.1% mild, 8.1% moderate/severe) and PD (3.8% mild, 1% moderate/severe), P = .003. Splenectomy prior to HSCT was observed more frequently in patients with CID (15.8%) and PAD (11.4%) than in patients with PD (0.8%), P ≤ .001. Bronchiectasis at HSCT was observed more frequently in patients with PAD (43.2%) than in patients with CID (26.5%) and PD (21.6%), P = .032 (Table 2).
Distribution of IEI-related comorbidities and age at HSCT by IEI subgroup
| . | Group . | CID . | PAD . | PD . | P value . | |||
|---|---|---|---|---|---|---|---|---|
| Median (IQR) . | Median (IQR) . | Median (IQR) . | ||||||
| Age at HSCT | Median (IQR) | 17.6 (16.4-21.2) | 22.8 (17.4-37.9) | 18.9 (16.8-22.5 | .001 | |||
| Missing (%) | N (%) | Missing (%) | N (%) | Missing (%) | N (%) | P | ||
| Hepatic comorbidity | No | 28 (17.2) | 113 (83.7) | 3 (7.7) | 27 (75.0) | 23 (18.1) | 99 (95.2) | .003 |
| Mild | 11 (8.1) | 7 (19.4) | 4 (3.8) | |||||
| Moderate/severe | 11 (8.1) | 2 (5.6) | 1 (1.0) | |||||
| Splenectomy prior to HSCT | No | 5 (3.1) | 133 (84.2) | 4 (10.3) | 31 (88.6) | 5 (3.9) | 121 (99.2) | <.001 |
| Yes | 25 (15.8) | 4 (11.4) | 1 (0.8) | |||||
| Bronchiectasis at HSCT | Absent | 1 (0.6) | 119 (73.5) | 2 (5.1) | 21 (56.8) | 2 (1.6) | 98 (78.4) | .032 |
| Present | 43 (26.5) | 16 (43.2) | 27 (21.6) | |||||
| HCT-CI score | 0 | 5 (3.1) | 51 (32.3) | 1 (2.6) | 9 (23.7) | 1 (0.8) | 29 (23.0) | .096 |
| 1-2 | 56 (35.4) | 12 (31.6) | 60 (47.6) | |||||
| >2 | 51 (32.3) | 17 (44.7) | 37 (29.4) | |||||
| . | Group . | CID . | PAD . | PD . | P value . | |||
|---|---|---|---|---|---|---|---|---|
| Median (IQR) . | Median (IQR) . | Median (IQR) . | ||||||
| Age at HSCT | Median (IQR) | 17.6 (16.4-21.2) | 22.8 (17.4-37.9) | 18.9 (16.8-22.5 | .001 | |||
| Missing (%) | N (%) | Missing (%) | N (%) | Missing (%) | N (%) | P | ||
| Hepatic comorbidity | No | 28 (17.2) | 113 (83.7) | 3 (7.7) | 27 (75.0) | 23 (18.1) | 99 (95.2) | .003 |
| Mild | 11 (8.1) | 7 (19.4) | 4 (3.8) | |||||
| Moderate/severe | 11 (8.1) | 2 (5.6) | 1 (1.0) | |||||
| Splenectomy prior to HSCT | No | 5 (3.1) | 133 (84.2) | 4 (10.3) | 31 (88.6) | 5 (3.9) | 121 (99.2) | <.001 |
| Yes | 25 (15.8) | 4 (11.4) | 1 (0.8) | |||||
| Bronchiectasis at HSCT | Absent | 1 (0.6) | 119 (73.5) | 2 (5.1) | 21 (56.8) | 2 (1.6) | 98 (78.4) | .032 |
| Present | 43 (26.5) | 16 (43.2) | 27 (21.6) | |||||
| HCT-CI score | 0 | 5 (3.1) | 51 (32.3) | 1 (2.6) | 9 (23.7) | 1 (0.8) | 29 (23.0) | .096 |
| 1-2 | 56 (35.4) | 12 (31.6) | 60 (47.6) | |||||
| >2 | 51 (32.3) | 17 (44.7) | 37 (29.4) | |||||
Transplant characteristics are described in detail in Table 3. Most patients underwent MUD or MMUD transplants (n = 200, 62.1%).
Transplant characteristics
| . | Donor type . | N (%)∗ . | . | Graft manipulation . | N (%)∗ . |
|---|---|---|---|---|---|
| Missing: 7 (2.1%) | MRD | 103 (32) | Missing: 1 (0.3%) | Yes | 31 (9.5) |
| MUD (10/10) | 136 (42.2) | No | 297 (90.5) | ||
| MMUD (9/10) | 56 (17.4) | ||||
| MMUD (<9/10) | 8 (2.5) | TCR α-β depletion | 16 | ||
| UCB | 1 (0.3) | CD34+ selection | 10 | ||
| MMRD (Haplo) | 18 (5.8) | Other | 5 | ||
| Stem cell source | Conditioning regimen groups | ||||
| Missing: 1 (0.3%) | BM | 151 (46) | Missing: 2 (0.6%) | Busulfan-based | 133 (40.4) |
| PB | 169 (51.5) | Treosulfan-based | 86 (26.1) | ||
| BM+PB | 1 (0.3) | Melphalan-based | 74 (22.5) | ||
| CB† | 4 (1.2) | Other | 34 (10.4) | ||
| BM+CB | 1 (0.3) | ||||
| PB+CB | 2 (0.6) | ||||
| Conditioning regimen intensity | Most commonly used conditioning regimens | ||||
| Missing: 13 (4%) | Myeloablative | 82 (25.9) | Fludarabine, busulfan | 97 | |
| Reduced toxicity | 208 (65.8) | Fludarabine, melphalan | 56 | ||
| Nonmyeloablative | 26 (8.2) | Fludarabine, treosulfan | 42 | ||
| Fludarabine, treosulfan, thiotepa | 37 | ||||
| Busulfan, cyclophosphamide | 17 | ||||
| Fludarabine, cyclophosphamide | 13 | ||||
| Fludarabine, melphalan, thiotepa | 11 | ||||
| Fludarabine, busulfan, thiotepa | 7 | ||||
| Fludarabine, melphalan, BCNU | 7 | ||||
| Fludarabine, thiotepa | 5 | ||||
| Fludarabine | 5 | ||||
| Other | 30 | ||||
| Serotherapy | |||||
| Missing: 2 (0.6%) | Alemtuzumab | 138 (42.2) | |||
| ATG/ALG | 109 (33.3) | ||||
| No serotherapy | 80 (24.5) | ||||
| CMV match (donor/recipient) | |||||
| Missing: 69 (21%) | −/− | 74 (28.5) | |||
| −/+ | 18 (6.9) | ||||
| +/− | 60 (23.1) | ||||
| +/+ | 108 (41.5) | ||||
| Year of HSCT | |||||
| Median (IQR) | 2014 (2010-2016) | ||||
| . | Donor type . | N (%)∗ . | . | Graft manipulation . | N (%)∗ . |
|---|---|---|---|---|---|
| Missing: 7 (2.1%) | MRD | 103 (32) | Missing: 1 (0.3%) | Yes | 31 (9.5) |
| MUD (10/10) | 136 (42.2) | No | 297 (90.5) | ||
| MMUD (9/10) | 56 (17.4) | ||||
| MMUD (<9/10) | 8 (2.5) | TCR α-β depletion | 16 | ||
| UCB | 1 (0.3) | CD34+ selection | 10 | ||
| MMRD (Haplo) | 18 (5.8) | Other | 5 | ||
| Stem cell source | Conditioning regimen groups | ||||
| Missing: 1 (0.3%) | BM | 151 (46) | Missing: 2 (0.6%) | Busulfan-based | 133 (40.4) |
| PB | 169 (51.5) | Treosulfan-based | 86 (26.1) | ||
| BM+PB | 1 (0.3) | Melphalan-based | 74 (22.5) | ||
| CB† | 4 (1.2) | Other | 34 (10.4) | ||
| BM+CB | 1 (0.3) | ||||
| PB+CB | 2 (0.6) | ||||
| Conditioning regimen intensity | Most commonly used conditioning regimens | ||||
| Missing: 13 (4%) | Myeloablative | 82 (25.9) | Fludarabine, busulfan | 97 | |
| Reduced toxicity | 208 (65.8) | Fludarabine, melphalan | 56 | ||
| Nonmyeloablative | 26 (8.2) | Fludarabine, treosulfan | 42 | ||
| Fludarabine, treosulfan, thiotepa | 37 | ||||
| Busulfan, cyclophosphamide | 17 | ||||
| Fludarabine, cyclophosphamide | 13 | ||||
| Fludarabine, melphalan, thiotepa | 11 | ||||
| Fludarabine, busulfan, thiotepa | 7 | ||||
| Fludarabine, melphalan, BCNU | 7 | ||||
| Fludarabine, thiotepa | 5 | ||||
| Fludarabine | 5 | ||||
| Other | 30 | ||||
| Serotherapy | |||||
| Missing: 2 (0.6%) | Alemtuzumab | 138 (42.2) | |||
| ATG/ALG | 109 (33.3) | ||||
| No serotherapy | 80 (24.5) | ||||
| CMV match (donor/recipient) | |||||
| Missing: 69 (21%) | −/− | 74 (28.5) | |||
| −/+ | 18 (6.9) | ||||
| +/− | 60 (23.1) | ||||
| +/+ | 108 (41.5) | ||||
| Year of HSCT | |||||
| Median (IQR) | 2014 (2010-2016) | ||||
ATG/ALG, antithymocyte globulin/antilymphocyte globulin; BCNU, carmustine; BM, bone marrow; CB, cord blood; Haplo, haploidentical; MAbs, monoclonal antibodies; MMRD, mismatched-related donor; MMUD, mismatched unrelated donor; MRD, matched-related donor; MUD, matched unrelated donor; PB, peripheral blood; TCR, T-cell receptor; UCB, unrelated umbilical cord blood.
Percentages in this column are calculated from the nonmissing.
One unrelated cord blood, all others from related donors.
Engraftment and graft failure
Median time to neutrophil engraftment was 18 days (95% CI, 17-19 days), with 87% (83% to 91%) of patients achieving neutrophil engraftment by 28 days. The median time to platelet recovery was 18 days (17-20), with 72% (66% to 77%) of patients achieving platelet engraftment by 28 days.
GF was observed in 28 patients (8%). These were categorized as primary in 12 and secondary in 16. For those patients developing GF (n = 28), this was not related to IEI subgroups (P = .204, data not shown). The incidence of primary and secondary graft failure, combined as a single endpoint (with second transplant and death as competing events), was not influenced by IEI subgroup, donor, stem cell source, or conditioning intensity, P = .15, P = .5, P = .4, and P = .07, respectively (supplemental Table 4).
Survival
Estimated OS and EFS at 6 months, 1 year, and 5 years for the whole cohort and IEI subgroups are shown in Table 4.
Estimated OS and EFS for whole cohort and IEI subgroups
| Group . | N . | OS . | N . | EFS . | ||||
|---|---|---|---|---|---|---|---|---|
| 6 mo (95% CI) . | 1 y (95% CI) . | 5 y (95% CI) . | 6 mo (95% CI) . | 1 y (95% CI) . | 5 y (95% CI) . | |||
| Whole cohort | 329 | 83% (79-87) | 78% (74-83) | 71% (66-76) | 319 | 70% (65-75) | 65% (60-71) | 62% (56-67) |
| CID | 163 | 80% (74-86) | 76% (69-83) | 68% (61-76) | 157 | 68% (61-75) | 64% (56-71) | 58% (50-67) |
| PD∗ | 127 | 93% (88-97) | 87% (81-93) | 78% (71-86) | 123 | 77% (70-85) | 72% (64-80) | 69% (61-77) |
| CGD alone† | 83 | 92% (86-98%) | 83% (75-91%) | 77% (67-86%) | 80 | 74% (64-83%) | 69% (59-79%) | 67% (57-78%) |
| PAD‡ | 39 | 64% (49-79) | 59% (44-74) | 59% (44-74) | 39 | 54% (38-69) | 51% (36-67) | 51% (36-67) |
| CVID alone§ | 27 | 56% (37-74) | 48% (29-67) | 48% (29-67) | 27 | 44% (26-63) | 41% (22-59) | 41% (22-59) |
| Group . | N . | OS . | N . | EFS . | ||||
|---|---|---|---|---|---|---|---|---|
| 6 mo (95% CI) . | 1 y (95% CI) . | 5 y (95% CI) . | 6 mo (95% CI) . | 1 y (95% CI) . | 5 y (95% CI) . | |||
| Whole cohort | 329 | 83% (79-87) | 78% (74-83) | 71% (66-76) | 319 | 70% (65-75) | 65% (60-71) | 62% (56-67) |
| CID | 163 | 80% (74-86) | 76% (69-83) | 68% (61-76) | 157 | 68% (61-75) | 64% (56-71) | 58% (50-67) |
| PD∗ | 127 | 93% (88-97) | 87% (81-93) | 78% (71-86) | 123 | 77% (70-85) | 72% (64-80) | 69% (61-77) |
| CGD alone† | 83 | 92% (86-98%) | 83% (75-91%) | 77% (67-86%) | 80 | 74% (64-83%) | 69% (59-79%) | 67% (57-78%) |
| PAD‡ | 39 | 64% (49-79) | 59% (44-74) | 59% (44-74) | 39 | 54% (38-69) | 51% (36-67) | 51% (36-67) |
| CVID alone§ | 27 | 56% (37-74) | 48% (29-67) | 48% (29-67) | 27 | 44% (26-63) | 41% (22-59) | 41% (22-59) |
PD including CGD.
Subset of PD.
PAD including CVID.
Subset of PAD.
OS
Two hundred thirty-seven patients were alive at last follow up (median 44.3 months post-HSCT) with an estimated OS at 1 year and 5 years post-HSCT of 78% and 71%, respectively, for the whole cohort (Figure 1A).
Kaplan-Meier estimated probabilities of OS at 5 years. (A) The estimated OS for the whole cohort was 78% (95% CI, 74% to 83%) at 1 year and 71% (66% to 76%) at 5 years post-HSCT. OS at both 1 and 5 years post-HSCT were influenced by (B) IEI subgroup, (C) the presence of bronchiectasis at HSCT, (D) splenectomy prior to HSCT, (E) hepatic comorbidity at HSCT, (F) the number of IEI-associated risk factors at HSCT (bronchiectasis, colitis, malignancy, GLILD, hepatic comorbidity, and splenectomy), (G) HCT-CI score, and (H) year of transplant. OS at both 1 and 5 years post-HSCT were not influenced by (I) age at HSCT. Estimated OS at 5 years (95% CI) are shown.
Kaplan-Meier estimated probabilities of OS at 5 years. (A) The estimated OS for the whole cohort was 78% (95% CI, 74% to 83%) at 1 year and 71% (66% to 76%) at 5 years post-HSCT. OS at both 1 and 5 years post-HSCT were influenced by (B) IEI subgroup, (C) the presence of bronchiectasis at HSCT, (D) splenectomy prior to HSCT, (E) hepatic comorbidity at HSCT, (F) the number of IEI-associated risk factors at HSCT (bronchiectasis, colitis, malignancy, GLILD, hepatic comorbidity, and splenectomy), (G) HCT-CI score, and (H) year of transplant. OS at both 1 and 5 years post-HSCT were not influenced by (I) age at HSCT. Estimated OS at 5 years (95% CI) are shown.
In univariate analysis, OS at 1 year and 5 years post-HSCT was significantly influenced by IEI subgroup being 93% and 78% for PD (n = 127), 76% and 68% for CID (n = 163), and 59% at both time points for PAD (n = 39), P = .007 (Figure 1B). The presence (n = 126) or absence (n = 37) of a genetic diagnosis in the CID group had no impact on OS, P = .5 (data not shown).
The presence of various individual IEI-related complications at the time of HSCT were shown to adversely affect OS at both 1 and 5 years post-HSCT, including bronchiectasis (P = .026), prior splenectomy (P = .008), and liver disease (P = .017), as shown in Figure 1C-E. We assessed the impact of the number of IEI-associated complications at the time of HSCT on OS, including bronchiectasis, colitis, malignancy, GLILD, hepatic comorbidity, and splenectomy. OS was significantly worse for patients with 2 or more complications at the time of HSCT, P = .02 (Figure 1F).
As in other adult HSCT cohorts, higher pretransplant HCT-CI scores (≥2) identified patients with inferior OS at 1 and 5 years post-HSCT, P = .01 (Figure 1G). Finally, the year of transplant significantly influenced outcome, and better OS was observed for those transplanted in 2014 or later compared with those transplanted before that, P = .017 (Figure 1H).
Neither age at HSCT (Figure 1I) nor donor type significantly influenced OS at 1 and 5 years post-HSCT, P = .177 and P = .5, respectively, although there was a trend for better OS in the younger patients. Univariate analysis for the whole cohort identified other factors that did not significantly impact OS, including conditioning intensity (P = .6), pre-HSCT infection (P = .2), active colitis at HSCT (P = .6), pre-HSCT malignancy (P = .8), and GLILD (P = .16), as shown in supplemental Figure 1. Neither prior immunosuppressive therapy nor remission status of malignancy (where present) influenced OS (data not shown).
Subsequent multivariate analysis identified no risk factors statistically significant for OS, but IEI subgroup and number of IEI complications had a trend to significance (Table 5).
Multivariable analyses of risk factors for OS and EFS
| Risk factor . | Group . | At risk (N) . | Events (N) . | HR (95% CI) . | P value . | LRT∗ . |
|---|---|---|---|---|---|---|
| OS | ||||||
| IEI subgroup | CID | 127 | 39 | 0.052 | ||
| PAD | 30 | 15 | 1.49 (0.78-2.86) | .2 | ||
| PD | 102 | 21 | 0.62 (0.36-1.07) | .09 | ||
| IEI complications | No complication | 88 | 18 | 0.062 | ||
| 1 complication | 91 | 24 | 1.02 (0.54-1.94) | .9 | ||
| ≥2 complications | 80 | 33 | 1.84 (0.98-3.48) | .06 | ||
| Age at HSCT | (decades) | 259 | 75 | 1.13 (0.87-1.46) | .4 | 0.378 |
| HCT-CI score | 0 | 68 | 17 | 0.103 | ||
| 1-2 | 105 | 23 | 0.87 (0.45-1.67) | .7 | ||
| >2 | 86 | 35 | 1.54 (0.82-2.9) | .18 | ||
| Conditioning intensity | MA | 69 | 26 | 0.32 | ||
| RTC | 169 | 44 | 0.69 (0.42-1.13) | .14 | ||
| NMA | 21 | 5 | 0.66 (0.25-1.76) | .4 | ||
| EFS | ||||||
| IEI subgroup | CID | 122 | 50 | 0.142 | ||
| PAD | 30 | 17 | 1.38 (0.76-2.48) | .3 | ||
| PD | 101 | 33 | 0.73 (0.47-1.16) | .18 | ||
| IEI complications | No complication | 86 | 27 | 0.05 | ||
| 1 complication | 88 | 32 | 1.06 (0.62-1.8) | .8 | ||
| ≥2 complications | 79 | 41 | 1.78 (1.04-3.04) | .035 | ||
| Age at HSCT | (decades) | 253 | 100 | 1.15 (0.92-1.44) | .2 | 0.249 |
| HCT-CI score | 0 | 64 | 23 | 0.774 | ||
| 1-2 | 104 | 38 | 0.92 (0.53-1.59) | 0.8 | ||
| > 2 | 85 | 39 | 1.09 (0.62-1.91) | 0.8 | ||
| Conditioning intensity | MA | 68 | 27 | 0.958 | ||
| RTC | 164 | 65 | 1.05 (0.66-1.66) | 0.8 | ||
| NMA | 21 | 8 | 0.96 (0.43-2.15) | 0.9 | ||
| Risk factor . | Group . | At risk (N) . | Events (N) . | HR (95% CI) . | P value . | LRT∗ . |
|---|---|---|---|---|---|---|
| OS | ||||||
| IEI subgroup | CID | 127 | 39 | 0.052 | ||
| PAD | 30 | 15 | 1.49 (0.78-2.86) | .2 | ||
| PD | 102 | 21 | 0.62 (0.36-1.07) | .09 | ||
| IEI complications | No complication | 88 | 18 | 0.062 | ||
| 1 complication | 91 | 24 | 1.02 (0.54-1.94) | .9 | ||
| ≥2 complications | 80 | 33 | 1.84 (0.98-3.48) | .06 | ||
| Age at HSCT | (decades) | 259 | 75 | 1.13 (0.87-1.46) | .4 | 0.378 |
| HCT-CI score | 0 | 68 | 17 | 0.103 | ||
| 1-2 | 105 | 23 | 0.87 (0.45-1.67) | .7 | ||
| >2 | 86 | 35 | 1.54 (0.82-2.9) | .18 | ||
| Conditioning intensity | MA | 69 | 26 | 0.32 | ||
| RTC | 169 | 44 | 0.69 (0.42-1.13) | .14 | ||
| NMA | 21 | 5 | 0.66 (0.25-1.76) | .4 | ||
| EFS | ||||||
| IEI subgroup | CID | 122 | 50 | 0.142 | ||
| PAD | 30 | 17 | 1.38 (0.76-2.48) | .3 | ||
| PD | 101 | 33 | 0.73 (0.47-1.16) | .18 | ||
| IEI complications | No complication | 86 | 27 | 0.05 | ||
| 1 complication | 88 | 32 | 1.06 (0.62-1.8) | .8 | ||
| ≥2 complications | 79 | 41 | 1.78 (1.04-3.04) | .035 | ||
| Age at HSCT | (decades) | 253 | 100 | 1.15 (0.92-1.44) | .2 | 0.249 |
| HCT-CI score | 0 | 64 | 23 | 0.774 | ||
| 1-2 | 104 | 38 | 0.92 (0.53-1.59) | 0.8 | ||
| > 2 | 85 | 39 | 1.09 (0.62-1.91) | 0.8 | ||
| Conditioning intensity | MA | 68 | 27 | 0.958 | ||
| RTC | 164 | 65 | 1.05 (0.66-1.66) | 0.8 | ||
| NMA | 21 | 8 | 0.96 (0.43-2.15) | 0.9 | ||
LRT, likelihood ratio test.
P value for: does this risk factor add information when the other risk factors are already in the model? (<.05 = yes).
EFS
EFS for the whole cohort at 1 and 5 years was 65% and 62%, respectively (Figure 2A), and was also affected by IEI subgroup being 72% and 69% for PD (n = 123), 64% and 58% for CID (n = 157), and 51% at both time points for PAD (n = 39), P = .023 (Figure 2B). Again, no impact of a genetic diagnosis was seen within the CID group, P > .99 (data not shown). The presence of bronchiectasis, prior splenectomy, and hepatic comorbidity at HSCT adversely affected EFS at both 1 and 5 years post-HSCT, P = .021, P = .033, and P = .01, respectively (Figure 2C-E). EFS was also influenced by the number of IEI-associated risk factors present at HSCT, P = .029 (Figure 2F).
Kaplan-Meier estimated probabilities of EFS at 5 years. (A) The estimated EFS for the entire cohort was 65% (95% CI, 60% to 71%) at 1 year and 62% (56% to 57%) at 5 years post-HSCT. EFS at both 1 and 5 years post-HSCT were influenced by (B) IEI subgroup, (C) the presence of bronchiectasis at HSCT, (D) splenectomy prior to HSCT, and (E) the presence of hepatic dysfunction at HSCT. EFS at both 1 and 5 years post-HSCT were not influenced by (F) number of IEI-associated risk factors (bronchiectasis, colitis, malignancy, GLILD, hepatic comorbidity, and splenectomy), (G) HCT-CI score, (H) year of transplant, or (I) age at HSCT. Estimated EFS at 5 years (95% CI) are shown.
Kaplan-Meier estimated probabilities of EFS at 5 years. (A) The estimated EFS for the entire cohort was 65% (95% CI, 60% to 71%) at 1 year and 62% (56% to 57%) at 5 years post-HSCT. EFS at both 1 and 5 years post-HSCT were influenced by (B) IEI subgroup, (C) the presence of bronchiectasis at HSCT, (D) splenectomy prior to HSCT, and (E) the presence of hepatic dysfunction at HSCT. EFS at both 1 and 5 years post-HSCT were not influenced by (F) number of IEI-associated risk factors (bronchiectasis, colitis, malignancy, GLILD, hepatic comorbidity, and splenectomy), (G) HCT-CI score, (H) year of transplant, or (I) age at HSCT. Estimated EFS at 5 years (95% CI) are shown.
Neither the pretransplant HCT-CI score, P = .168 (Figure 2G); the era of transplant, P = .108 (Figure 2H); age at HSCT, P = .4 (Figure 2I); donor type, P = .9 (data not shown); prior immunosuppressive therapy (data not shown); or remission status of malignancy, where present, had a significant influence on EFS (data not shown).
Subsequent multivariable analysis confirmed only the number of IEI-associated complications at HSCT as a significant risk factor for EFS (P = .05), as shown in Table 5.
OS and EFS analysis within IEI subgroups
In patients with CID (n = 162), the presence of bronchiectasis and the number of IEI-associated complications at HSCT adversely affected OS (P = .033 and P = .009, respectively), whereas GLILD and the number of IEI-associated complications adversely affected EFS (P = .018 and P = .038, respectively). For patients with PD (n = 125), only bronchiectasis adversely affected OS and EFS, P = .008. Despite small numbers in PAD patients (n = 36), both OS and EFS were significantly influenced by hepatic comorbidity and age at HSCT, P = .003 and P = .005, respectively (supplemental Figure 2).
GVHD
In keeping with the frequent use of serotherapy, the cumulative incidence of grades 2 to 4 acute (aGVHD) was low at 22%, with only 8% of patients developing grade 3 to 4 aGVHD (Figure 3A). The cumulative incidence of extensive cGVHD was 7%. The incidence of combined limited and extensive GVHD was 16% at 1 year and 19% at 5 years post-HSCT (Figure 3B). Notably, IEI subgroup had no impact on rates of acute or chronic GVHD (supplemental Table 5). As expected, the absence of serotherapy increased the risk of grades 2 to 4 aGVHD (P = .039).
Cumulative incidence of aGVHD and cGVHD. The cumulative incidence of (A) grades 2 to 4 aGVHD was 22% (17% to 26%) at both 6 and 12 months post-HSCT (blue line), with 8% (5% to 11%) developing grades 3 to 4 aGVHD at both 6 and 12 months post-HSCT (red line). The cumulative incidence of (B) all cGVHD (limited and extensive) was 16% (12% to 20%) at 1 year and 19% (15% to 24%) at 5 years (blue line), whereas extensive chronic GVHD was seen in 7% (4% to 9%) at 1 year and 7% (4% to 10%) at 5 years (red line).
Cumulative incidence of aGVHD and cGVHD. The cumulative incidence of (A) grades 2 to 4 aGVHD was 22% (17% to 26%) at both 6 and 12 months post-HSCT (blue line), with 8% (5% to 11%) developing grades 3 to 4 aGVHD at both 6 and 12 months post-HSCT (red line). The cumulative incidence of (B) all cGVHD (limited and extensive) was 16% (12% to 20%) at 1 year and 19% (15% to 24%) at 5 years (blue line), whereas extensive chronic GVHD was seen in 7% (4% to 9%) at 1 year and 7% (4% to 10%) at 5 years (red line).
Chimerism
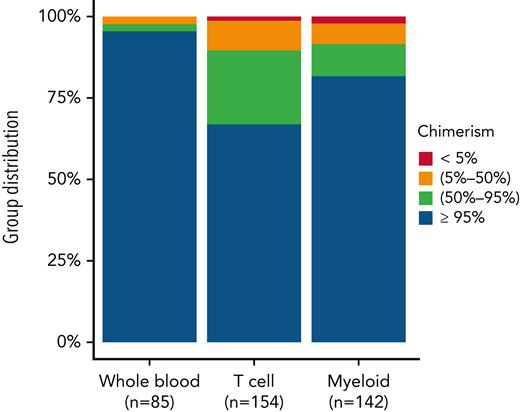
Either WB or lineage-specific peripheral blood chimerism was available at last follow-up for a total of 154 patients.
For those with only WB chimerism results (n = 85), rates of >95% donor, 50% to 95% donor, 5% to 50% donor, and <5% donor chimerism were 95.3%, 2.4%, 2.4%, and 0%, respectively. Lineage-specific T-cell chimerism (n = 154) and myeloid chimerism (n = 142) results at last follow-up were also available, with rates of >95% donor, 50% to 95% donor, 2% to 50% donor, and <5% donor T-cell chimerism of 66.9%, 22.7%, 9.1%, and 1.3%, respectively, and myeloid chimerism of 81.7%, 9.9%, 6.3%, and 2.1%, respectively (Figure 4).
WB, T-cell, and myeloid chimerism at last follow-up. Degree of donor chimerism at last follow-up in patients with available data (numbers indicated for each category). Left column, WB; middle column, T-cell; right column, myeloid chimerism.
WB, T-cell, and myeloid chimerism at last follow-up. Degree of donor chimerism at last follow-up in patients with available data (numbers indicated for each category). Left column, WB; middle column, T-cell; right column, myeloid chimerism.
When stratified by IEI subgroup, no differences were observed between rates of full donor chimerism at 1 year post-HSCT and at last follow-up (data not shown).
Immune reconstitution
Total lymphocytes and lymphocyte subsets and the requirement for IGRT at 2 years post-HSCT (n = 225) or at last assessment (n = 329) are shown in Table 6. Of note, at 2 years post-HSCT, 24.2% of patients were receiving IgRT and median lymphocyte subset counts were within the normal adult range.
Immune reconstitution
| . | . | At last assessment . | . | |||
|---|---|---|---|---|---|---|
| <2 y post-HSCT∗ . | ≥2 y post-HSCT† . | |||||
| Time to last assessment | Median (IQR) | 4.8 mo (2.4-12.3) | 60.9 mo (42-91.5) | |||
| Missing (%) | ‡N (%)∗ | Missing (%) | ‡N (%)∗ | |||
| Total | 104 (100) | 225 (100) | ||||
| IgRT | No | 44 (42.3) | 23 (38.3) | 9 (4) | 185 (85.6) | |
| Yes | 22 (36.7) | 25 (11.6) | ||||
| Not evaluated | 8 (13.3) | 2 (0.9) | ||||
| Unknown | 7 (11.7) | 4 (1.9) | ||||
| Missing (%) | Median (IQR) | Missing (%) | Median (IQR) | Normal range† | ||
| Lymphocytes (109/L) | 66 (63.5) | 1.1 (0.6-2.2) | 50 (22.2) | 2.0 (1.3-3.2) | 1.00-4.80 | |
| CD3+ T-cells (109/L) | 79 (76) | 0.8 (0.3-1.6) | 107 (47.6) | 1.2 (0.7-2.0) | 0.79-2.01 | |
| CD4+ T-cells (109/L) | 81 (77.9) | 0.2 (0.1-0.5) | 104 (46.2) | 0.6 (0.4-1.2) | 0.45-1.25 | |
| CD8+ T-cells (109/L) | 81 (77.9) | 0.5 (0.2-0.6) | 104 (46.2) | 0.6 (0.3-1.5) | 0.22-0.69 | |
| CD56+ NK-cells (109/L) | 81 (77.9) | 0.2 (0.1-0.3) | 113 (50.2) | 0.2 (0.1-3.1) | 0.10-0.44 | |
| CD19+ B-cells (109/L) | 81 (77.9) | 0 (0-0.3) | 118 (52.4) | 0.4 (0.2-1.1) | 0.11-0.36 | |
| Naïve T cells (109/L) | 96 (92.3) | 0 (0-0.1) | 177 (78.7) | 0.5 (0.2-1.5) | 0.19-0.98 | |
| . | . | At last assessment . | . | |||
|---|---|---|---|---|---|---|
| <2 y post-HSCT∗ . | ≥2 y post-HSCT† . | |||||
| Time to last assessment | Median (IQR) | 4.8 mo (2.4-12.3) | 60.9 mo (42-91.5) | |||
| Missing (%) | ‡N (%)∗ | Missing (%) | ‡N (%)∗ | |||
| Total | 104 (100) | 225 (100) | ||||
| IgRT | No | 44 (42.3) | 23 (38.3) | 9 (4) | 185 (85.6) | |
| Yes | 22 (36.7) | 25 (11.6) | ||||
| Not evaluated | 8 (13.3) | 2 (0.9) | ||||
| Unknown | 7 (11.7) | 4 (1.9) | ||||
| Missing (%) | Median (IQR) | Missing (%) | Median (IQR) | Normal range† | ||
| Lymphocytes (109/L) | 66 (63.5) | 1.1 (0.6-2.2) | 50 (22.2) | 2.0 (1.3-3.2) | 1.00-4.80 | |
| CD3+ T-cells (109/L) | 79 (76) | 0.8 (0.3-1.6) | 107 (47.6) | 1.2 (0.7-2.0) | 0.79-2.01 | |
| CD4+ T-cells (109/L) | 81 (77.9) | 0.2 (0.1-0.5) | 104 (46.2) | 0.6 (0.4-1.2) | 0.45-1.25 | |
| CD8+ T-cells (109/L) | 81 (77.9) | 0.5 (0.2-0.6) | 104 (46.2) | 0.6 (0.3-1.5) | 0.22-0.69 | |
| CD56+ NK-cells (109/L) | 81 (77.9) | 0.2 (0.1-0.3) | 113 (50.2) | 0.2 (0.1-3.1) | 0.10-0.44 | |
| CD19+ B-cells (109/L) | 81 (77.9) | 0 (0-0.3) | 118 (52.4) | 0.4 (0.2-1.1) | 0.11-0.36 | |
| Naïve T cells (109/L) | 96 (92.3) | 0 (0-0.1) | 177 (78.7) | 0.5 (0.2-1.5) | 0.19-0.98 | |
Last assessment is before 2 years post-HSCT for 104 patients who were lost to follow-up or died before 2 years.
Only patients alive and in follow-up by 2 years post-HSCT.
Percentages in this column are calculated from the nonmissing.
Status at last follow-up and causes of death
Two hundred thirty-seven patients (72% of total cohort) were alive at last follow-up at a median of 44.3 months post-HSCT. Performance status was available for 186 patients (78.5% of surviving patients), with 91.9% of patients reporting performance status (Lansky or Karnofsky) of 90% or greater. Accordingly, 87.3% of patients were attending school, higher education, or were employed (data not shown).
Limited data on post-HSCT fertility was available. A total of 16 patients reported conception (for themselves or their partner) following HSCT. Of those, 12 conceptions resulted in a live birth; 1 unwanted pregnancy was interrupted; for 3, no data were available; and there was 1 stillbirth/miscarriage.
For those 91 patients who died within the 5-year follow-up period, causes of death was transplant-related mortality in 73 (81.1%), recurrence of IEI (or IEI-related complication) in 11 (12.2%), other in 6 (6.7%), and not available in 1 (1.1%).
Discussion
This important, timely study reports detailed outcomes following HSCT for the largest cohort to date of IEI patients aged 15 years or more at transplant. As 75% of patients in this cohort were transplanted after 2011, it reflects contemporary transplant practice and supportive care. Until now, published HSCT outcome data in this rare patient group have been limited to single-center adult studies,11,12 or larger, predominantly pediatric studies including small numbers of older patients.3-5,23
Older IEI patients referred for HSCT have usually developed disease-associated complications such as opportunistic infections, autoimmunity, autoinflammation, organ damage, and/or malignancies. Reflecting this, 72.4% of our cohort had HCT-CI scores of 1 or greater at HSCT, including 32.6% with an HCT-CI ≥ 3. A previous study validating the HCT-CI score in 796 immunodeficiency patients was a much lower-risk cohort, with a median age at HSCT of 9 years, and only 37.5% had an HCT-CI score of 1 of greater, including 15.7% with a score ≥3. Over 50% of our cohort had active IEI-related complications at HSCT as currently it is much less common for asymptomatic adults with a genetic diagnosis of IEI (with or without family history) to be referred for consideration of HSCT, unless there is clear evidence of poor clinical outcomes in adulthood.18,24
IEI-related factors, which adversely affected outcomes, were bronchiectasis, prior splenectomy, hepatic comorbidity, and cumulative number of IEI-related complications at HSCT. These were all more important than patient age, donor, or conditioning intensity, suggesting that older age per se does not adversely affect HSCT outcome in carefully selected patients. Notably, hepatic comorbidity and bronchiectasis were more frequently seen in patients with PAD, particularly those with complex CVID, which may contribute to poorer outcomes observed in these patients. Our data strongly support the notion that HSCT should be considered for adult patients prior to development of significant end organ complications, specifically impairment of hepatic and pulmonary function, and that availability of an HLA-matched donor matters less, given the equally good outcomes achieved with 9/10 MUD and MMRD in this cohort. This is relevant for IEI patients, where chances of identifying suitable MRDs are reduced by the genetic origin of the disease.25 Active colitis, autoimmunity or treated malignancy prior to HSCT did not confer inferior outcomes and should not prevent transplant referral. Patients who had undergone prior splenectomy had worse outcomes (both OS and EFS), which is likely multifactorial. In this patient group, splenectomy is used for severe, refractory autoimmunity or thrombocytopenia, or advanced liver disease, and thus may indicate advanced disease and may increase risks of post-HSCT overwhelming sepsis.24,26 As indications for splenectomy were not available, further interpretation is difficult.
For the entire cohort, OS and EFS are remarkable considering the degree of pre-HSCT morbidity. Survival was influenced by underlying IEI being significantly better for patients with PD and CID compared with those with PAD. The OS observed for patients with PD and CID is excellent, only exceeded by a few single-center studies with established experience in transplanting older IEI patients.11-13 Our findings in the PAD group are consistent with previously reported inferior outcomes post-HSCT for CVID patients, although only 27 of the 39 patients in this group were characterized as classic CVID.10 Notably, we did not observe the same high rates of GVHD in CVID patients described by Wehr et al. CVID patients referred for HSCT typically have complex CVID and associated inflammatory or autoimmune complications such as nodular regenerative hyperplasia with hepatic impairment and/or GLILD.10,27,28 Although our PAD subgroup was small, it is striking that younger patients with less pre-HSCT morbidity, particularly hepatic comorbidity, had a much better outcome, strongly supporting early consideration of HSCT, prior to the development of significant noninfectious complications. There remains an urgent need for additional validated biomarkers to predict disease progression in CVID.29 The overall very low rates of severe acute or chronic GVHD in this cohort are encouraging, especially for patients with nonmalignant diseases. These may be explained by the high percentages (∼75% each) of transplants incorporating serotherapy, an HLA-matched donor, and reduced-toxicity conditioning regimens.
The few available data in adults with IEI indicate worsening quality of life (QOL) with age, but comparable data on transplanted patients are absent.30,31 QOL analysis pre- and post-HSCT should be addressed in prospective trials for older IEI patients. Nevertheless, >90% of survivors in our study had excellent performance scores and were attending school, university, or work, suggesting an acceptable QOL after successful HSCT, consistent with the limited published data.4,30,31
Assessing the influence of conditioning regimens on outcome is difficult in a retrospective study as disease-specific considerations, patient age, and comorbidity likely influence regimen selection. Accordingly, no impact of conditioning on outcome was seen in our study. Nevertheless, for patients with significant organ damage, regimens with reduced intensity and toxicity are likely to be preferred as supported by a recent prospective trial in a cohort of relatively old (50% >18 years) IEI patients with a median HCT-CI score of 3.13 The current Inborn Errors Working Party guidelines for HSCT for IEI recommend 3 different reduced-intensity regimens for adult patients.16 Assessing longer term outcomes will be important to confirm the positive impact of HSCT on reducing recurrent events associated with underlying IEIs observed in untransplanted adults.
In conclusion, we report the largest cohort of older adolescents and adults with IEI having undergone HSCT and have identified IEI subgroup, HCT-CI score, and the cumulative number of IEI-related comorbidities as risk factors for adverse outcome. These data provide much-needed evidence for physicians counseling IEI patients and their families, regarding the role of HSCT as a therapeutic intervention, and they should aid in designing appropriate prospective studies to define the preferred transplant approach for these patients. This data supports early referral of IEI patients, regardless of their age, prior to the development of significant organ damage.
Acknowledgements
The authors thank Alessandro Aiuti, Alexei Maschan, Mahmoud Aljurf, Tobias Gedde-Dahl, Gunhan Gurman, Victoria Bordon, Gergely Kriván, Franco Locatelli, Fulvio Porta, David Valcárcel, Yves Beguin, Maura Faraci, Nicolaus Kröger, Aleksandr Kulagin, Peter J. Shaw, Joan Hendrik Veelken, Cristina Diaz de Heredia, Franca Fagioli, Matthias Felber, Bernd Gruhn, Wolfgang Holter, Claudia Rössig, Petr Sedlacek, Jane Apperley, Mouhab Ayas, Ivana Bodova, Goda Choi, J.J. Cornelissen, Anne Sirvent, Anjum Khan, Alphan Kupesiz, Stig Lenhoff, Hakan Ozdogu, Nicolas von der Weid, Montserrat Rovira, Rik Schots, and Donald C. Vinh for providing patient data.
Authorship
Contribution: E.C.M., M.H.A., A.C.L., N.M., F.S., M.L.F., and A.R.G. conceived and designed the study; T.S. managed the data; D.-J.E. and K.B. performed the statistical analysis; M.H.A. and E.C.M. analyzed the data and wrote the manuscript; all authors except D.-J.E., T.S., and K.B. contributed clinical data; all authors reviewed and edited the manuscript; and T.S. and D.-J.E. directly accessed and verified the underlying data.
Conflict-of-interest disclosure: M.H.A. reports honoraria for educational events from Grifols and Octapharma, support for attending meetings from Octapharma, and stock ownership in and consulting fees from CSL Behring. C.W. reports participation on a data safety monitoring board or advisory board for Takeda. The remaining authors declare no competing financial interests.
Complete lists of the members of the Inborn Errors Working Party of the European Society for Blood and Marrow Transplantation and the European Society for Immunodeficiencies appear in “Appendix 1” and “Appendix 2,” respectively.
Correspondence: Emma C. Morris, UCL Institute of Immunity & Transplantation, The Pears Building, Rosslyn Hill, London NW3 2PP, United Kingdom; e-mail: e.morris@ucl.ac.uk.
Inborn Errors Working Party of the European Society for Blood and Marrow Transplantation members involved in this study
Alessandro Aiuti, Alexei Maschan, Mahmoud Aljurf, Tobias Gedde-Dahl, Gunhan Gurman, Victoria Bordon, Gergely Kriván, Franco Locatelli, Fulvio Porta, David Valcárcel, Yves Beguin, Maura Faraci, Nicolaus Kröger, Aleksandr Kulagin, Peter J. Shaw, Joan Hendrik Veelken, Cristina Diaz de Heredia, Franca Fagioli, Matthias Felber, Bernd Gruhn, Wolfgang Holter, Claudia Rössig, Petr Sedlacek, Jane Apperley, Mouhab Ayas, Ivana Bodova, Goda Choi, J. J. Cornelissen, Anne Sirvent, Anjum Khan, Alphan Kupesiz, Stig Lenhoff, Hakan Ozdogu, Nicolas von der Weid, Montserrat Rovira, Rik Schots, and Donald C. Vinh.
European Society for Immunodeficiencies members involved in this study
Alessandro Aiuti, Alexei Maschan, Mahmoud Aljurf, Tobias Gedde-Dahl, Gunhan Gurman, Victoria Bordon, Gergely Kriván, Franco Locatelli, Fulvio Porta, David Valcárcel, Yves Beguin, Maura Faraci, Nicolaus Kröger, Aleksandr Kulagin, Peter J. Shaw, Joan Hendrik Veelken, Cristina Diaz de Heredia, Franca Fagioli, Matthias Felber, Bernd Gruhn, Wolfgang Holter, Claudia Rössig, Petr Sedlacek, Jane Apperley, Mouhab Ayas, Ivana Bodova, Goda Choi, J. J. Cornelissen, Anne Sirvent, Anjum Khan, Alphan Kupesiz, Stig Lenhoff, Hakan Ozdogu, Nicolas von der Weid, Montserrat Rovira, Rik Schots, and Donald C. Vinh.
References
Author notes
Summarized, deidentified data are available from the corresponding author upon written request.
The online version of this article contains a data supplement.
There is a Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.





This feature is available to Subscribers Only
Sign In or Create an Account Close Modal