Background
Novel agents in chronic lymphocytic leukemia (CLL) have demonstrated improved outcomes while resulting in more complex treatment decision making. Agents vary in efficacy, tolerability, and duration of therapy, including fixed-duration (FD) therapy and treatment to progression (TTP) regimens, resulting in the need to make trade-offs in the selection of treatment. In the absence of mature overall survival data that clearly demonstrates long-term advantages of each regimen, it is important to understand the treatment preferences of patients and oncologists in the first line CLL setting.
Aims
We aimed to examine and quantify the preferences for key treatment attributes, including duration of therapy, of first line CLL therapies among oncologists and CLL patients.
Methods
Oncologists and CLL patients in the US, UK, Germany, France, and Australia were recruited into the study. They were asked to complete an online survey including a discrete choice experiment (DCE) where, in 12 tasks, participants were asked to choose between two hypothetical first-line therapies with levels that varied across five attributes: progression-free survival (PFS); risk of an adverse event (such as headache, muscle pain, or diarrhea); risk of treatment discontinuation due to side effect; FD intravenous (IV) regimens; FD and TTP oral regimens. The percentages of time each level was chosen when it appeared in a profile was reported. Bayesian models were used to estimate attribute level preference weights and attribute relative importance (RI) estimates, which capture how influential changes in an attribute are in treatment choice.
Results
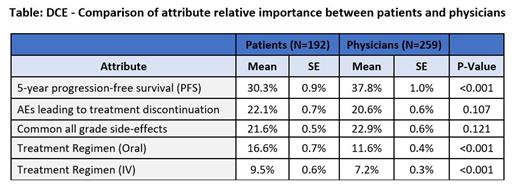
In total, 259 oncologists (74% male, mean age: 50 + 8 years, mean years of practice: 17 + 6) and 192 patients (mean age 63 + 11 years; time since diagnosis 8 + 7 years; 69% treatment experienced) participated. Patients selected ‘daily oral medication taken indefinitely’ 40% of the time, which was significantly lower than the percentage of times they selected ‘daily oral medication taken for 24 months’ (53% of the time) and ‘daily oral medication taken for 12 months’ (54% of the time) (p<0.001). Conversely, there was no significant difference in the proportions of time that FD and continuous oral medications were selected by oncologists. Efficacy was the most important attribute for both patients and physicians, although more important for physicians than for patients (p<0.001), followed by reducing the risks of the two side effect attributes, which were similar in importance. Mode of administration attributes were not as important as efficacy and safety: changes in the oral regimens (FD therapy and TTP) was the fourth most important attribute, with patients placing a greater importance on this attribute compared to physicians (p<0.001); changes among the FD therapy IV regimens was least important. Changes in the oral and IV regimens were more important for patients than for physicians (p<0.001, Table). Patients with experience with FD therapies (n=101) had a higher preference for the 12-month FD oral regimens compared to those without FD therapy experience. Those with TTP regimen experience (n=73) had a higher preference for the TTP oral regimens compared to those without prior TTP therapy experience. There was no significant difference in attribute importance according to patient's treatment history (currently treated, previously treated but off treatment now, never treated).
Conclusions
In contrast to oncologists, patients preferred FD oral therapy over TTP regimens. Efficacy and safety of the medications were important to both patients and oncologists, and both were willing to make trade-offs in relation to duration of therapy if efficacy and safety were established. Our results suggest that patients may be more sensitive to type of regimen, whether it is FD or TTP than oncologists. Understanding factors that affect the preferences and perceptions of patients is important and may lead to improved treatment outcomes and better management of adverse events, leading to greater patient satisfaction. Further work is required to explore the determinants/drivers of these preferences.
Disclosures
Sile:AstraZeneca: Consultancy, Current Employment. Horchi:Cerner Enviza an Oracle Company: Other: Cerner Enviza provides consulting services to AstraZeneca. Rault:Cerner Enviza an Oracle Company: Other: Cerner Enviza provides consulting services to AstraZeneca. Mulvihill:Cerner Enviza an Oracle Company: Other: Cerner Enviza provides consulting services to AstraZeneca; Cerner Enviza: Current Employment. Beusterien:Cerner Enviza an Oracle Company: Other: Cerner Enviza provides consulting services to AstraZeneca; Kite, a Gilead Company: Consultancy. Teschemaker:AstraZeneca: Current Employment, Current equity holder in publicly-traded company. Stewart:AstraZeneca: Current Employment. Palhares De Miranda:AstraZeneca: Current Employment, Current equity holder in private company, Current holder of stock options in a privately-held company. Guillaume:Cerner Enviza an Oracle Company: Other: Cerner Enviza provides consulting services to AstraZeneca.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal