In this issue of Blood, Buka et al have identified myeloproliferative leukemia oncogene (MPL)-mediated Janus kinase (JAK) 2 activation as a signaling mechanism through which platelet factor 4 (PF4) triggers platelet aggregation.1
In the JAK/signal transducer and activator of transcription (STAT) signaling cascade, ligand occupation of transmembrane receptors initiates activation of JAKs, tyrosine kinases that then phosphorylate STAT factors that go on to modulate gene expression. In the minds of many hematologists, JAK/STAT signaling is foremost associated with myeloproliferative neoplasms.2 Furthermore, now, almost 2 decades since the identification of the JAK2V617F mutation in polycythemia vera, there are multiple illustrations of the pathway’s clinical relevance in the pathogenesis of hematological disorders; also, there is widespread use of drugs that inhibit the pathway or act as agonists.3
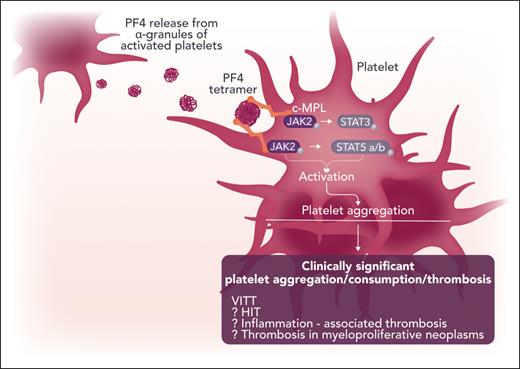
Buka et al did not set out to add to the catalogue of roles of JAK/STAT signaling in clinical hematology. Rather, they wanted to investigate the mechanism by which the chemokine PF4 induces platelet aggregation. PF4 is a chemokine released from platelet α granules that became notorious for its role in the pathogenesis of heparin-induced thrombocytopenia (HIT) and vaccine-induced immune thrombocytopenia and thrombosis (VITT).4 However, relatively little is known of the direct effect of PF4 on platelets. Hypothesizing that PF4 acts through a tyrosine kinase signaling cascade, they initially took an exploratory approach by using an antibody to phosphorylated tyrosine to compare Western blots of cell lysates from untreated and PF4-treated platelets. This revealed a 95-kDa protein tyrosine phosphorylated only in platelets treated with PF4, which was confirmed, using mass spectrometry, as STAT5. In a series of elegant experiments, they moved progressively upstream through the JAK/STAT signaling pathway to establish which pathway components were involved and had an active role in triggering platelet aggregation. First, Western blots with antibodies specific for phosphorylated JAK2 showed a dose-dependent increase in JAK2 phosphorylation with increasing PF4 concentrations. More important, blocking JAK2 activation with the JAK1/2 inhibitor ruxolitinib prevented platelet aggregation by PF4. Knowing that thrombopoietin (TPO) triggers JAK2 activation in platelets,5 they next examined whether PF4 binds to the TPO receptor MPL. Sure enough, surface plasmon resonance showed direct binding of PF4 to MPL with the dose-response relationships tracking that of JAK2 phosphorylation and platelet aggregation. Interestingly, in silico modeling suggests that PF4 binds MPL at a site adjacent to that of TPO binding. This is supported by in vitro results, which showed that PF4 reduced, but did not entirely abrogate, binding of TPO to MPL. Crucially, when MPL was blocked with an anti-MPL antibody, PF4 was unable to trigger platelet aggregation. In summary, the data support a model whereby PF4 binds to MPL and activates JAK2, leading to platelet aggregation.
In addition to this basic science mechanism, the authors show us the potential of their findings to clinical medicine using the example of VITT. In platelet aggregation triggered by serum from patients with VITT and exogenous PF4, ruxolitinib reduced platelet aggregation.
The discovery that PF4 binds to MPL and acts through JAK2 to cause platelet activation and aggregation generates several interesting questions. It is intriguing to consider that the phosphorylated STATs may play a part in platelet activation. First, what are the mediators downstream of JAK2? This work was focused strictly on tyrosine phosphorylation but other concurrent pathways, including those potentially activated by JAK2 in other kinases (eg, MAPK, extracellular signal-regulated kinase, or phosphatidylinositol 3-kinase), are likely mechanisms.
Second, MPL signaling is crucial in hematopoietic stem cells, megakaryopoiesis, and megakaryocyte function. What role does PF4 play here? This is particularly interesting in the setting of evidence that PF4 inhibits megakaryopoiesis.6 Does the microenvironment influence the contribution of PF4 to MPL signaling? For instance, locally high PF4 concentrations may occur in the vicinity of activated platelets because it is released from α granules. PF4 can be adsorbed onto cell surface and endovascular glycoproteins, which could also lead to localized increases in PF4 concentrations. On a broader scale, systemic PF4 has been identified in recent research7 as a beneficial factor (and a potential therapeutic target) in abating inflammation—and restoring immune and cognitive function with aging.
Further work is needed to fully explore the clinical significance of these findings (see figure). Outside of VITT and HIT phenomena, the role of PF4 in the TPO-MPL-JAK2 axis and consequent platelet activation is potentially relevant in any setting of chronic inflammatory disorders. Another aspect is the consequence of these findings to therapy: For example, we know that JAK inhibitors by themselves can exacerbate thrombocytopenia but without a concomitant reduction in thrombotic events.8 Therefore, it would be interesting to evaluate how PF4-mediated platelet activation contributes to risk for thrombosis in general—and how a possible interplay between the Fc γ immunoglobulin receptor (FCGR2A; studied in this work in the context of VITT) and PF4 could have a modulating effect (eg, in patients with a myeloproliferative neoplasm [MPN]). This is particularly intriguing considering the authors’ data showing reduction of PF4- (and JAK-STAT–mediated) platelet activation by ruxolitinib. Our recent MPN platelet transcriptomic data9 also point to a possible synergy between FCGR2A and PF4, and the data implicate their potential cross-linking role in MPN thrombosis risk.
Possible clinical and biological implications of the role of platelet factor 4 (PF4) in the activation of classic c-MPL and JAK-STAT pathways in the human platelet. Professional illustration by Somersault18:24.
Possible clinical and biological implications of the role of platelet factor 4 (PF4) in the activation of classic c-MPL and JAK-STAT pathways in the human platelet. Professional illustration by Somersault18:24.
Finally, this study has implications for designing drugs acting on MPL. Insights into how this novel ligand and binding site intersects with the classic TPO-mediated MPL signaling may ultimately enable us to selectively reduce or enhance signaling, which could impact diseases, such as MPNs and immune thrombocytopenia.10
Conflict-of-interest disclosure: The authors declare no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal