Abstract
Background: Ruxolitinib, a potent and selective oral Janus kinase 1/2 inhibitor, is approved by the US Food and Drug Administration for treatment of intermediate and high-risk myelofibrosis (MF) and has demonstrated improvements in symptoms, spleen size, and overall survival. Although ruxolitinib treatment is associated with dose-dependent anemia and thrombocytopenia, both conditions are generally manageable and rarely lead to treatment discontinuations. The objective of this analysis was to describe the characteristics, ruxolitinib treatment patterns, and clinical outcomes of patients with MF with anemia or thrombocytopenia who received ruxolitinib in real-world clinical practice.
Methods: This was a secondary analysis of a medical chart review conducted in May 2018 that included 491 patients aged ≥18 years diagnosed with MF between January 1, 2012, and December 31, 2016. Data were abstracted from charts of patients being treated by one of 45 providers in the Cardinal Health Oncology Provider Extended Network (OPEN). Included in this analysis were patients treated with ruxolitinib as initial therapy. Anemia was defined as hemoglobin <10 g/dL at MF diagnosis; patients were also grouped based on platelet count (<50 × 109/L, 50-100 × 109/L, and >100 × 109/L) at diagnosis. Patient characteristics and treatment patterns in patients who received ruxolitinib as initial MF treatment, the subset of patients with anemia, and the subsets with low platelet counts were reported. Median duration of ruxolitinib treatment (defined as time from treatment initiation to discontinuation or end of data availability [censored at the time of data collection, May 2018], whichever occurred first) was estimated using Kaplan-Meier analysis.
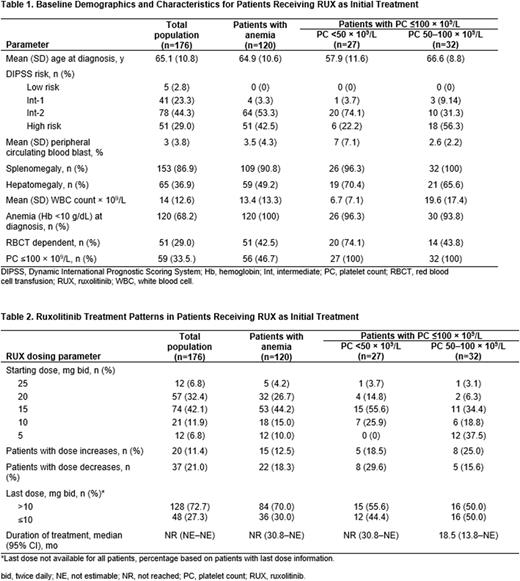
Results: Of 176 patients who received ruxolitinib as first MF treatment, 120 had anemia at diagnosis; baseline demographics and patient characteristics are shown in Table 1. Of the 120 patients with anemia, mean age was 64.9 years, 34.2% were female, and 60.0% were White; 3.3%, 53.3%, and 42.5% had intermediate-1, intermediate-2, and high-risk MF, respectively. Additionally, 42.5% were transfusion dependent per the treating physician, and 90.8% had splenomegaly (Table 1). The most common ruxolitinib starting doses were 15 mg twice daily (bid; 44.2%) and 20 mg bid (26.7%); 12.5% had dose increases, 18.3% had dose decreases, and the last ruxolitinib dose was >10 mg bid in 70% of patients (Table 2). Per the treating physician, 78.3% of patients had improved symptoms and 59.6% of those with a palpable spleen had reductions in spleen size. Ruxolitinib discontinuation occurred in 23.9% of the total population (30.8% in the anemia subset) and was rarely due to anemia or thrombocytopenia (n=1 [0.6%] for each); the most common reason for ruxolitinib treatment discontinuation was lack of or loss of response (total population, n=15; anemia subset, n=13). Overall estimated median duration of ruxolitinib treatment was not reached in either group (Table 2).
Of the 27 patients with platelet counts <50 × 109/L, the mean age at diagnosis was 57.9 years, 74.1% had intermediate-2 MF, 96.3% had splenomegaly, and 74.1% were transfusion dependent (Table 1). The most common ruxolitinib starting doses were 15 mg (55.6%) and 10 mg (25.9%) bid, 18.5% had dose increases, and 29.6% had dose decreases; most patients (55.6%) were receiving a final ruxolitinib dose >10 mg bid (Table 2). Per treating physician, 85.2% (23/27) of patients had improved symptoms and 65.4% (17/26) of those with a palpable spleen had spleen size reductions. Treatment discontinuation occurred in 18.5% of patients with platelet counts <50 × 109/L, none of which were due to anemia or thrombocytopenia (43.8% of patients with platelet counts 50-100 × 109/L discontinued treatment, which in 1 patient was due to thrombocytopenia). As was observed for patients with anemia, median duration of ruxolitinib treatment was not reached in those with platelet counts <50 × 109/L at diagnosis (95% CI, 30.8-not evaluable; Table 2).
Conclusions: In real-world practice, patients with MF with baseline anemia and thrombocytopenia can derive clinical benefits from and tolerate ruxolitinib treatment even at relatively high doses (15 or 20 mg bid). Most patients had ongoing ruxolitinib treatment for ≥2.5 years (median duration of treatment not reached) and rarely discontinued ruxolitinib owing to anemia or thrombocytopenia.
Disclosures
Verstovsek:Incyte: Consultancy, Research Funding; ItalPharma: Research Funding; Protagonist Therapeutics: Research Funding; Roche: Research Funding; Sierra Oncology: Consultancy, Research Funding; Promedior: Research Funding; PharmaEssentia: Research Funding; Novartis: Consultancy, Research Funding; NS Pharma: Research Funding; Gilead: Research Funding; Genentech: Research Funding; CTI BioPharma Corp.: Research Funding; Constellation Pharmaceuticals: Consultancy; Celgene: Consultancy, Research Funding; Blueprints Medicines Corp.: Research Funding; AstraZeneca: Research Funding; Pragmatist: Consultancy. Yu:Incyte Corporation: Current Employment, Current equity holder in publicly-traded company. Bland:Cardinal Health Specialty Solutions: Current Employment. Schuler:Cardinal Health Specialty Solutions: Current Employment. Cordaro:Incyte Corporation: Current Employment, Current equity holder in private company, Current holder of stock options in a privately-held company. Braunstein:Incyte Corporation: Current Employment, Current equity holder in publicly-traded company.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal