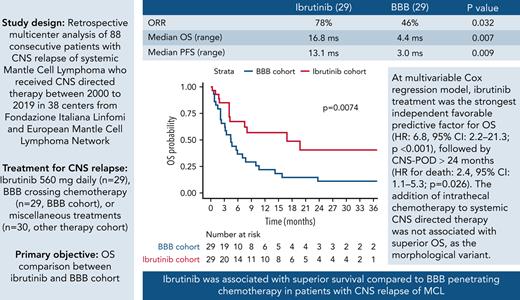
Key Points
Ibrutinib provided superior survival compared with BBB crossing chemotherapy in patients with CNS relapse of mantle cell lymphoma.
Addition of intrathecal therapy was not associated with improved OS, and CNS POD ≤24 months was an independent adverse prognostic factor.
Abstract
Central nervous system (CNS) relapse of mantle cell lymphoma (MCL) is a rare phenomenon with dismal prognosis, where no standard therapy exists. Since the covalent Bruton tyrosine kinase (BTK) inhibitor ibrutinib is effective in relapsed/refractory MCL and penetrates the blood–brain barrier (BBB), on behalf of Fondazione Italiana Linfomi and European Mantle Cell Lymphoma Network we performed a multicenter retrospective international study to investigate the outcomes of patients treated with ibrutinib or chemoimmunotherapy. In this observational study, we recruited patients with MCL with CNS involvement at relapse who received CNS-directed therapy between 2000 and 2019. The primary objective was to compare the overall survival (OS) of patients treated with ibrutinib or BBB crossing chemotherapy. A propensity score based on a multivariable binary regression model was applied to balance treatment cohorts. Eighty-eight patients were included. The median age at study entry was 65 years (range, 39-87), 76% were males, and the median time from lymphoma diagnosis to CNS relapse was 16 months (range, 1-122). Patients were treated with ibrutinib (n = 29, ibrutinib cohort), BBB crossing chemotherapy (ie, high-dose methotrexate ± cytarabine; n = 29, BBB cohort), or miscellaneous treatments (n = 30, other therapy cohort). Both median OS (16.8 vs 4.4 months; P = .007) and median progression-free survival (PFS) (13.1 vs 3.0 months; P = .009) were superior in the ibrutinib cohort compared with the BBB cohort. Multivariable Cox regression model revealed that ibrutinib therapeutic choice was the strongest independent favorable predictive factor for both OS (hazard ratio [HR], 6.8; 95% confidence interval [CI], 2.2-21.3; P < .001) and PFS (HR, 4.6; 95% CI, 1.7-12.5; P = .002), followed by CNS progression of disease (POD) >24 months from first MCL diagnosis (HR for death, 2.4; 95% CI, 1.1-5.3; P = .026; HR for death or progression, 2.3; 95% CI, 1.1-4.6; P = .023). The addition of intrathecal (IT) chemotherapy to systemic CNS-directed therapy was not associated with superior OS (P = .502) as the morphological variant (classical vs others, P = .118). Ibrutinib was associated with superior survival compared with BBB-penetrating chemotherapy in patients with CNS relapse of MCL and should be considered as a therapeutic option.
Introduction
Mantle cell lymphoma (MCL) represents a relatively uncommon form of non-Hodgkin lymphoma, with a relapsing and remitting course.1 The disease is characterized by genomic instability and worsening clinical and histopathological features at each subsequent relapse.2,3 Immunochemotherapy induction,4-8 often followed by rituximab maintenance,5,9 remains a standard of care in the frontline setting.
Targeting Bruton tyrosine kinase (BTK) with oral inhibitors including ibrutinib,10,11 acalabrutinib, or zanubrutinib monotherapy12,13 represents a well-tolerated and established treatment approach in patients with relapsed or refractory MCL, with overall response rate (ORR) and a complete response rate (CRR) in the range of 70% and 20%, respectively.
Central nervous system (CNS) relapse of MCL is a rare phenomenon with a poor outcome, for which a therapeutic standard of care has not been identified. Approximately 4% of patients with MCL develop CNS relapse14,15; baseline poor risk clinical, biological and histopathological features correlate with a higher risk of CNS involvement at recurrence.14-16 Traditional management strategies include chemotherapy, IV or intrathecal (IT), combined chemoradiotherapy, or radiotherapy alone. The use of high-dose antimetabolites such as high-dose methotrexate (HD-MTX) and high-dose arabinoside-cytarabine (HD-Ara-C) is widespread, but evidence of utility is mostly extrapolated from diffuse large B-cell lymphoma.17-20 Patients with CNS MCL have a median survival of 3 to 6 months, and the majority of patients die of progressive CNS disease.14
Ibrutinib is known to penetrate the blood–brain barrier (BBB) in preclinical models,21,22 and is active when administered as monotherapy in patients with a range of B-cell malignancies presenting with CNS involvement.23,24 Some groups have described case reports25-27 and small series suggesting efficacy of ibrutinib in CNS MCL. A series of 5 patients from the United Kingdom demonstrated a response in all patients receiving ibrutinib, combined with high-dose antimetabolites in 2 patients and steroids in 3 patients.28 A separate case series of 3 MCL patients with CNS relapse documented responses to ibrutinib monotherapy in all patients with minimal toxicity.29
However, no large series have investigated the efficacy and toxicity of ibrutinib in CNS relapsed MCL, and no comparisons are available with high-dose antimetabolite-based chemoimmunotherapy.
To address these key unanswered questions, we conducted a multicenter, international cohort study to compare the clinical outcomes of patients with CNS relapsed MCL treated with ibrutinib or chemoimmunotherapy.
Methods
Study design
We conducted a multicenter observational retrospective cohort study of consecutive BTK inhibitor-naïve MCL patients with documented CNS involvement at relapse who received CNS-directed therapy between January 2000 and December 2019. Thirty-eight centers participated, including 18 from the FIL (Fondazione Italiana Linfomi) and 20 international sites from the EMCLN (European MCL Network). Overall, 39 patients who received standard therapy14,30 and 10 patients treated with ibrutinib that were previously described were included.25,26,28,29
CNS involvement with MCL at relapse was defined by ≥1 of the following criteria: histologically confirmed CNS involvement, neuroradiological findings compatible with CNS involvement, and/or positive cerebrospinal fluid (CSF) (cytology and flow cytometry). CNS involvement pattern was defined according to neuroradiological and CSF findings as parenchymal and/or leptomeningeal.
According to the treatment received, patients were divided into 3 cohorts: (1) the ibrutinib cohort, comprising patients treated with ibrutinib at a standard dose of 560 mg by mouth once daily; (2) the BBB cohort, comprising patients treated with chemotherapeutic agents known to cross the BBB at defined dosage (MTX ≥2 g/m2, Ara-C ≥2 g/m2, and ifosfamide ≥3 g/m2), hereafter referred to as high dose (HD); and (3) the other therapy cohort, comprising patients treated with chemotherapy not able to cross the BBB, IT chemotherapy alone, or palliative therapies. Miscellaneous treatments included in cohort 3 were considered suboptimal, and therefore the outcomes of these patients were analyzed separately and not included in the survival comparison and propensity score.
The study was approved by the institutional review board of all participating centers.
Endpoints and assessments
The primary objective of the study was to compare OS in the ibrutinib and BBB cohorts. Secondary endpoints were to compare progression-free survival (PFS) in the ibrutinib and BBB cohort; to describe OS and PFS in the entire study population and in the other therapy cohort; to describe the ORR and safety in the entire study population; to explore prognostic factors such as time from the initial diagnosis to CNS relapse and pattern of CNS involvement (parenchymal vs leptomeningeal vs both); and impact of additional IT chemotherapy in the ibrutinib and BBB cohort. A cutoff point of 24 months for time to CNS progression of disease (POD) from initial MCL diagnosis was used, as previously described.31 Patients were assessed for response according to International Primary Central Nervous System Lymphoma Collaborative Group Criteria32 for the MCL CNS component and according to response criteria published in 199933 for the systemic MCL component. Toxicity assessment was defined according to the CTCAE (Common Terminology Criteria for Adverse Events) Version 4.0; data collection was intentionally limited to grade ≥3 nonhematological events.
Statistical analysis
OS time was calculated from the date of initiation of CNS-directed therapy until the date of death from any cause or censored at the last follow-up. PFS time was calculated from the date of initiation of CNS-directed therapy until the date of progression or relapse (at CNS or systemic) or death from any cause or censored at the last follow-up. OS and PFS curves were estimated using the Kaplan-Meier method and compared using the log-rank test. Uni- and multivariable analyses of OS and PFS were carried out using Cox regression models. Overfitting because of the high number of predictors in the model in relation to the low number of deaths was controlled by the penalized maximum likelihood estimation method.34 To detect a different distribution of characteristics between treatment assignment cohorts (ibrutinib cohort vs BBB cohort), the Wilcoxon-Mann-Whitney rank test was applied to continuous variables, while the Fisher-Freeman-Halton test35 to factors.
To account for the selection bias resulting from nonrandom treatment assignment, the statistical analyses involving a comparison between treatment groups were performed by adjusting for the propensity score (PS) in multivariable models.36 PS by 1-to-1 pair matching or matching weights using PS37 were infeasible because of significant sample size shrinkage. PS was estimated using a multivariable binary logistic regression model where treatment was the response variable, and the covariates were the possible confounding factors that could influence ibrutinib choice. These confounding factors included sex, age at CNS relapse, morphological variant, ECOG (Eastern Cooperative Oncology Group) performance status, MIPI (MCL International Prognostic Index) score at diagnosis, B symptoms at diagnosis, serum lactate dehydrogenase (LDH) concentration over upper limits of normal,38 CNS POD within 24 months, previous HD-Ara-C, and autologous stem cell transplantation (SCT) for systemic MCL. The year of treatment for CNS relapse was not included in the list of confounding factors because of the known differences in treatment eras following the later approval of ibrutinib for routine use in clinical practice between 2013 and 2016 across participating international centers, because including variables that are related to the exposure but not to the outcome will decrease the precision of the estimated treatment effect.39
Statistical analyses were performed with SAS (SAS Institute; Cary, NC) and R-software (R-Foundation for Statistical Computing; Vienna, Austria). The database was locked on 30 June 2020 for analysis.
Results
Patients and treatments
Eighty-eight patients with CNS relapse of MCL were included in the analysis: 29 patients (33%) in the ibrutinib cohort, 29 patients (33%) in the BBB cohort, and 30 patients (34%) in the other therapy cohort. Baseline characteristics and frontline therapy for systemic MCL for the 3 cohorts are displayed in Table 1. The median age of the entire study population at initial MCL diagnosis was 63 years (range, 39-84); 76% were male; MIPI score was high in 65% of 68 evaluable patients, and blastoid variant was diagnosed in 26% of 81 evaluable cases. Patient characteristics at CNS relapse are summarized in Table 2. The median age at CNS relapse was 65 years (range, 39-87); the median number of treatment lines preceding CNS relapse was 1 (range, 1-5). Concurrent systemic relapse was detected in 28 (43%) of 65 evaluable patients and was more frequent in the BBB cohort (53%). The median time to CNS relapse from initial diagnosis in the entire study population was 16 months (range, 1-122). Considering 2013, the year of ibrutinib availability in routine practice, as a cutoff, 18 patients (62%) in the BBB cohort and 13 patients (43%) in the other therapies cohort were treated in the pre-2013 era. Radiology was consistent with CNS involvement in all patients; CNS involvement was confirmed by parenchymal biopsy in 2 patients and by cytology on CSF in 54 patients (61%). Isolated leptomeningeal infiltration was the most common pattern of CNS involvement (51%).
Systemic MCL diagnosis characteristics
| . | Ibrutinib Cohort (n = 29), n (%) . | BBB Cohort (n = 29), n (%) . | Other Therapy Cohort (n = 30), n (%) . | Total (n = 88), n (%) . |
|---|---|---|---|---|
| Patient characteristics | ||||
| Age (yr), median (range) | 66 (48-80) | 56 (39-84) | 65 (39-78) | 63 (39-84) |
| Gender (male) | 24 (83) | 20 (69) | 23 (77) | 67 (76) |
| Disease characteristics | ||||
| B symptoms (Yes) | 10/26 (38) | 18/28 (64) | 12/29 (41) | 40/83 (48) |
| Clinical stage (IV) | 25/28 (89) | 28/29 (97) | 29/29 (100) | 82/86 (95) |
| ECOG | ||||
| 0-1 | 23/28 (82) | 13/19 (68) | 14/20 (70) | 50/67 (75) |
| 2-4 | 5/28 (18) | 6/19 (32) | 6/20 (30) | 17/67 (25) |
| LDH (>ULN) | 14/21 (67) | 15/19 (79) | 14/21 (67) | 43/61 (70) |
| MIPI (high) | 18/26 (69) | 17/27 (63) | 16/25 (64) | 51/68 (65) |
| Morphology variant | ||||
| Classic | 21/28 (75) | 17/28 (67) | 15/25 (60) | 53/81 (65) |
| Pleomorphic | 1/28 (4) | 5/28 (18) | 1/21 (4) | 7/81 (9) |
| Blastoid | 6/28 (21) | 6/28 (21) | 9/25 (36) | 21/81 (26) |
| Ki67 (>30%) | 11/17 (65) | 20/21 (95) | 16/18 (89) | 47/56 (84) |
| Extranodal site involvement | 22/28 (79) | 27/28 (96) | 21/23 (91) | 70/79 (89) |
| First-line therapy for systemic MCL | ||||
| Rituximab-containing regimen | 29/29 (100) | 25/28 (89) | 28/29 (97) | 82/86 (95) |
| Anthracycline-containing regimen | 14/21 (67) | 26/26 (100) | 29/29 (100) | 69/76 (91) |
| HD-ARA-C–containing regimen | 15/29 (52) | 23/29 (79) | 17/29 (59) | 55/87 (63) |
| ASCT | 9/29 (31) | 15/28 (54) | 7/29 (24) | 31/86 (36) |
| . | Ibrutinib Cohort (n = 29), n (%) . | BBB Cohort (n = 29), n (%) . | Other Therapy Cohort (n = 30), n (%) . | Total (n = 88), n (%) . |
|---|---|---|---|---|
| Patient characteristics | ||||
| Age (yr), median (range) | 66 (48-80) | 56 (39-84) | 65 (39-78) | 63 (39-84) |
| Gender (male) | 24 (83) | 20 (69) | 23 (77) | 67 (76) |
| Disease characteristics | ||||
| B symptoms (Yes) | 10/26 (38) | 18/28 (64) | 12/29 (41) | 40/83 (48) |
| Clinical stage (IV) | 25/28 (89) | 28/29 (97) | 29/29 (100) | 82/86 (95) |
| ECOG | ||||
| 0-1 | 23/28 (82) | 13/19 (68) | 14/20 (70) | 50/67 (75) |
| 2-4 | 5/28 (18) | 6/19 (32) | 6/20 (30) | 17/67 (25) |
| LDH (>ULN) | 14/21 (67) | 15/19 (79) | 14/21 (67) | 43/61 (70) |
| MIPI (high) | 18/26 (69) | 17/27 (63) | 16/25 (64) | 51/68 (65) |
| Morphology variant | ||||
| Classic | 21/28 (75) | 17/28 (67) | 15/25 (60) | 53/81 (65) |
| Pleomorphic | 1/28 (4) | 5/28 (18) | 1/21 (4) | 7/81 (9) |
| Blastoid | 6/28 (21) | 6/28 (21) | 9/25 (36) | 21/81 (26) |
| Ki67 (>30%) | 11/17 (65) | 20/21 (95) | 16/18 (89) | 47/56 (84) |
| Extranodal site involvement | 22/28 (79) | 27/28 (96) | 21/23 (91) | 70/79 (89) |
| First-line therapy for systemic MCL | ||||
| Rituximab-containing regimen | 29/29 (100) | 25/28 (89) | 28/29 (97) | 82/86 (95) |
| Anthracycline-containing regimen | 14/21 (67) | 26/26 (100) | 29/29 (100) | 69/76 (91) |
| HD-ARA-C–containing regimen | 15/29 (52) | 23/29 (79) | 17/29 (59) | 55/87 (63) |
| ASCT | 9/29 (31) | 15/28 (54) | 7/29 (24) | 31/86 (36) |
ULN, upper limits of normal.
CNS MCL relapse characteristics
| . | Ibrutinib Cohort (n = 29), n (%) . | BBB Cohort (n = 29), n (%) . | Other Therapy Cohort (n = 30), n (%) . | Total (n = 88), n (%) . |
|---|---|---|---|---|
| Patient characteristics | ||||
| Age (yr), median (range) | 67 (48-82) | 60 (39-87) | 70 (43-80) | 65 (39-87) |
| Disease characteristics | ||||
| Time to CNS relapse, mo (range) | 24 (1-85) | 13 (3-99) | 18 (2-122) | 16 (1-122) |
| CNS POD ≤24 mo | 14/29 (48) | 20/29 (69) | 18/30 (60) | 52/88 (59) |
| Extra CNS involvement | 10/29 (35) | 9/17 (53) | 9/19 (47) | 28/65 (43) |
| CNS involvement pattern | ||||
| Leptomeningeal | 13/29 (45) | 14/24 (58) | 13/25 (52) | 40/78 (51) |
| Parenchymal | 10/29 (34) | 2/24 (9) | 6/25 (24) | 18/78 (23) |
| Both | 6/29 (21) | 8/24 (33) | 6/25 (24) | 20/78 (26) |
| CNS MCL therapy | ||||
| Ibrutinib | 29/29 (100) | — | — | 29/88 (33) |
| HD-MTX | — | 7/29 (24) | — | 7/88 (8) |
| HD-MTX-ARAC | — | 20/29 (69) | — | 20/88 (23) |
| HD-IFO | — | 2/29 (7) | — | 2/88 (2) |
| IT therapy | — | — | 17/30 (57) | 17/88 (19) |
| Bendamustine-based regimen | — | — | 6/30 (20) | 6/88 (7) |
| Palliation | — | — | 7/30 (23) | 7/88 (8) |
| Additional CNS MCL therapies | ||||
| IT therapy | 12/29 (41) | 23/29 (79) | 5/30 (17) | 40/88 (46) |
| Rituximab | 4/29 (14) | 10/28 (36) | 7/27 (26) | 21/84 (25) |
| Consolidative RT | 1/26 (4) | 2/17 (12) | 7/18 (39) | 10/61 (16) |
| Number of prior therapies before CNS relapse | 1 (1-2) | 1 (1-5) | 1 (1-3) | 1 (1-5) |
| . | Ibrutinib Cohort (n = 29), n (%) . | BBB Cohort (n = 29), n (%) . | Other Therapy Cohort (n = 30), n (%) . | Total (n = 88), n (%) . |
|---|---|---|---|---|
| Patient characteristics | ||||
| Age (yr), median (range) | 67 (48-82) | 60 (39-87) | 70 (43-80) | 65 (39-87) |
| Disease characteristics | ||||
| Time to CNS relapse, mo (range) | 24 (1-85) | 13 (3-99) | 18 (2-122) | 16 (1-122) |
| CNS POD ≤24 mo | 14/29 (48) | 20/29 (69) | 18/30 (60) | 52/88 (59) |
| Extra CNS involvement | 10/29 (35) | 9/17 (53) | 9/19 (47) | 28/65 (43) |
| CNS involvement pattern | ||||
| Leptomeningeal | 13/29 (45) | 14/24 (58) | 13/25 (52) | 40/78 (51) |
| Parenchymal | 10/29 (34) | 2/24 (9) | 6/25 (24) | 18/78 (23) |
| Both | 6/29 (21) | 8/24 (33) | 6/25 (24) | 20/78 (26) |
| CNS MCL therapy | ||||
| Ibrutinib | 29/29 (100) | — | — | 29/88 (33) |
| HD-MTX | — | 7/29 (24) | — | 7/88 (8) |
| HD-MTX-ARAC | — | 20/29 (69) | — | 20/88 (23) |
| HD-IFO | — | 2/29 (7) | — | 2/88 (2) |
| IT therapy | — | — | 17/30 (57) | 17/88 (19) |
| Bendamustine-based regimen | — | — | 6/30 (20) | 6/88 (7) |
| Palliation | — | — | 7/30 (23) | 7/88 (8) |
| Additional CNS MCL therapies | ||||
| IT therapy | 12/29 (41) | 23/29 (79) | 5/30 (17) | 40/88 (46) |
| Rituximab | 4/29 (14) | 10/28 (36) | 7/27 (26) | 21/84 (25) |
| Consolidative RT | 1/26 (4) | 2/17 (12) | 7/18 (39) | 10/61 (16) |
| Number of prior therapies before CNS relapse | 1 (1-2) | 1 (1-5) | 1 (1-3) | 1 (1-5) |
HD-ARA-C, high dose of cytosine arabinoside; HD-IFO, high dose ifosfamide; HD-MTX, high dose methotrexate; Other therapy cohort, nonpenetrating blood brain barrier therapy cohort; RT, radiotherapy.
As shown in supplemental Figure 1 in the data supplement, at CNS relapse, the ibrutinib and BBB cohorts displayed similar characteristics in terms of sex, morphological variant, presence of B symptoms, elevated LDH, previous exposure to HD-Ara-C–containing regimens, previous autologous SCT, and time to CNS relapse. Conversely, the median age at relapse was significantly higher in the ibrutinib cohort (67 vs 60 years; P = .005), whereas the ECOG score was 0 to 1 in 79.3% of patients in the ibrutinib cohort and 44.4% of patients in the BBB cohort (P = .006). The incidence of concomitant systemic and CNS disease in the ibrutinib and BBB cohorts is shown in supplemental Figure 2.
CNS-directed therapy in the ibrutinib cohort consisted of ibrutinib at a standard dose of 560 mg once daily; in the BBB cohort, 27 patients (93%) received HD-MTX, combined with HD-Ara-C in 69%; in the other therapy cohort treatment consisted of IT chemotherapy alone in 17 (57%), bendamustine-based regimen in 6 (20%), and palliation in 7 (23%) patients. IV rituximab was added in 25% of all active treatments delivered: 14% in the ibrutinib cohort, 36% in the BBB cohort, and 26% in the other therapy cohort, respectively. Cerebral radiotherapy was given as consolidation in 16% of all cases. Two patients in the BBB cohort and 1 in the ibrutinib cohort underwent subsequent allogeneic SCT consolidation. When excluding those receiving IT chemotherapy only, additional IT chemotherapy was delivered to 40 of 71 (56%) patients: 12 of 29 (41%) patients in the ibrutinib cohort, 23 of 29 (79%) in the BBB cohort, and 5 of 13 (38%) in the other therapy cohort.
Response and toxicity
The response was evaluable in 77 patients (87.5%); data for response assessment were missing in 7% of patients in the ibrutinib and BBB cohorts. The ORR was 78% (CRR, 46%) in the ibrutinib cohort, 46% (CRR, 21%) in the BBB cohort, and 33% (CRR, 13%) in the other therapy cohort, respectively. The ORR was higher in the ibrutinib cohort than in the BBB cohort (P = .031).
Grade ≥3 nonhematological toxicity observed in each cohort was infection (n = 2 grade 3), sepsis (n = 1 grade 4), and hepatic failure (n = 1 grade 4) in the BBB cohort, grade 3 herpes zoster infection (n = 1) and grade 3 diarrhea (n = 1) in the other therapy cohort, and grade 3 atrial fibrillation (n = 1) and grade 5 heart failure (n = 1) in the ibrutinib cohort. The only grade 5 adverse event because of heart failure occurred in a 75-year-old patient with a previous history of severe cardiomyopathy who was in complete remission on ibrutinib; the relationship with ibrutinib was deemed uncertain by the local investigator. In the ibrutinib cohort, no major bleeding or invasive pulmonary or CNS fungal infection was reported.
Survival
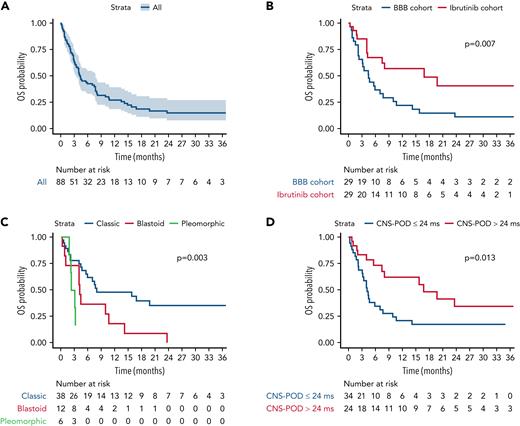
With a median follow-up of 29.5 (interquartile range [IQR], 14.0-35.0) months, the median OS for the whole study population was 4.3 (95% confidence interval [CI], 3.4-7.7] months (Figure 1A), and the median PFS was 3.0 (95% CI, 2.4-6.0) months (Figure 2A). In the other therapy cohort, the median OS and PFS were 2.8 (95% CI, 2.5-4.3) and 2.2 (95% CI, 1.4-3.7) months, respectively (supplemental Figures 3 and 4).
OS. (A) Entire study population; (B) ibrutinib cohort vs BBB cohort; (C) morphological variant: classic vs blastoid vs pleomorphic variant; and (D) CNS POD >24 months vs ≤24 months. P refers to P values from the log-rank test. Other therapy cohort was not included in (B-D).
OS. (A) Entire study population; (B) ibrutinib cohort vs BBB cohort; (C) morphological variant: classic vs blastoid vs pleomorphic variant; and (D) CNS POD >24 months vs ≤24 months. P refers to P values from the log-rank test. Other therapy cohort was not included in (B-D).
PFS. (A) Entire study population; (B) ibrutinib cohort vs BBB cohort; (C) morphological variant: classic vs blastoid vs pleomorphic variant; (D) CNS POD >24 months vs ≤24 months. P refers to P values from the log-rank test. Other therapy cohort was not included in (B-D).
PFS. (A) Entire study population; (B) ibrutinib cohort vs BBB cohort; (C) morphological variant: classic vs blastoid vs pleomorphic variant; (D) CNS POD >24 months vs ≤24 months. P refers to P values from the log-rank test. Other therapy cohort was not included in (B-D).
When focusing on the comparison between 58 patients in the ibrutinib cohort and BBB cohort, after a median follow-up of 29.5 months (IQR, 9.6-35), median OS and median PFS were 16.8 (95% CI, 7.5-not reached [NR]) and 13.1 (95% CI, 4.0-NR) months for the 29 patients receiving ibrutinib, and 4.4 (95% CI, 3.1-9.9) and 3.0 (95% CI, 1.6-6.6) months for the 29 patients receiving BBB crossing therapies, respectively. Survival differences between ibrutinib and BBB cohort were statistically significant for both OS (P = .007) (Figure 1B) and PFS (P = .009) (Figure 2B).
To address differences in the year of treatment as a potential confounding factor, we performed an analysis of outcomes according to treatment era, dichotomized at 2013. The treatment era did not impact survival among nonibrutinib-treated patients (BBB and other therapy cohorts) (supplemental Figures 5 and 6). In the BBB cohort only, the median OS for 18 patients treated before and for 11 patients treated after 2013 was 3.9 (95% CI, 2.3-14.2) and 4.7 (95% CI, 2.2-NR) months, respectively (P = .89); median PFS for patients treated before and after 2013 was 2.9 (95% CI, 1.6-8.8) and 3.0 (95% CI, 1.1-NA) months (P = .80) (supplemental Figures 7 and 8). When confining analysis to patients treated after 2013, ibrutinib remained associated with improved OS (P < .001) and PFS (P < .001) relative to the pooled nonibrutinib cohorts (supplemental Figures 9 and 10). A statistically significant difference in survival in favor of ibrutinib was also observed when considering patients treated after 2013 in the BBB cohort only. In particular, the median OS for patients in the ibrutinib cohort and the BBB cohort treated after 2013 was 16.8 (95% CI, 7.5-NA) and 4.7 (95% CI, 2.2-NA) months, respectively (P = .032), whereas median PFS was 13.1 (95% CI, 4.0-NA) months and 3.0 (95% CI, 1.1-6.0) months, respectively (P = .035) (supplemental Figures 11 and 12).
At the last follow-up, 17 patients on ibrutinib remain alive, with a median time on therapy of 11.8 months (range, 0.6-68.9). In the BBB cohort, 5 patients are alive, 2 of them after allogeneic SCT consolidation. All 3 patients receiving allogeneic SCT consolidation (n = 2 in the BBB cohort and n = 1 in the ibrutinib cohort) are alive and in CR at the last follow-up, after 1.2 (ibrutinib cohort) and 2.4 and 5.1 (BBB cohort) years, respectively.
The classical morphological variant and a late (>24 months) CNS POD were associated with superior OS and PFS, whereas no difference in survival was found according to additional IT therapy and CNS involvement pattern. Specifically, the classical morphological variant, compared with blastoid and pleomorphic, was associated with superior median OS (8.0 [95% CI, 5.9-NR] vs 4.2 [95% CI, 4.0-NR] vs 2.7 [95% CI, 2.2-NR] months; P = .003) (Figure 1C) and median PFS (7.5 [95% CI, 3.4-NR] vs 4.0 [95% CI, 2.4-NR] vs 1.7 [95% CI, 1.5-NR] months; P = .008) (Figure 2C). Similarly, CNS POD >24 months was associated with superior median OS (16.8 [95% CI, 7.5-NR] vs 4.2 [95% CI, 3.2-7.7] months; P = .013) (Figure 1D) and median PFS (13.1 [95% CI, 6.1-NA] vs 2.5 [95% CI, 1.6-6.6] months; P = .012) (Figure 2D). Survival curves stratified according to CNS involvement pattern and additional IT therapy are shown in supplemental Figures 13-16. In particular, the median OS was 14.2 (95% CI, 4.2-NR) months for patients not receiving additional IT chemotherapy and 5.6 (95% CI, 4.0-16.8) months for patients receiving additional IT chemotherapy (P = .652), whereas the median PFS was 7.5 (95% CI, 3.32-NA) months for patients not receiving IT and 4.0 (95% CI, 2.37-8.84) months for patients receiving IT (P = .838), respectively. Furthermore, no OS impact was observed in either the ibrutinib (P = .508) or the BBB cohort (P = .721), according to additional IT therapy.
The putative prognostic variables analyzed in the univariate Cox models for OS and PFS are listed in Table 3. Univariable Cox regression model demonstrated statistical significance in OS and PFS in favor of ibrutinib compared with BBB cohort: hazard ratio (HR) for death, 2.46 (95% CI, 1.24-4.90; P = .010) and HR for progression or death, 2.29 (95% CI, 1.21-4.36; P = .011), respectively. Variables other than ibrutinib that were significantly associated with superior survival outcomes were the classical morphological variant and a late (>24 months) CNS POD. Specifically, HR for death was 2.38 (95% CI, 1.14-4.98) for the comparison between classical vs blastoid morphological variant and 4.85 (95% CI, 1.61-14.57) for the comparison between classical vs pleomorphic morphological variant (P = .005); while HR for progression or death was 2.47 (95% CI, 1.18-5.20) classical vs blastoid morphological variant and 3.67 (95% CI, 1.32-10.79) for the comparison between classical and pleomorphic morphological variant (P = .010). Similarly, in univariate analysis, an early (≤24 months) CNS POD was associated with an inferior survival if compared with a late CNS POD: HR for death was 2.37 (95% CI, 1.18-4.77; P = .015), and HR for progression or death was 2.31 (95% CI, 1.20-4.44; P = .012).
Univariate Cox regression model estimates
| Variable . | Comparisons . | OS . | PFS . | ||
|---|---|---|---|---|---|
| HR (CI 95%) . | Wald P value . | HR (CI 95%) . | Wald P value . | ||
| Cohort | BBB vs ibrutinib | 2.46 (1.24-4.90) | .010 | 2.29 (1.21-4.36) | .011 |
| Additional IT chemotherapy | No vs yes | 0.86 (0.44-1.68) | .653 | 0.93 (0.49-1.77) | .832 |
| Age at CNS relapse | 3rd vs 1st quartile | 1.05 (0.68-1.61) | .836 | 1.11 (0.73-1.68) | .631 |
| Sex | Female vs male | 0.84 (0.41-1.73) | .629 | 0.88 (0.44-1.76) | .720 |
| Morphology variant | Blastoid vs classic | 2.38 (1.14-4.98) | .005 | 2.47 (1.18-5.20) | .010 |
| Pleomorphic vs classic | 4.85 (1.61-14.57) | 3.77 (1.32-10.79) | |||
| MIPI | Intermediate vs low | 0.47 (0.16-1.41) | .401 | 0.43 (0.14-1.32) | .273 |
| High vs low | 0.69 (0.27-1.71) | 0.79 (0.32-1.95) | |||
| CNS involvement pattern | Both vs leptomeningeal | 1.31 (0.61-2.82) | .602 | 1.25 (0.59-2.64) | .844 |
| Parenchyma vs leptomeningeal | 0.77 (0.29-2.11) | 1.13 (0.47-2.73) | |||
| Extra CNS involvement | No vs yes | 1.15 (0.53-2.47) | .730 | 1.45 (0.69-3.06) | .326 |
| CNS POD ≤24 mo | Yes vs no | 2.37 (1.18-4.77) | .015 | 2.31 (1.20-4.44) | .012 |
| Variable . | Comparisons . | OS . | PFS . | ||
|---|---|---|---|---|---|
| HR (CI 95%) . | Wald P value . | HR (CI 95%) . | Wald P value . | ||
| Cohort | BBB vs ibrutinib | 2.46 (1.24-4.90) | .010 | 2.29 (1.21-4.36) | .011 |
| Additional IT chemotherapy | No vs yes | 0.86 (0.44-1.68) | .653 | 0.93 (0.49-1.77) | .832 |
| Age at CNS relapse | 3rd vs 1st quartile | 1.05 (0.68-1.61) | .836 | 1.11 (0.73-1.68) | .631 |
| Sex | Female vs male | 0.84 (0.41-1.73) | .629 | 0.88 (0.44-1.76) | .720 |
| Morphology variant | Blastoid vs classic | 2.38 (1.14-4.98) | .005 | 2.47 (1.18-5.20) | .010 |
| Pleomorphic vs classic | 4.85 (1.61-14.57) | 3.77 (1.32-10.79) | |||
| MIPI | Intermediate vs low | 0.47 (0.16-1.41) | .401 | 0.43 (0.14-1.32) | .273 |
| High vs low | 0.69 (0.27-1.71) | 0.79 (0.32-1.95) | |||
| CNS involvement pattern | Both vs leptomeningeal | 1.31 (0.61-2.82) | .602 | 1.25 (0.59-2.64) | .844 |
| Parenchyma vs leptomeningeal | 0.77 (0.29-2.11) | 1.13 (0.47-2.73) | |||
| Extra CNS involvement | No vs yes | 1.15 (0.53-2.47) | .730 | 1.45 (0.69-3.06) | .326 |
| CNS POD ≤24 mo | Yes vs no | 2.37 (1.18-4.77) | .015 | 2.31 (1.20-4.44) | .012 |
The distributions of putative confounding factors included in the propensity scoring multivariable logistic model according to the treatment cohorts are shown in supplemental Figure 1. As shown by the nonparametric test results, the ibrutinib cohort and the BBB cohort were significantly imbalanced for patient age at relapse (median age, 67 years in the ibrutinib vs 60 years in the BBB cohort; P = .005) and ECOG performance status (ECOG 0 to 1, 79.3% in the ibrutinib vs 44.4% in the BBB cohort; P = .006). Other parameters considered putative confounding factors were well balanced. We performed a multivariable Cox analysis, including the main treatment variable (ibrutinib vs BBB crossing chemotherapy) together with propensity score and the 2 significant factors at univariable analysis (ie, morphological variant and CNS POD24 and additional IT chemotherapy). Treatment with ibrutinib was the strongest independent favorable prognostic factor for both OS (HR, 6.8; 95% CI, 2.2-21.3; P < .001) and PFS (HR, 4.6; 95% CI, 1.7-12.5; P = .002), followed by a CNS POD >24 months from previous therapy (HR for death, 2.4; 95% CI, 1.1-5.3; P = .026; HR for death or progression, 2.3; 95% CI, 1.1-4.6; P = .023) (Table 4).
Multivariate Cox regression model estimates
| Variable . | Comparisons . | OS . | PFS . | ||
|---|---|---|---|---|---|
| HR (CI 95%) . | Wald P value . | HR (CI 95%) . | Wald P value . | ||
| Cohort | BBB vs ibrutinib | 6.81 (2.18-21.25) | <.001 | 4.62 (1.72-12.45) | .002 |
| Additional IT chemotherapy | No vs yes | 1.29 (0.61-2.73) | .502 | 1.43 (0.73-2.82) | .301 |
| Morphology variant | Blastoid vs classic | 1.69 (0.73-3.92) | .118 | 1.50 (0.68-3.29) | .265 |
| Pleomorphic vs classic | 3.25 (1.02-10.36) | 2.26 (0.79-6.48) | |||
| CNS-POD ≤24 mo | Yes vs no | 2.41 (1.11-5.25) | .026 | 2.27 (1.12-4.61) | .023 |
| Variable . | Comparisons . | OS . | PFS . | ||
|---|---|---|---|---|---|
| HR (CI 95%) . | Wald P value . | HR (CI 95%) . | Wald P value . | ||
| Cohort | BBB vs ibrutinib | 6.81 (2.18-21.25) | <.001 | 4.62 (1.72-12.45) | .002 |
| Additional IT chemotherapy | No vs yes | 1.29 (0.61-2.73) | .502 | 1.43 (0.73-2.82) | .301 |
| Morphology variant | Blastoid vs classic | 1.69 (0.73-3.92) | .118 | 1.50 (0.68-3.29) | .265 |
| Pleomorphic vs classic | 3.25 (1.02-10.36) | 2.26 (0.79-6.48) | |||
| CNS-POD ≤24 mo | Yes vs no | 2.41 (1.11-5.25) | .026 | 2.27 (1.12-4.61) | .023 |
Discussion
In this large series of patients with relapsed CNS MCL, the use of ibrutinib was associated with higher response rates and longer PFS and OS than high-dose antimetabolite-based chemotherapy regimens. The median OS of 16.8 months in patients treated with ibrutinib represents a substantial and clinically meaningful improvement over the 4.4 months observed with intensive CNS-directed chemotherapy.14,38,40,41 To minimize selection bias because of nonrandom treatment assignment, we applied a propensity score adjustment in the multivariable context that revealed the difference in OS between patients treated with ibrutinib or BBB crossing regimens was even more substantial, strengthening the overall study findings. Results did not change when the 6 patients treated with bendamustine-based regimens were added to the BBB cohort.
Other than scant anecdotal reports suggesting activity of ibrutinib in CNS relapsed MCL,28,29 and the widespread knowledge of the adverse outcome of CNS involvement in MCL,14,15,38,40,41 no robust analysis has examined treatment approaches in relapsed MCL with CNS involvement in the literature to date. For this reason, we collaborated with many international sites and experts well-versed in treatment and response assessment in MCL. The rarity of CNS relapse in MCL makes performing prospective clinical trials extremely challenging. This strengthens the importance of the international collaborative retrospective series in providing the best available evidence to guide clinical management.
While high-dose antimetabolites require hospitalization and are unsuitable for elderly, frail, and comorbid patients, ibrutinib is a once-daily, oral, and active treatment suitable even for frail patients because of its relatively favorable toxicity profile. Although we did not collect detailed toxicity data, we did not observe invasive fungal infection or major bleeding adverse events. In contrast, the systemic and neurological toxicity of BBB-penetrating high-dose regimens is well described.6,40-43 We believe these data support the use of ibrutinib as a preferred therapy for patients with relapsed MCL involving the CNS who are BTK inhibitor-naïve. No patients treated with more selective covalent BTK inhibitors, acalabrutinib or zanubrutinib, were included, but these agents may also provide activity in this setting. Our analysis was not designed to assess the optimum treatment for patients developing CNS relapse while on or following a prior covalent BTK inhibitor. This represents an extremely rare and poorly studied area.
There was no survival benefit from the addition of IT chemotherapy, irrespectively of CNS-directed systemic therapy, suggesting a limited role for this treatment, which can be associated with morbidity and logistic burden. We did, however, observe that time to CNS relapse (CNS POD24) was a strong predictor of outcome, confirmed by multivariate analysis, with a median OS of the early CNS POD population of only 4.2 months. Although the use of ibrutinib was the strongest predictor of outcome in the multivariable analysis, these results parallel the well-described association of time to first POD in MCL patients and point to the importance of this clinical variable in the longitudinal clinical behavior of the disease.30,31 Finally, although evidence supporting consolidation strategies is lacking in CNS relapse of MCL, all 3 patients undergoing allogeneic SCT in our series are alive and in prolonged continuous remission. Consolidation strategies, including allogeneic SCT or chimeric antigen receptor T-cell therapy,44 may be considered in younger, fit patients with an initial favorable response to CNS-directed therapy and retain activity even in chemorefractory patients.
Our study has several limitations inherent to the retrospective analysis: the lack of uniformity in allocating treatment; unavailability of detailed toxicity data; the prolonged time frame that was needed to collect a sufficient number of cases in the 3 cohorts (over 20 years); and the absence of centralized imaging review for response assessment. Patient characteristics at CNS relapse were relatively well balanced between ibrutinib and BBB cohorts. We recognize that the BBB cohort had a numerically higher proportion of patients with increased LDH levels, more extranodal site involvement, and higher Ki67 percent concentrations. Conversely, patients in the ibrutinib cohort were older and more frequently scored <2, according to ECOG. The MIPI score, the best validated prognostic score in MCL, was slightly numerically higher in the ibrutinib cohort (69% vs 63%). In the attempt to overcome any potential bias in terms of outcomes because of unbalanced characteristics in treatment cohorts, we applied the propensity score, and the putative confounding factors have been included in the estimation. Extra CNS systemic involvement at relapse also was more frequent in the BBB cohort than in the ibrutinib cohort, but this variable has not been included in the propensity score because of unclear prognostic relevance in this clinical scenario. The year of treatment for CNS relapse was another unavailable potential bias because patients in the ibrutinib cohort were treated more recently and may have benefited from improved supportive care strategies. The era of treatment was intentionally not included in the propensity score because of methodological considerations, having been considered as a covariate that primarily predicts treatment. Nevertheless, survival rates in BBB cohort patients treated before and after 2013 were similar, and a superior survival in favor of ibrutinib was also confirmed when a comparison was made with the proportion of patients in the BBB cohort treated more recently (since 2013). The results of the survival comparison according to treatment era support a key role of ibrutinib in determining a superior outcome, irrespectively of potential bias because of different treatment periods.
Conclusions
In the present study, we described the outcome of a large cohort of patients treated with ibrutinib for CNS relapse of MCL. Despite the limitations of a retrospective analysis, ibrutinib was associated with superior survival compared with BBB penetrating chemotherapy and should be considered as a therapeutic option in the routine care of this difficult-to-treat population.
Acknowledgments
The authors thank all the patients participating in this study and all investigators and nurses of the enrolling centers for their contribution. The authors would also like to thank Rosalba Miceli from the Unit of Clinical Epidemiology and Trial Organization, Department of Applied Research and Technology Development, Fondazione IRCCS Istituto Nazionale dei Tumori, Milan, for statistical supervision, and Emilia Elizbieta Florea and Anna Bordin at the University of Verona for the precious administrative assistance. C.R. acknowledges Paolo Corradini for scientific advice and critical revision of the manuscript, Mario Lazzarino and the dearly departed Ercole Brusamolino for introducing her to clinical research, her parents Rosanna and Vittorio for teaching the value of hard work, and Dario for his love and support and the many dinners cooked.
P.K. was supported by Czech Health Research Council number AZV NU21-03-00386.
Authorship
Contribution: C.R., L.C., C.M., and C.V. conceived and designed the study; C.R., C.Y.C., T.A.E., D.T., P.K., E.G., L.C., C.M., S.B., R.L.A., C.P., M.D.-D., H.M., T.v.M., E.C., A.M.-N., M.E.W., P.A., P.N., A.A., L.M., D.M., O.V., R. Sartori, A.C., R. Sciarra, P.M.S., M.D., J.F.S., and C.V. acquired clinical data; S.I. and G.I. performed statistical analysis; C.R., T.A.E., L.C., C.M., and C.V. interpreted data and drafted the manuscript; C.Y.C., T.A.E., J.F.S., and C.V. critically revised the manuscript; and all authors discussed the results and contributed to and approved the final manuscript.
Conflict-of-interest disclosure: C.R.: Celgene: advisory board honorarium; Janssen: advisory board honorarium, congress participation; Takeda: congress participation; Roche: congress participation. T.A.E.: Roche: honorarium, advisory board honorarium; Gilead: honorarium, research support, travel to scientific conferences; KITE: advisory board honorarium; Janssen: honorarium; Abbvie: honorarium, travel to scientific conferences; AstraZeneca: honorarium, research founding; Loxo Oncology: advisory board, trial steering committee; Beigene: advisory board honorarium, research funding; Incyte: advisory board honorarium; Secura Bio: advisory board honorarium. The remaining authors declare no competing financial interests.
Correspondence: Chiara Rusconi, Division of Hematology and Bone Marrow Transplantation, Fondazione IRCCS Istituto Nazionale dei Tumori, via Giacomo Venezian 1, 20133 Milan, Italy; e-mail: chiara.rusconi@istitutotumori.mi.it.
References
Author notes
The online version of this article contains a data supplement.
There is a Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal