Abstract
Introduction: Sickle cell disease (SCD) is the most commonly inherited monogenic hemoglobinopathy and results from a mutation in the β-globin gene (HbS), leading to red blood cell (RBC) sickling, vaso-occlusive episodes (VOE), and organ damage. Chronic hemolysis, inflammation, and repeated red blood cell transfusions in SCD can disrupt iron homeostasis. Patients who receive multiple blood transfusions develop iron-overload, and another subpopulation of SCD patients manifest iron deficiency. SCD mice exhibit increased iron absorption via upregulation of the intestinal transcription factor hypoxia inducible factor-2a(HIF-2a) leading to the development of iron overload. Disruption of intestinal HIF-2a or limiting iron intake by dietary iron restriction in SCD mice, significantly reduced systemic iron overload, lowered cellular sickle hemoglobin, and consequently improved hemolysis and ameliorated anemia. Our lab previously demonstrated that microbial signals from the gut also participated in initiation of VOE and organ damage in SCD mice. To elucidate connections between dietary iron, the microbiota-gut axis, and SCD pathogenesis, we hypothesize that dietary iron restriction may reduce microbiota load and gut permeability, and contributes to amelioration of acute and chronic complications in SCD mice.
Methods: SCD mice were generated by transplanting bone marrow cells from Berkeley SCD (Tg[Hu-miniLCRα1GγAγδβS]Hba−/− Hbb−/−) mice into 8-week old, lethally-irradiated C57BL/6J mice. Immediately after transplantation, SCD mice were fed either a regular-iron diet (Ctrl) containing 185 parts per million (ppm) iron or an otherwise identical iron-restricted diet (IRD) containing 3 ppm iron. VOE and organ damage analysis were conducted 3-4 months after treatments.
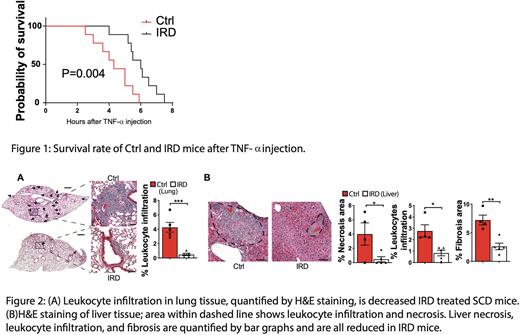
Results: IRD resulted in a reduced organ iron concentration in SCD compared to Ctrl group(0.6±0.1 v.s. 4.0±0.7ug iron/mg liver tissue; P=0.001), confirming that dietary iron restriction reverses organ iron deposition in SCD mice. SCD mice treated with IRD exhibited reduced MCHC (19.7±0.3 v.s. 20.1±0.2 pg; P=0.006) and serum indirect bilirubin (0.4±0.04 v.s. 0.7±0.07 mg/dL; P=0.001) indicating a decrease in cellular sickle hemoglobin and hemolysis. Furthermore, IRD reduced the fraction of aged neutrophils by >50% in SCD mice which corresponded with improved VOE parameters (RBC velocity: 2.0±0.2 v.s. 1.6±0.1mm/s; P=0.01; blood flow rate: 314±33 v.s. 230±13pL/s; P=0.01; leukocyte adhesion: 10±1 v.s. 14±1cells/100um vein; P=0.01; IRD v.s. Ctrl). Importantly, IRD led to prolonged survival after TNF-a challenge in SCD mice (Fig1). IRD resulted in a dramatic reduction in lung leukocyte infiltration, indicating significantly reduced lung inflammation in SCD mice (Fig2A). Compared to Ctrl liver samples, IRD resulted in significantly less liver necrosis, leukocyte infiltration and fibrosis (Fig2B). We further hypothesized that, in addition to reducing iron concentration in organs, IRD ameliorates organ damage by preventing translocation of microbial compounds. Microbiota load in IRD treated SCD mice were decreased to 25% of Ctrl level. IRD inhibited translocation of FITC-Dextran by 5-fold compared to Ctrl suggesting functionally improved gut permeability. We detected significantly decreased TLR2 ligands in serum of IRD compared to Ctrl mice (0.31±0.04 v.s. 0.46±0.05 TLR2 ligand activities O.D. 650nm; P=0.04) suggesting ligands from gram-positive bacteria (TLR2 agonists) may play a role in triggering SCD complications.
Conclusion: Our study demonstrates first time that dietary iron restriction reduces VOE severity and alleviates chronic organ damage in SCD mice. These findings provide strong evidence for the therapeutic potential of manipulating iron homeostasis and the gut microbiome to ameliorate SCD pathophysiology. Many treatments under development focus on lowering the systemic iron concentration to relieve disease complications, and our data suggest that iron-induced changes in microbiota load and gut integrity are related and novel therapeutic targets.
Disclosures
Ginzburg:Ionis: Consultancy; Takeda: Consultancy; Dexcel: Consultancy; Repare: Research Funding; Protagonist: Consultancy, Research Funding. Manwani:Global Blood Therapeutics: Consultancy; Novartis: Consultancy; Pfizer: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal