Sickle Cell Disease (SCD) is characterized by severe hemolytic anemia and low Hct. This contributes to elevated cerebral cerebrometabolic strain, as evidenced by increased cerebral blood flow (CBF), increased blood oxygen extraction fraction (OEF), and increased incidence of silent cerebral infarct. Recent reports have shown that approved and experimental therapies ameliorate these physiological indicators of strain in SCD patients using advanced MRI-based techniques (Fields, et al., Blood, 2019; Hulbert, et al., Blood 2022), but the effect on these same MRI endpoints has not been demonstrated in animal models of SCD. The purpose of this work was to perform these same measurements in Townes SCD mice treated with tucaresol, which is known to increase the oxygen affinity hemoglobin, thus building a translational platform for non-invasive assessment of these endpoints to support characterization of novel therapies and their relationship to cerebrometabolic strain that leads to end organ damage in the brain.
All animal procedures were performed under an IACUC-approved protocol. Forty-three male Townes SCD mice were assigned to two treatment groups (n=21 / 22, mean age = 8.1±0.8 / 8.4 ±0.6 weeks, mean weight = 26±2 / 26±2 g). Animals were anesthetized and scanned on a 9.4 T Bruker MRI scanner at MGH. CBF was measured using a pseudo-continuous arterial spin labeling sequence (pCASL) (Hirschler, et al., NMR in Biomedicine, 2018), and OEF was estimated from a T2 measurement in the superior sagittal sinus using a TRUST sequence (Wei, et al., Magn. Reson. Med., 2018) and a hematocrit measure which was taken from a tail vein bleed immediately following the scan.
Following anesthesia recovery from the first imaging timepoint, animals were placed in cages according to group assignment and drinking water was replaced by one of two masked dosing solutions. prepared off-site by an independent team, containing either 1.14 mM tucaresol or pH and color-matched water. The tucaresol concentration was chosen to approximate an oral dose of 60 mg/kg/day comparable to a clinical dose and be consistent with previous studies. After two weeks of ad lib exposure to the dosing solutions, the scanning and blood sampling procedure was repeated. A terminal cardiac punch was also performed under anesthesia to assess tucaresol blood exposure. Due to technical challenges Hct measures were not performed in the first five animals from each group, and two animals had Hct measures at only the second timepoint. CBF is reported in whole bilateral striatum.
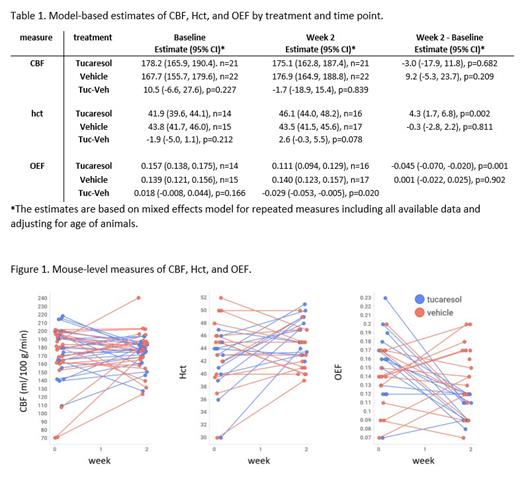
Whole blood analysis revealed tucaresol exposures of 0.1-0.6 mM in the active arm. While still within the range for hemoglobin modification, the exposures were similar but somewhat lower than the exposures of 0.5-1.6 mM reported from oral gavage at 30 mg/kg twice per day in SCD mice (Knee et al. Blood, 2016). Table 1 shows CBF, Hct, and OEF values at baseline and after two weeks of dosing for each group. OEF values assume arterial oxygen saturation of 1. Spaghetti plots in figure 1 display changes in these values for individual animals.
CBF elevations are observed very early in clinical SCD populations and begin to plateau as the disease progresses, but OEF has been shown to increase as the disease progresses (Juttukunda, et al., J. Magn. Reson. Imaging, 2019). As a result, therapeutic interventions in advanced disease may be expected to first affect OEF, with decreases in CBF to follow. That may, in part explain why a treatment effect was observed with OEF and not with CBF. Additionally, the proximity of the labeling plane of inflowing arterial blood to the brain tissue being perfused and the high carotid artery flow rate in these animals means that the CBF measure by pCASL MRI is considerably more challenging in mice than in humans.
The sensitivity of MRI-derived OEF to therapeutic intervention in mice is similar to what has been reported in SCD patients, where elevated OEF is particularly associated with regions of the brain at highest risk for developing silent infarct. CBF and OEF are currently being evaluating for an approved HbS modifier (NCT05228821). The ability to use the same techniques in both mice and humans to non-invasively and quantitatively demonstrate the amelioration of the cerebrophysiological compensatory measures that precede end CNS organ damage is a valuable step in characterizing potential benefit of novel therapeutics beyond standard regulatory endpoints associated with pain crises.
Disclosures
Goodman:Pfizer, Inc: Current Employment, Current equity holder in publicly-traded company, Current holder of stock options in a privately-held company. Yen:Pfizer, Inc: Research Funding. Chen:Pfizer, Inc: Research Funding. Chan:Pfizer, Inc: Research Funding. Weigand Wittier:Pfizer, Inc: Research Funding. Janz:Pfizer, Inc: Ended employment in the past 24 months. Arkin:Pfizer: Current Employment, Current equity holder in publicly-traded company. Rybin:Pfizer, Inc: Current Employment. Knee:Pfizer, Inc: Ended employment in the past 24 months. Pittman:Pfizer: Current Employment, Current equity holder in publicly-traded company. Mandeville:Pfizer, Inc: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal