Background: Cancer and major abdominopelvic surgeries pose significant thrombosis risks, making cancer-related surgery highly susceptible to venous thromboembolism (VTE) despite prophylaxis. Without post-operative thromboprophylaxis (TPX), VTE risk can reach 29% in the weeks following major surgeries. National Comprehensive Cancer Network (NCCN) guidelines recommend anticoagulation for up to 4 weeks after such surgeries, primarily using low molecular weight heparin (LMWH) for 28 days postoperatively. This study aimed to determine VTE rates linked to various gynecologic cancer-related surgeries and explore TPX utilization in a large cancer-center cohort. Additionally, we sought to further understand the necessity for post-discharge TPX in this setting.
Methods: Patients (pts) diagnosed with gynecologic cancer between January 1st, 2012, through September 15th, 2022, at our institution were identified through our tumor registry. Using an in-house automated text search tool from our electronic medical record (Epic®), combined with natural language processing review, venous thromboembolism (VTE) events were identified, with secondary adjudication by the study leader (GAS). VTE was defined as either lower extremity deep vein thrombosis (DVT), or pulmonary embolism (PE) diagnosed from the day of surgery until 90 days after the first day of surgery. Logistic regression was performed on binary outcomes for the presence of VTE and TPX use at discharge. Kaplan-Meier curves and Cox regression were used for overall survival analysis.
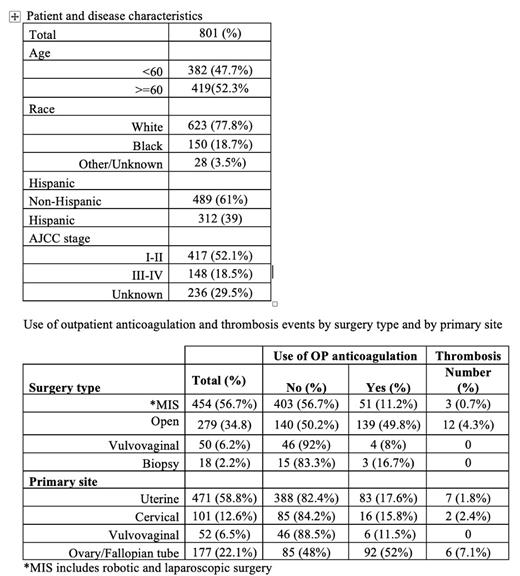
Results: 801 pts who underwent gynecologic cancer-related surgeries were identified. Patient demographics and surgery types are described in Table 1. VTE occurred in 1.9% (n=15) of the overall population but was unequally distributed. The number of VTE in those undergoing minimally invasive surgery (MIS), vulvovaginal surgery, and biopsies collectively was low (0.57%, n=3), even when considering that only 11.1% of those pts received TPX at discharge. VTE was more commonly diagnosed in patients who underwent open surgery (4.3%, n=12), with 5 pts (1.8%) diagnosed before discharge and 7 pts (2.5%) after discharge. In multivariable logistic regression analysis, during the full study period, open procedures were associated with a higher risk of thrombosis, (relative risk 7.5, p < 0.001) and higher use of post-discharge TBX (RR 4.55, p < 0.001). Notably, only 50% (139 of 279) of pts who underwent open surgery received TPX at the time of discharge. Within the open surgery cohort, the rate of post-discharge VTE correlated with the use of post-discharge TPX. Of pts on TPX, 0.7% (n=1) were diagnosed with VTE compared to 4.3% (n=6) of pts who did not receive TPX (RR=0.17, p= 0.12). There was no difference in overall survival in pts who received TPX and those who did not after open surgery up to 36 months after the surgery.
Conclusions: Our findings suggest a tiered risk of post-operative VTE after gynecologic cancer surgery, with a clinically relevant risk observed only in women undergoing open surgery. Our gynecologic oncology practice already reflects this, as post-operative TPX is not routinely prescribed for lower-risk procedures. In patients who underwent open surgery, TPX at discharge showed a trend towards a protective effect, although this result did not reach statistical significance, probably due to sample size, it supports the need for a targeted prospective study. If this trend towards benefit holds in a prospective study, the number needed to treat to avoid one clinically detected post-operative VTE would be 28 pts. The relatively high fraction of post-op VTE events during the inpatient hospital stay also supports efforts for early TPX, as has been done with a prophylactic dose of pre-operative unfractionated heparin or LMWH in other high-risk cancer surgery settings (Selby et al, J Am Coll Surg. 2016). The limitations of this study include being conducted at a single institution, retrospective and non-randomized.
Disclosures
Trabolsi:Johnson & Johnson: Current equity holder in publicly-traded company. Kronenfeld:Bayer Healthcare Pharmaceuticals: Research Funding. Caballero:Exact Sciences Corp: Current equity holder in publicly-traded company; Organon & Co: Current equity holder in publicly-traded company; Microsoft Corp: Current equity holder in publicly-traded company; Merck And Co Inc: Current equity holder in publicly-traded company. Soff:Sanofi: Consultancy; Anthos Therapeutics: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal