Abstract
Background: Glofitamab, a CD20xCD3 T-cell-engaging bispecific monoclonal antibody with a novel 2:1 (CD20:CD3) configuration, redirects T cells to eliminate normal and malignant B cells.
In ongoing Phase I/II trials, glofitamab has shown high response rates and durable complete responses in patients with relapsed/refractory (R/R) B-cell lymphomas (Hutchings et al. 2021, J Clin Oncol), including diffuse large B-cell lymphoma (DLBCL; Dickinson et al. ASCO 2022).
In this imaging study (NCT03533283), the glofitamab-mediated effects on the distribution of CD8-expressing cells, such as cytotoxic T cells, in patients with R/R B-cell non-Hodgkin lymphoma (NHL) were captured with the novel 89Zr-radiolabeled anti-CD8 humanized minibody crefmirlimab berdoxam (CD8 tracer) for positron emission tomography (PET).
Methods: Patients received obinutuzumab (1000 mg) once 7 days before the 1st glofitamab dose; glofitamab was then given as step-up doses on Day 1 (2.5 mg) and Day 8 (10 mg). The baseline infusion of the CD8 tracer was administered prior to obinutuzumab and the on-treatment infusion of the CD8 tracer was performed 48 h (±24 h) after the 1st or 2nd glofitamab dose. The tracer was infused at a fixed dose of 37 MBq (1.5 mg protein); PET/CT scans were performed 24 h post infusion. The primary endpoint was quantification of changes of CD8 tracer uptake (SUVmax) in tumor lesions before and after glofitamab treatment. Paired biopsies for CD8 immunohistochemistry (IHC) and serum pharmacokinetic samples were taken at baseline and on-treatment.
Results: To date, 9 evaluable patients with a median of 2 prior treatment lines (range: 1-6) with B-cell NHL (follicular lymphoma (FL), n=5; transformed FL, n=1; DLBCL, n=3) were enrolled. CD8 tracer infusions were well tolerated with no reported tracer-related adverse events. No difference in serum clearance of the CD8 tracer was observed between baseline and on-treatment infusions. Tracer uptake was detected in CD8 rich tissues (e.g. spleen, bone marrow, lymph nodes) and clearance organs (liver, kidney, intestine).
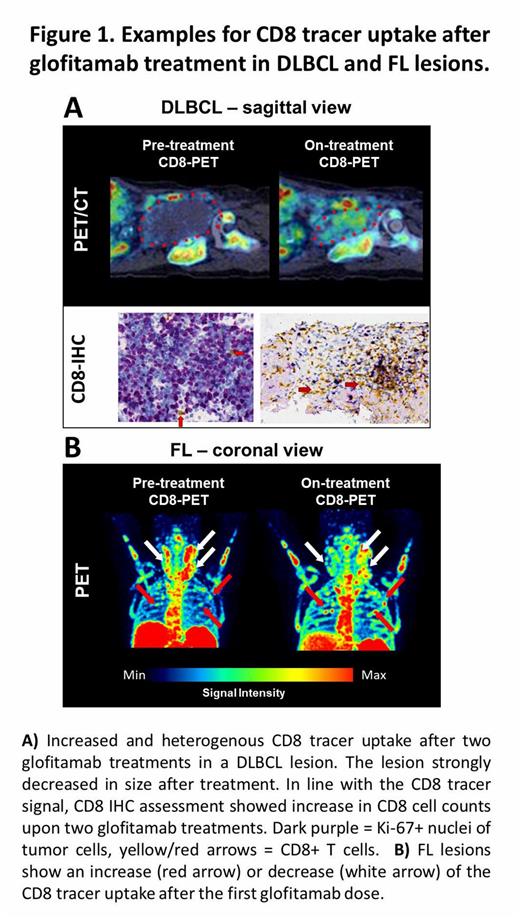
Three patients with DLBCL received on-treatment CD8-PET/CT scans after the 2nd glofitamab dose. 11 analyzed DLBCL lesions showed variable CD8 tracer uptake at baseline between SUVmax 2.6-38.27. Following treatment with glofitamab, the signal in 64% of the analyzed lesions was increased 1.2-3.3-fold while the signal in 18% of the lesions was stable and 18% showed a decreased signal compared to baseline. The distribution of the CD8 tracer in individual lesions was highly heterogeneous with areas of low and higher uptake (Fig. 1A). The CD8 IHC of paired biopsy samples taken after PET scans in 1 patient with DLBCL showed good correspondence between PET and IHC signal. The on-treatment biopsy contained quantifiable T cell amounts without detection of viable tumor tissue, suggesting a fast onset of glofitamab activity prior to the selected imaging/biopsy timepoint.
6 patients with FL were analyzed. In 87% of the analyzed 39 FL lesions, a high and heterogenous CD8-tracer uptake between SUVmax 5.3-29.2 was observed at baseline. After one dose of 2.5 mg glofitamab, 36% of the analyzed lesions showed a stable or ≤1.3-fold increase of CD8-tracer uptake. In 64% of the analyzed lesions a ≤2.1-fold decrease of CD8-tracer uptake was observed (Fig.1B). After the 2nd glofitamab dose, CD8-PET-scans of patients with FL revealed that in 86% of the analyzed lesions CD8 tracer uptake decreased 1.2-11.9-fold compared with baseline scans, accompanied by shrinkage of lesion size as detected on low dose CT.
Conclusions: Changes in the uptake of the CD8 tracer were observed early after treatment with glofitamab. Preliminary results suggest indication-specific differences in T-cell dynamics after two treatment interventions with glofitamab. In DLBCL, the CD8 tracer confirmed recruitment of T cells to the majority of tumor lesions after two glofitamab treatments. In FL, the preliminary CD8-tracer results suggest that the intra tumoral T cells exhibit their antitumoral effects immediately after glofitamab administration and thus faster compared to the glofitamab-mediated effects in DLBCL. In addition these data indicate that the optimal imaging time point is after the 1st glofitamab dose. 89Zr-crefmirlimab berdoxam is a promising innovative tool to assess the presence and dynamic change of CD8 T cells after treatment with novel immunotherapies for hematological malignancies.
Disclosures
Hutchings:Roche: Consultancy, Research Funding; Genmab: Consultancy, Research Funding; Celgene: Consultancy, Research Funding; Janssen: Consultancy, Research Funding; Incyte: Research Funding; Takeda: Consultancy, Research Funding; AbbVie: Consultancy; Novartis: Research Funding; Genentech: Research Funding; Celgene, Genentech, Genmab, Incyte, Janssen, Novartis, Roche, Takeda: Research Funding; AbbVie, Celgene, Genmab, Janssen, Roche, Takeda: Membership on an entity's Board of Directors or advisory committees. Osborne:Roche, Takeda, Pfizer, Kite Gilead, Astra Zeneca, Novartis, Kyowa Kirin: Honoraria; Takeda, Roche, Pfizer, Kite Gilead, Astra Zeneca, Novartis, Kyowa Kirin, BMS Celegene: Speakers Bureau; Roche, Takeda, Servier, Kite Gilead, MSD, Novartis, Beigene, Syneos, Autolus, Kyowa Kirin, Incyte: Membership on an entity's Board of Directors or advisory committees. Petrides:Newcastle Upon Tyne Hospitals NHS Foundation Trust: Current Employment; GE Healthcare and Gilead: Honoraria; On Alliance Medical Limited UK national PET contract governance board: Membership on an entity's Board of Directors or advisory committees; Alliance Medical Limited and GE Healthcare: Other: Fees received for scan reporting. De Vriendt:UZ Gent - University Hospital Ghent: Current Employment; Janssen, Gilead, Novartis: Consultancy; Novartis, Janssen, Takeda: Speakers Bureau; Young Hematology Academy, Janssen: Membership on an entity's Board of Directors or advisory committees; Roche: Other: Congress Registration Fee. Gritti:Genmab: Other: Advisory Board (2022); IQVIA: Other: Advisory Board (2020); Beigene: Other: Training activity (2022); Clinigen: Other: Training activity (2021); Ideogen: Other: Advisory Board (2022), Training activity (2022); Italfarmaco: Other: Advisory Board (2021); Kite-Gilead: Other: Advisory Board (2020); Sandoz: Other: Support for attending meetings (2021); Takeda: Other: Advisory Board (2020, 2021, 2022), Training activity (2020, 2022), Individual scientific consultancy (2021-ongoing); Roche: Other: Advisory Board (2021) Training activity (2020), Support for attending meetings (2021); Incyte: Other: Training activity (2022). Townsend:Takeda, Roche: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Gilead: Consultancy, Honoraria. Kayani:University College Hospital, London, UK: Current Employment. Wilson:ImaginAb Inc: Current Employment, Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees, Other: Inventor of intellectual property regarding ImaginAb technology. Wang:F. Hoffmann-La Roche Ltd: Current Employment; Peking University Third Hospital, China (2021): Ended employment in the past 24 months. Jadhav:Roche Glycart AG: Current Employment; Roche Holding AG: Current equity holder in publicly-traded company. Santini:Roche: Current Employment; Roche, BMS: Current equity holder in publicly-traded company. Kelly:Roche Products Ltd: Current Employment. Kornacker:F. Hofmann-La Roche Ltd.: Current Employment, Current equity holder in private company. Tessier:Hoffmann-La-Roche: Current Employment, Current equity holder in publicly-traded company. Bröske:Roche Diagnostics GmbH: Current Employment. Lahr:Roche: Current Employment, Ended employment in the past 24 months. Boehnke:F.Hoffman-La Roche (Roche): Current Employment, Current equity holder in publicly-traded company, Divested equity in a private or publicly-traded company in the past 24 months. Griessinger:F. Hoffmann-LaRocheLtd.: Current Employment; Roche Genusscheine: Current equity holder in private company.
OffLabel Disclosure:
Glofitamab is a full-length, humanized immunoglobulin G1 bispecific monoclonal antibody with a 2:1 (CD20:CD3) configuration that facilitates bivalent binding to CD20 on B cells, and monovalent binding to CD3 on T cells. Glofitamab redirects T cells to eliminate normal and malignant B cells. Glofitamab is an investigational agent. Obinutuzumab (Gazyva) is a CD20-directed cytolytic antibody indicated: in combination with chlorambucil, for the treatment of patients (pts) with previously untreated CLL; in combination with bendamustine followed by obinutuzumab monotherapy, for the treatment of pts with FL who relapsed after, or are refractory to, a rituximab-containing regimen; in combination with chemo followed by obinutuzumab monotherapy in pts achieving at least a PR, for the treatment of adult pts with previously untreated stage II bulky, III or IV FL.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal