Abstract
Background: Daratumumab (Dara) combination therapy is approved in transplant ineligible newly diagnosed multiple myeloma (MM), and ongoing trials evaluating Dara in transplant eligible newly diagnosed MM may result in increased first-line (1L) use. Because Dara combinations are also approved in relapsed/refractory MM, patients may receive Dara retreatment in subsequent lines of therapy (LOTs); however, data regarding optimal sequencing and benefit of Dara retreatment are lacking. Here, we present a real-world evaluation of treatment patterns and effectiveness of retreatment with Dara-based regimens.
Methods: This retrospective observational study utilized deidentified electronic health records data from the Concert AI Patient360 database, representing primarily United States community-based oncology centers. The analysis included adult patients who met the following inclusion criteria: diagnosed with MM on or after January 1, 2011 through December 31, 2021, received ≥1 LOT postdiagnosis, had no prior stem cell transplantation, and had no other primary cancer diagnosis at baseline. Within this population, patients who received treatment with Dara (1L and beyond) and subsequent retreatment with Dara (second line [2L] and beyond) were identified. 1L was defined as all MM medications used within 35 days of MM-related medication initiation. Advancement of LOT was based on regimen change and documented progression events or stem cell transplant. The primary objective was to determine the proportion of MM patients initially treated and retreated with Dara-based regimens overall and by LOT. Key secondary objectives were to determine duration of treatment (DOT) and time to next treatment (TTNT) using the Kaplan-Meier method. A maximum follow-up date was determined for each patient using activity dates from multiple data files to identify the last active date of any medical encounter. Patients without subsequent LOT or death dates were censored at the maximum follow-up. Data were reported for the overall group and by the LOT in which initial Dara treatment was received.
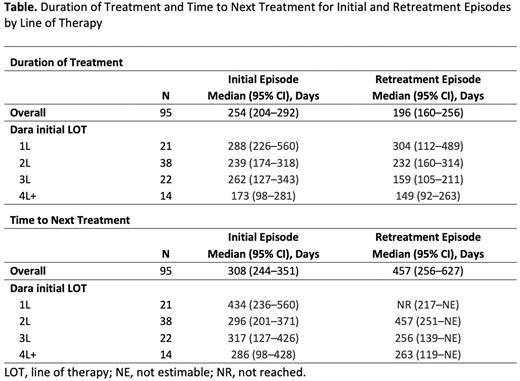
Results: Among 2367 patients with MM who met the inclusion criteria, 713 (30%) were treated with a Dara-based regimen, of which 97 (4%) were retreated with Dara and included in the analysis. Among the 97 patients analyzed, 21 (22%) started initial Dara treatment in 1L, 38 (39%) in 2L, 23 (24%) in third line (3L), and 15 (15%) in fourth or later line (4L+). Median age at diagnosis was 61-68.5 years across these groups, and 35%-50% of patients were male. Most patients had a baseline ECOG performance status of 1-2 (61%-65% for 1L, 2L, and 3L) with the exception of 4L+ (33%), and stage 2 (20%-42%) or 3 (27%-48%) disease at diagnosis. Patients were mostly treated at community (67%-81%) vs academic centers (19%-33%). Initial treatment regimens were Dara + immunomodulatory drug (IMiD) ± proteasome inhibitor (PI) (67%), Dara + PI (21%), or Dara monotherapy (12%). Dara retreatment occurred in the LOT subsequent to the initial Dara treatment episode for 90%-100% of patients; the most common retreatment regimen was Dara + IMiD (43%), followed by Dara + IMiD + PI (33%) and Dara + PI (15%). The Table shows clinical outcomes for initial Dara treatment and retreatment episodes in the same set of patients, with data by LOT indicating the line in which initial Dara treatment was received. Among 95 Dara retreated patients who had a maximum follow-up date, median DOT was 254 days and 196 days for initial Dara treatment and retreatment episodes, respectively. Median DOT by LOT for initial Dara treatment episodes ranged from 173 days (4L+ initiation) to 288 days (1L initiation). Median DOT by LOT for Dara retreatment episodes ranged from 149 days (4L+ initiation) to 304 days (1L initiation). Median TTNT was 308 days and 457 days for initial Dara treatment and retreatment episodes, respectively. Median TTNT by LOT for initial Dara treatment episodes ranged from 286 days (4L+ initiation) to 434 days (1L initiation). Median TTNT by LOT for Dara retreatment episodes ranged from 256 days (3L initiation) to not yet reached (1L initiation).
Conclusion: This analysis of real-world data from adult patients diagnosed with MM showed that DOT and TTNT for Dara retreatment episodes were reasonably long and aligned with clinical expectations for subsequent therapy, suggesting that patients continue to receive meaningful clinical benefit with Dara retreatment despite prior exposure.
Disclosures
Girvan:Eli Lilly: Current equity holder in publicly-traded company, Ended employment in the past 24 months; AbbVie: Current Employment, Current equity holder in publicly-traded company. Yu:AbbVie: Current Employment, Current equity holder in publicly-traded company; Guardant Health: Divested equity in a private or publicly-traded company in the past 24 months, Ended employment in the past 24 months. Emechebe:AbbVie: Current Employment, Current equity holder in publicly-traded company. Kamalakar:AbbVie: Current Employment, Current equity holder in publicly-traded company. Luo:AbbVie: Current Employment, Current equity holder in publicly-traded company.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal