Autologous CD5 CAR T cells induce clinical responses in r/r mature TCLs.
CD5 CAR T cells were associated with low-grade cytokine release syndrome and neurotoxicity but no significant infectious complications.
Visual Abstract
Despite newer targeted therapies, patients with primary refractory or relapsed (r/r) T-cell lymphoma have a poor prognosis. The development of chimeric antigen receptor (CAR) T-cell platforms to treat T-cell malignancies often requires additional gene modifications to overcome fratricide because of shared T-cell antigens on normal and malignant T cells. We developed a CD5-directed CAR that produces minimal fratricide by downmodulating CD5 protein levels in transduced T cells while retaining strong cytotoxicity against CD5+ malignant cells. In our first-in-human phase 1 study (NCT0308190), second-generation autologous CD5.CAR T cells were manufactured from patients with r/r T-cell malignancies. Here, we report safety and efficacy data from a cohort of patients with mature T-cell lymphoma (TCL). Among the 17 patients with TCL enrolled, CD5 CAR T cells were successfully manufactured for 13 out of 14 attempted lines (93%) and administered to 9 (69%) patients. The overall response rate (complete remission or partial response) was 44%, with complete responses observed in 2 patients. The most common grade 3 or higher adverse events were cytopenias. No grade 3 or higher cytokine release syndrome or neurologic events occurred. Two patients died during the immediate toxicity evaluation period due to rapidly progressive disease. These results demonstrated that CD5.CAR T cells are safe and can induce clinical responses in patients with r/r CD5-expressing TCLs without eliminating endogenous T cells or increasing infectious complications. More patients and longer follow-up are needed for validation. This trial was registered at www.clinicaltrials.gov as #NCT0308190.
Introduction
Despite impressive progress in the treatment of refractory or relapsed (r/r) B-cell lymphomas, T-cell lymphomas (TCLs) have proven to be more intractable.1,2 The prognosis for patients with primary refractory or relapsed TCLs is poor, with a 3-year event-free survival of <30% across all subtypes despite intensive chemotherapies and targeted therapies.2 In these situations, hematopoietic stem cell transplantation (HSCT) often provides the only hope for long-term disease-free survival3 and is recommended in first remission for patients with high-risk disease according to the current National Comprehensive Cancer Network guidelines.2 However, current salvage regimens generally fail to induce the responses required to allow patients to proceed to undergo HSCT, and those who relapse after allogeneic HSCT (allo-HSCT) have a very poor prognosis.2
Although chimeric antigen receptor (CAR)-modified T cells have successfully treated r/r B-cell lymphoma,4,5 similar approaches for TCL are more challenging because of the complexities associated with targeting T-lineage antigens. First, the expression of T-antigen-specific CARs in T cells can promote fratricide (self-targeting) and, therefore, compromise ex vivo expansion. Second, the sustained activity of these CAR T cells in patients increases the risk of prolonged T-cell aplasia and associated infectious complications.6,7 We have previously reported the preclinical development of a CAR specific to a pan-T-cell antigen CD5, which is broadly expressed in T-cell malignancies, including various subtypes of TCLs.8 In normal tissues, CD5 is expressed on T cells, committed thymic progenitors, and a minor population of B cells, is absent on other hematopoietic and nonhematopoietic tissues, and has been targeted in T-cell malignancies using monoclonal antibody-based immunotoxins.9 We created a second-generation CD5.CAR derived from a clinically used antibody (clone H65).8 We previously showed that the expression of CD5.CAR with a CD28 costimulatory endodomain on primary human T cells via gammaretroviral transduction elicits a rapid loss of detectable CD5 on T cells, resulting in limited and transient fratricide without impairing ex vivo expansion. The resulting CD5.CAR T cells produced robust cytotoxicity in vitro and in vivo against CD5+ malignant T-cell lines and primary malignant T cells but spared a subset of normal T cells.8
Based on these findings, we conducted a first-in-human phase 1 trial to assess the safety and efficacy of escalating doses of unedited CD5.CAR T cells as a treatment for r/r CD5+ T-cell malignancies. Here, we report safety and efficacy data from a cohort of patients with mature TCL.
Methods
Study design
We conducted a phase 1 study (NCT03081910) to assess the feasibility and safety of treating pediatric and adult patients (≤75 years of age) with r/r T-cell malignancies with >50% CD5 expression in tumor cells, as confirmed by flow cytometry or immunohistochemical staining. The patients were treated with escalating doses of peripheral blood–derived autologous T cells genetically modified to express a CD5.CAR (CD5.CAR T cell). The study was designed to serve as a bridge to allo-HSCT for patients who achieved remission after CD5.CAR T-cell infusion. Patients were required to be eligible for allo-HSCT with a potential donor identified before CAR T-cell treatment. Detailed eligibility criteria are provided in the supplemental Data, available on the Blood website.
Eligible patients underwent peripheral blood collection to obtain peripheral blood mononuclear cells (PBMCs). Bridging therapy, at the discretion of the treating oncologist, was allowed while awaiting the manufacture of CAR T cells. Once sufficiently expanded to meet the assigned dose level, CAR T cells were cryopreserved, and release testing was performed. All patients treated had active disease, confirmed within 4 weeks before CD5.CAR T-cell infusion. Patients received lymphodepleting chemotherapy with fludarabine 30 mg/m2 and cyclophosphamide 500 mg/m2 for 3 consecutive days, finishing at least 48 hours before the administration of a single dose of CD5.CAR T cells. Three dose levels (DLs) were evaluated: 1 × 107 (DL1), 5 × 107 (DL2), and 1 × 108 (DL3) CAR+ cells per m2. We used a modified continual reassessment method with cohorts of 3 to guide dose escalation in all the enrolled patients.10,11 Dose-limiting toxicity (DLT) within 6 weeks of infusion was factored into the modified continual reassessment method calculations to determine the recommended dose for the subsequent cohort.
Adverse events (AEs) were graded according to the National Institutes of Health Common Terminology Criteria for Adverse Events version 4.X. We assessed the responses by imaging and pathology studies (where available) between weeks 4 and 6 after CAR T infusion. Clinical responses for the mature TCL cohort were based on the 2007 international working group revised criteria for response assessment of non-Hodgkin lymphoma.12
Based on a favorable initial safety profile, we later amended the study to allow up to 3 additional infusions of CD5.CAR T cells for patients with evidence of clinical benefit (defined as at least stable disease and/or clinical improvement) and without DLT, infection, or other safety concerns. Additional infusions of CD5.CAR T cells were at the initial cell dose or lower, if sufficient cells were unavailable.
Patients were monitored for evidence of cytokine release syndrome (CRS),13 neurotoxicity, and infectious complications. Antibacterial, antifungal, antiviral, and Pneumocystis jirovecii pneumonia prophylaxis were administered to all patients per institutional standard operating procedures. Patients received supportive care for acute or chronic toxicity, including blood components, antibiotics, and other interventions, as appropriate. Patients received no other antineoplastic agents for at least 6 weeks after T cells.
CD5.CAR T-cell generation
Preclinical development and optimization of second-generation CD5.CAR with CD28 costimulatory endodomain and a CH3 immunoglobulin 1 spacer has been reported previously.8 Patients’ PBMCs were stimulated with plate-bound CD3 and CD28 antibodies and transduced with a CD5.CAR gammaretroviral vector 3 days later, followed by expansion in the presence of recombinant interleukin-7 (IL-7) and IL-15, as previously described.14,15 CD5.CAR T-cell products were cryopreserved between days 4 and 7 after transduction. The final products were analyzed for the presence of CAR-transduced malignant cells by flow cytometry and were not infused if those population levels were detected above the release cutoff of 0.5%.
Transgene detection
Study approval
The study was conducted according to the principles of the Declaration of Helsinki. It was approved by the institutional review boards at Baylor College of Medicine and Houston Methodist Hospital and operated under an Investigational New Drug application from the Food and Drug Administration. All participants gave informed consent before enrollment.
Statistical analysis
Descriptive statistics were used to summarize the patient and clinical characteristics, laboratory data, and AEs. The data analyses cutoff date was 6 September 2022. The overall response (complete response [CR] or partial response [PR]) rate was estimated along with exact 95% binomial confidence intervals. A swimmer plot was generated to visualize the pattern of the responses of the individual subjects. Statistical analyses were performed using Statistical Analysis System (SAS) 9.2. Statistical analyses of correlative data were performed using the methods indicated in the text using the GraphPad Prism software.
Results
Patient characteristics
Seventeen patients with mature TCL were enrolled in the procurement portion of the trial, and 13 of 14 (93%) autologous CD5.CAR T cells lines were successfully manufactured. Nine patients were subsequently infused. Three lines were not initiated because of patient death (n = 2) and 1 patient proceeding to undergo HSCT. The remaining 4 patients who had CD5.CAR T cells manufactured but not infused died from progressive disease before treatment (n = 2), achieved remission with alternative therapy (n = 1), or failed eligibility (n = 1). One product manufactured from a patient with human T-lymphotropic virus 1 (HTLV-1) associated adult T-cell leukemia/lymphoma was abandoned because of malignant cell contamination. The clinical characteristics of the treated patients are shown in Table 1. All CD5.CAR T cells were manufactured from peripheral blood collected by venipuncture. We administered a single dose (n = 7) or 2 doses (n = 2) of CD5.CAR T cells to patients at 1 of the 3 dose levels (DL1, 1 × 107; DL2, 5 × 107; and DL3, 1 × 108 CAR+ cells per m2).
Baseline characteristics of the treated patients
| DL . | Patient no. . | Disease type . | Age . | Sex . | Race . | Prior no. of therapies . | Prior HSCT . | Percentage of CD5 expression (%) . |
|---|---|---|---|---|---|---|---|---|
| DL1, 1 × 107/m2 | 2 | MF/SS | 63 | F | White | 18 | Allogeneic | 51 |
| 3 | AITL | 70 | M | White | 2 | Autologous | 100 | |
| DL2, 5 × 107/m2 | 5 | AITL | 63 | F | White | 7 | Autologous | 75 |
| 6 | PTCL | 67 | M | White | 4 | No | 51 | |
| 14 | PTCL | 71 | M | White | 4 | Autologous | 100 | |
| 23 | PTCL | 48 | M | White | 10 | No | 55 | |
| DL3, 1 × 108/m2 | 28 | CTCL | 29 | F | White | 7 | No | 80 |
| 31 | PTCL | 63 | M | Asian | 5 | Allogeneic | 98 | |
| 32 | ATLL | 49 | F | Black | 5 | No | 56 |
| DL . | Patient no. . | Disease type . | Age . | Sex . | Race . | Prior no. of therapies . | Prior HSCT . | Percentage of CD5 expression (%) . |
|---|---|---|---|---|---|---|---|---|
| DL1, 1 × 107/m2 | 2 | MF/SS | 63 | F | White | 18 | Allogeneic | 51 |
| 3 | AITL | 70 | M | White | 2 | Autologous | 100 | |
| DL2, 5 × 107/m2 | 5 | AITL | 63 | F | White | 7 | Autologous | 75 |
| 6 | PTCL | 67 | M | White | 4 | No | 51 | |
| 14 | PTCL | 71 | M | White | 4 | Autologous | 100 | |
| 23 | PTCL | 48 | M | White | 10 | No | 55 | |
| DL3, 1 × 108/m2 | 28 | CTCL | 29 | F | White | 7 | No | 80 |
| 31 | PTCL | 63 | M | Asian | 5 | Allogeneic | 98 | |
| 32 | ATLL | 49 | F | Black | 5 | No | 56 |
ATLL, adult T-cell leukemia/lymphoma; CTCL, cutaneous T-cell lymphoma; F, female; M, male; MF/SS, mycosis fungoides/Sezary syndrome.
There were 5 male and 4 female patients with a median age of 63 years (range, 29-71 years) who received a median of 5 lines of prior therapy (range, 2-18). Three patients had previously undergone autologous HSCT, and 2 patients had relapsed disease after allo-HSCT. All patients had potential allo-HSCT donors identified before treatment. A median of 75% (range, 51%-100%) of the tumor cells expressed CD5, as measured by multiparameter flow cytometry (n = 6) or immunohistochemical (n = 3). The median time from enrollment (defined as the date procurement consent was obtained) to infusion was 56 days (range, 39-81). Five patients (56%) received bridging therapy within 30 days before starting lymphodepleting chemotherapy, at the discretion of their treating physician (supplemental Table 1). A minimum of 1 week separated bridging therapies and lymphodepleting chemotherapy, and all patients underwent repeat disease evaluation to confirm measurable disease before treatment. Patients received lymphodepleting chemotherapy for 3 consecutive days, except for 1 patient who developed fever, nausea/vomiting, and diarrhea on day 2, so the third day dose was not administered. CD5.CAR T cells were administered at least 48 hours after the completion of chemotherapy and infused in the outpatient setting for 8 of the 9 patients.
CD5.CAR T-cell products
The characteristics of the T-cell products are summarized in Table 2. CD5.CAR T-cell products were manufactured using either a standard expansion protocol (6-7 days between transduction and cryopreservation) or a shortened protocol (4-5 days). The median length of CD5.CAR T-cell manufacture was 5 days from gammaretroviral transduction to cryopreservation (8 days total), with an additional 14 days required for sterility assurance testing. CD5.CAR T cells had a median of 23.4-fold expansion (range, 9.2-133.3) between transduction and cryopreservation, with a median daily expansion of 4.1-fold (range, 2.3- to 19-fold). All manufactured products expressed CD5.CAR in >97% of T cells at the time of cryopreservation, with a CD4/CD8 ratio ranging from 0.1 to 1.7, and produced a median of 59.8% specific lysis (range, 43.4%-87.6%) of CD5+ Jurkat cells in a 51Cr release cytotoxicity assay. CAR-transduced malignant T cells were not detected by flow cytometry in any of the infused products. The median vector copy number per transduced T cell was 4.6 (range, 1.62-7.71).
Characteristics of infused T-cell products
| Patient no. . | Expansion after transduction, d . | Expansion after transduction, fold . | CAR transduction . | Cytotoxicity at 20:1 . | VCN per transduced cell . | Viability . | Frequency of CD4+ . | Daily expansion, fold . |
|---|---|---|---|---|---|---|---|---|
| 2 | 7 | 133.3 | 97.6% | 85.60% | 6.8 | 97.00% | 8.98% | 19.0 |
| 3 | 4 | 21.0 | 99.1% | 51.80% | 4.6 | 96.90% | 55.99% | 5.3 |
| 5 | 4 | 9.2 | 98.4% | 50.50% | 7.71 | 85.90% | 50.44% | 2.3 |
| 6 | 4 | 23.4 | 98.6% | 59.80% | 6.08 | 89.40% | 57.28% | 5.9 |
| 14 | 5 | 36.2 | 98.6% | 65.70% | 1.81 | 92.50% | 52.99% | 7.2 |
| 23 | 6 | 24.5 | 96.9% | 87.60% | 3.73 | 95.70% | 30.03% | 4.1 |
| 28 | 4 | 14.5 | 98.3% | 52.90% | 2.36 | 93.30% | 9.93% | 3.6 |
| 31 | 6 | 23.6 | 98.8% | 72.20% | 4.9 | 95.80% | 60.33% | 3.9 |
| 32 | 5 | 13.9 | 98.7% | 43.40% | 1.62 | 94.60% | 52.88% | 2.8 |
| Patient no. . | Expansion after transduction, d . | Expansion after transduction, fold . | CAR transduction . | Cytotoxicity at 20:1 . | VCN per transduced cell . | Viability . | Frequency of CD4+ . | Daily expansion, fold . |
|---|---|---|---|---|---|---|---|---|
| 2 | 7 | 133.3 | 97.6% | 85.60% | 6.8 | 97.00% | 8.98% | 19.0 |
| 3 | 4 | 21.0 | 99.1% | 51.80% | 4.6 | 96.90% | 55.99% | 5.3 |
| 5 | 4 | 9.2 | 98.4% | 50.50% | 7.71 | 85.90% | 50.44% | 2.3 |
| 6 | 4 | 23.4 | 98.6% | 59.80% | 6.08 | 89.40% | 57.28% | 5.9 |
| 14 | 5 | 36.2 | 98.6% | 65.70% | 1.81 | 92.50% | 52.99% | 7.2 |
| 23 | 6 | 24.5 | 96.9% | 87.60% | 3.73 | 95.70% | 30.03% | 4.1 |
| 28 | 4 | 14.5 | 98.3% | 52.90% | 2.36 | 93.30% | 9.93% | 3.6 |
| 31 | 6 | 23.6 | 98.8% | 72.20% | 4.9 | 95.80% | 60.33% | 3.9 |
| 32 | 5 | 13.9 | 98.7% | 43.40% | 1.62 | 94.60% | 52.88% | 2.8 |
Safety
All infusions of CD5.CAR T cells were well tolerated, with no immediate infusion-related toxicities within the first 4 hours after infusion. As anticipated, the most frequent AEs in these heavily pretreated patients was cytopenia attributable to lymphodepleting chemotherapy (Table 3). Cytopenias were transient and resolved within 28 days of infusion in 6 of 9 patients; 3 patients experienced prolonged cytopenias as detailed below.
AEs at least possibly related to the investigational agent
| AE . | Number of patients∗ (%) (N = 9) . | ||
|---|---|---|---|
| Any grade . | Grade 1-2 . | Grade ≥3 . | |
| CRS | 4 (44) | 4 (44) | 0 (0) |
| Neurologic event | |||
| Headache | 3 (33) | 3 (33) | 0 (0) |
| Confusion | 1 (11) | 1 (11) | 0 (0) |
| Delirium | 1 (11) | 1 (11) | 0 (0) |
| Hematologic | |||
| Anemia | 1 (11) | 0 (0) | 1 (11) |
| Neutrophil count decreased | 1 (11) | 0 (0) | 1 (11) |
| Platelet count decreased | 4 (44) | 0 (0) | 4 (44) |
| Lymphocyte count decreased | 1 (11) | 0 (0) | 1 (11) |
| Febrile neutropenia | 4 (44) | 0 (0) | 4 (44) |
| Cardiovascular | |||
| Hypotension | 1 (11) | 1 (11) | 0 (0) |
| Sinus tachycardia | 1 (11) | 1 (11) | 0 (0) |
| Respiratory | |||
| Dyspnea | 1 (11) | 1 (11) | 0 (0) |
| Pleural effusion | 1 (11) | 1 (11) | 0 (0) |
| Infection | |||
| Viremia | 1 (11) | 0 (0) | 1 (11) |
| Laboratory abnormality | |||
| Hypoalbuminemia | 1 (11) | 1 (11) | 0 (0) |
| Aspartate aminotransferase increased | 4 (44) | 2 (22) | 2 (22) |
| Blood bilirubin increased | 2 (22) | 1 (11) | 1 (11) |
| Alanine aminotransferase increased | 1 (11) | 0 (0) | 1 (11) |
| Alkaline phosphatase increased | 1 (11) | 1 (11) | 0 (0) |
| Other | |||
| Fatigue | 3 (33) | 2 (22) | 1 (11) |
| Fever† | 3 (33) | 3 (33) | 0 (0) |
| Edema | 1 (11) | 1 (11) | 0 (0) |
| Myalgia | 1 (11) | 1 (11) | 0 (0) |
| AE . | Number of patients∗ (%) (N = 9) . | ||
|---|---|---|---|
| Any grade . | Grade 1-2 . | Grade ≥3 . | |
| CRS | 4 (44) | 4 (44) | 0 (0) |
| Neurologic event | |||
| Headache | 3 (33) | 3 (33) | 0 (0) |
| Confusion | 1 (11) | 1 (11) | 0 (0) |
| Delirium | 1 (11) | 1 (11) | 0 (0) |
| Hematologic | |||
| Anemia | 1 (11) | 0 (0) | 1 (11) |
| Neutrophil count decreased | 1 (11) | 0 (0) | 1 (11) |
| Platelet count decreased | 4 (44) | 0 (0) | 4 (44) |
| Lymphocyte count decreased | 1 (11) | 0 (0) | 1 (11) |
| Febrile neutropenia | 4 (44) | 0 (0) | 4 (44) |
| Cardiovascular | |||
| Hypotension | 1 (11) | 1 (11) | 0 (0) |
| Sinus tachycardia | 1 (11) | 1 (11) | 0 (0) |
| Respiratory | |||
| Dyspnea | 1 (11) | 1 (11) | 0 (0) |
| Pleural effusion | 1 (11) | 1 (11) | 0 (0) |
| Infection | |||
| Viremia | 1 (11) | 0 (0) | 1 (11) |
| Laboratory abnormality | |||
| Hypoalbuminemia | 1 (11) | 1 (11) | 0 (0) |
| Aspartate aminotransferase increased | 4 (44) | 2 (22) | 2 (22) |
| Blood bilirubin increased | 2 (22) | 1 (11) | 1 (11) |
| Alanine aminotransferase increased | 1 (11) | 0 (0) | 1 (11) |
| Alkaline phosphatase increased | 1 (11) | 1 (11) | 0 (0) |
| Other | |||
| Fatigue | 3 (33) | 2 (22) | 1 (11) |
| Fever† | 3 (33) | 3 (33) | 0 (0) |
| Edema | 1 (11) | 1 (11) | 0 (0) |
| Myalgia | 1 (11) | 1 (11) | 0 (0) |
If a patient experienced multiple same AEs, only the highest grade per event listed is included in the analysis.
Two patients experienced nonneutropenic fever before CAR T infusion; 1 patient had nonneutropenic after CAR T infusion, but it was not attributed to CRS.
Other frequently reported AEs (occurring in ≥30% of patients) at least possibly attributed to CAR T cells, including fatigue, headache, elevated aspartate aminotransferase, and nonneutropenic fever (Table 3). All grade 3 chemical laboratory abnormalities improved to baseline or resolved during the 6-week toxicity monitoring period. Only 2 grade 4 nonhematologic events occurred: hyperuricemia secondary to tumor lysis syndrome and pericardial effusion requiring pericardiocentesis, both due to lymphoma progression in the same patient (no. 23).
AEs likely related to CAR T-cell infusion included CRS in 4 of 9 patients, limited to grade 2 (n = 1) or less (n = 3 [based on 2014 Lee criteria13; grading unchanged per the American Society of Transplantation and Cellular Therapy criteria]; Table 3). None of the patients experienced grade 3 or 4 CRS. The median time to onset for CRS was 2 days (range, 0.5-4), and the median duration was 2 days (range, 1-7 days). The clinical symptoms of CRS correlated with an increase in serum ferritin and/or C-reactive protein levels. CRS occurred irrespective of disease burden. A single patient developed neurotoxicity (grade 2) on day 4 after infusion, which occurred concurrently with grade 2 CRS (treated with a single dose of tocilizumab). Symptoms resolved with supportive care. This patient did not have evidence of central nervous system involvement in the lymphoma.
Severe lymphopenia (defined as an absolute lymphocyte count <200/μL) after CAR T-cell infusion occurred in 8 patients, lasting a median of 16 days (range 8-27) in all but 1 patient. Infectious complications were limited to catheter-associated bacteremias that responded to antibiotic therapy (n = 2); a urinary tract infection coinciding with bacteremia in 1 of the aforementioned patients; and reactivation of BK virus and cytomegalovirus infections, which was treated with preemptive antiviral therapy in 1 patient. No patient required administration of steroids or any other immunosuppressants to eradicate transduced CAR T cells due to toxicity.
We observed grade 3 to 4 cytopenias that persisted beyond 28 days in 3 patients. Two patients treated at DL2 (no. 6 and no. 14) experienced prolonged pancytopenia and thrombocytopenia, respectively, and 1 patient treated at DL3 (no. 31) had prolonged pancytopenia. The latter patient (no. 31) met the criteria for DLT because of severe pancytopenia persisting >56 days and required an unselected stem cell boost from his prior allogeneic stem cell donor because of ongoing transfusion dependence and minimal response to growth factor support. The cytopenias were likely related to lymphodepleting chemotherapy, although the contribution of lymphopenia is related to circulating CD5.CAR T cells could not be excluded. No patient had evidence of malignant involvement or dysplastic features on marrow analysis.
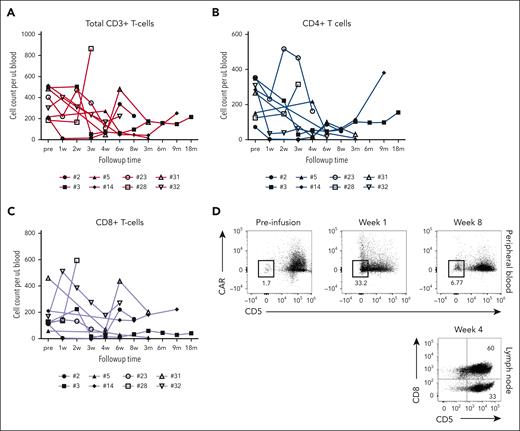
CD5.CAR T cells did not eliminate normal T cells in peripheral blood or lymph node biopsies. Circulating CD4+ and CD8+ T cells persisted after contraction of CD5.CAR T cells and comprised 7.7%-89.9% of total lymphocytes (Figure 1A-C; supplemental Figures 1 and 2). One subject (no. 6) experienced severe pancytopenia with grade 4 leukopenia and was excluded from the analysis. Most endogenous T cells in the peripheral blood and lymph node biopsies retained CD5 expression after CAR T contraction (Figure 1D). A population of CD5− T cells lacking CAR expression emerged concomitantly with the expansion of CD5.CAR T cells in the peripheral blood (Figure 1D).
Frequency of circulating T cells and CD5 expression after CD5.CAR T-cell infusion. (A) The overall frequency of total CD3+ T cells in the peripheral blood of patients after CD5.CAR T-cell infusion. Statistical significance was evaluated by one-way analysis of variance. Levels of CD3+ CD4+ (B) and CD3+ CD8+ (C) T cells in the peripheral blood. (D) Expression of CD5 and CD5.CAR in CD3+ T cells from the peripheral blood (top) and lymph node biopsy samples at the indicated time points.
Frequency of circulating T cells and CD5 expression after CD5.CAR T-cell infusion. (A) The overall frequency of total CD3+ T cells in the peripheral blood of patients after CD5.CAR T-cell infusion. Statistical significance was evaluated by one-way analysis of variance. Levels of CD3+ CD4+ (B) and CD3+ CD8+ (C) T cells in the peripheral blood. (D) Expression of CD5 and CD5.CAR in CD3+ T cells from the peripheral blood (top) and lymph node biopsy samples at the indicated time points.
Clinical responses
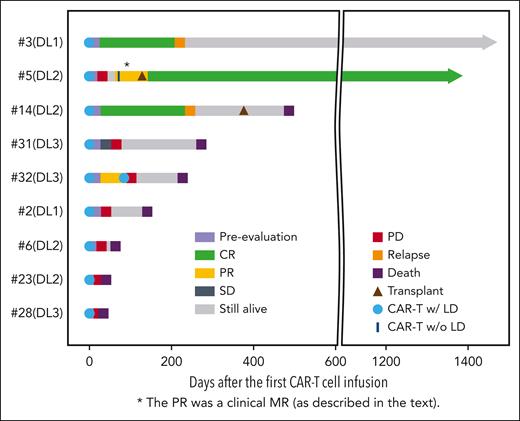
We observed objective clinical responses in 4 out of 9 patients (44%) at each DL. Four patients had progressive disease (PD) after infusion: patient no. 2 (mycosis fungoides/Sezary syndrome [MF/SS]), no. 6, no. 23, and no. 28 (peripheral TCL [PTCL]). All subsequently died of PD or complications related to PD between 4 and 17 weeks after CAR T-cell infusion. One patient with PTCL (no. 32) had stable disease at the initial disease evaluation after CAR T-cell infusion but died 9 months later in CR after receiving additional salvage therapy.
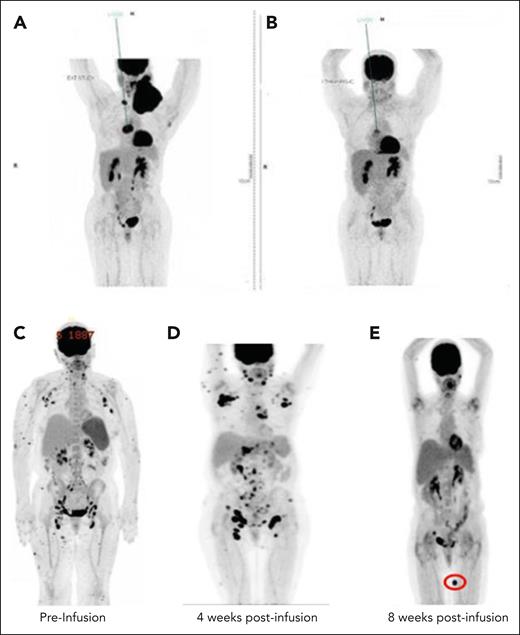
One patient with HTLV-1–driven adult TCL with bulky cervical and mediastinal involvement (no. 31; treated on DL3) achieved a partial response with significant reduction in left supraclavicular lymphadenopathy from 11.8 cm × 8.8 cm to 3.3 cm × 2.4 cm without abnormal uptake on positron emission tomography–computed tomography (PET-CT; Figure 2A-B). However, persistent activity was noted within a subcarinal/right hilar mass that was biopsy-confirmed as active disease with retained CD5 expression. Thus, the patient received a second infusion of CD5.CAR T cells at a dose of 6 × 107 cells per m2 (lower than the initial dose because of the availability of remaining cells) preceded by lymphodepleting chemotherapy for ∼2 months from the time of initial infusion. Although the second infusion was well tolerated, the patient showed disease progression and received salvage therapy. Of note, peripheral blood expansion of CD5.CAR T cells after the second infusion was minimal, and persistence was short-lived (data not shown).
Kinetics of CAR T responses by PET-CT imaging. Preinfusion (A) PET-CT of patients with ATLL treated at DL2 and postinfusion (B) PET/CT 4 weeks after CD5 CAR T-cell administration. (C) Preinfusion PET/CT of a patient with AITL (no. 5); (D) PET/CT 4 weeks after infusion showing increased markedly hypermetabolic lymph nodes and increased uptake in subcutaneous nodules; (E) PET/CT 8 weeks after CD5 CAR T-cell administration showed resolution of nearly all of the previous hypermetabolic abnormalities and appearance of 1 new FDG-avid lesion (indicated by red oval).
Kinetics of CAR T responses by PET-CT imaging. Preinfusion (A) PET-CT of patients with ATLL treated at DL2 and postinfusion (B) PET/CT 4 weeks after CD5 CAR T-cell administration. (C) Preinfusion PET/CT of a patient with AITL (no. 5); (D) PET/CT 4 weeks after infusion showing increased markedly hypermetabolic lymph nodes and increased uptake in subcutaneous nodules; (E) PET/CT 8 weeks after CD5 CAR T-cell administration showed resolution of nearly all of the previous hypermetabolic abnormalities and appearance of 1 new FDG-avid lesion (indicated by red oval).
In a patient with angioimmunoblastic T-cell lymphoma (AITL; no. 5; treated on DL2), we observed clinical resolution of cervical and axillary lymphadenopathy as well as multiple cutaneous lesions, indicating a possible disease response. However, restaging PET-CT performed 4 weeks after infusion demonstrated a marked increase in fludeoxyglucose F18 (FDG) activity (maximum standard uptake value [SUV] of up to 23.4) at the sites of previously involved nodal regions. Given the discordance between the clinical examination and imaging findings, excisional lymph node biopsies of 2 separate lymph node stations (axillary and inguinal) were obtained, revealing extensive necrosis and marked infiltration with CD5.CAR T cells. Notably, no viable malignant cells were identified. The patient was observed closely without additional antitumor therapy, and restaging PET-CT performed 8 weeks after infusion revealed interval resolution of nearly all prior FDG-avid lesions, with the appearance of a new FDG-avid lesion (Figure 2C-D) meeting the criteria for a mixed radiographic response. The patient subsequently received a second infusion of CD5.CAR T cells in an effort to consolidate the clinical response and ultimately proceeded to allo-HSCT and remained in CR 41 months after transplantation.
Another patient with AITL (no. 3) and 1 with PTCL (no. 14) achieved CR after a single dose of CD5.CAR T cells. Patient no. 14 had extensive multicompartmental lymphadenopathy above and below the diaphragm before the treatment. In accordance with the clinical protocol, patients who achieved CR after autologous CD5.CAR T cells were recommended to undergo consolidative allo-HSCT. However, both patients declined proceeding directly to HSCT, and duration of remission was 6.4 months (no. 3) and 7.2 months (no. 14), respectively. Patient no. 3 developed isolated right cervical lymph node recurrence, for which he received salvage therapy and remained alive at the time of the last follow-up, 48 months after CAR T-cell infusion. Patient no. 14, who initially presented with bulky multifocal disease, also relapsed with localized cervical lymph node disease and was treated with radiation therapy followed by allo-HSCT. Unfortunately, the subject died of transplant-related complications 99 days after the transplantation. Loss of CD5 expression in malignant cells was not observed in any patient (n = 7) who underwent biopsy at the time of disease progression or relapse.
Three patients died from lymphoma progression within 60 days of receiving CAR T cells (1 within 30 days). None of the deaths were attributed to the investigational agent or study overall. Of the remaining 6, 4 died at 4-15 months and 2 remained alive at 41 and 48 months (Figure 3).
MAGENTA disease responses. Swimmer plot for the mature TCL cohort. LD, lymphodepletion; w/, with; w/o, without. SD, stable disease.
MAGENTA disease responses. Swimmer plot for the mature TCL cohort. LD, lymphodepletion; w/, with; w/o, without. SD, stable disease.
CD5.CAR T-cell expansion and persistence
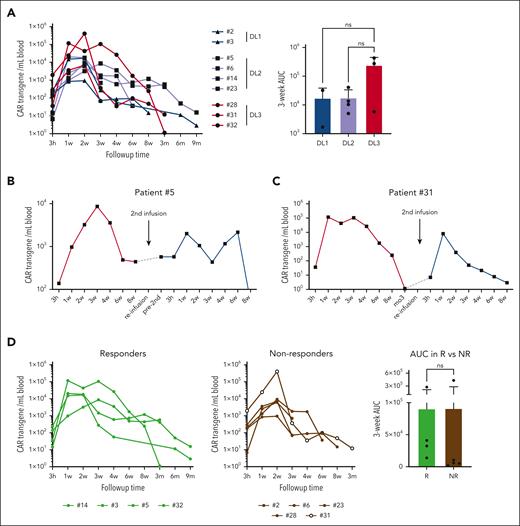
CD5.CAR T cells were detected in the peripheral blood of all infused patients starting at 3 hours after infusion, with CD5.CAR transgene copy numbers peaking between weeks 1 and 3, followed by a gradual contraction to low but detectable levels up to month 9 (Figure 4A). At the peak of expansion there was a mean of 9300 transgene copies per mL of blood (range, 938-405 955). In 2 patients (no. 5 and no. 31) who received a second CD5.CAR T-cell infusion, transgenic cells were detected in the peripheral blood up to 8 weeks after infusion, despite the lack of preceding lymphodepleting chemotherapy in subject 5 (Figure 4B-C).
Expansion and persistence of CD5.CAR T cells after infusion. (A) Levels of the CD5.CAR transgene per mL of blood. The area under the curve (AUC) for each dose level over the first 3 weeks is plotted on a bar graph. (B,C) Levels of the CD5.CAR transgene in patients no. 5 (B) and no. 31 (C), who received a second dose of CD5.CAR T cells. (D) CD5.CAR transgene levels and 3-week AUC in responders (Rs) and nonresponders (NRs). Statistical significance was evaluated using the unpaired Student t test.
Expansion and persistence of CD5.CAR T cells after infusion. (A) Levels of the CD5.CAR transgene per mL of blood. The area under the curve (AUC) for each dose level over the first 3 weeks is plotted on a bar graph. (B,C) Levels of the CD5.CAR transgene in patients no. 5 (B) and no. 31 (C), who received a second dose of CD5.CAR T cells. (D) CD5.CAR transgene levels and 3-week AUC in responders (Rs) and nonresponders (NRs). Statistical significance was evaluated using the unpaired Student t test.
Peak levels of CD5.CAR T cells and their persistence were not discernibly different between DLs or between responders and nonresponders (Figure 4D). There was no statistical difference in the CD4/CD8 ratio, cytotoxicity, expansion, or vector copy number (VCN; supplemental Figure 3).
All objective clinical responses were observed in patients treated with CD5.CAR T-cell products manufactured using the shortened protocol (4 out of 6 products), whereas none of the 3 patients treated with products manufactured using the standard protocol had an objective clinical response (supplemental Figure 4A). Shortened manufacture was associated with an increased frequency of minimally differentiated CD4+ and CD8+ T cells coexpressing CD62L and CCR7 (supplemental Figure 4B) and an elevated frequency of CD27+ T cells (supplemental Figure 4C). CD5.CAR T-cell products manufactured using the shortened method showed higher expansion and persistence after infusion (supplemental Figure 4D).
Discussion
We report the first clinical trial of nongene-edited CD5.CAR T cells for patients with r/r, CD5-expressing mature TCLs including PTCL, MF/SS, AITL, and HTLV-1-associated adult T-cell leukemia/lymphoma. We demonstrated the feasibility of generating autologous, unedited, second-generation CD5.CAR T cells from the peripheral blood of heavily pretreated patients with r/r TCL, including those who relapsed after allo-HSCT. Treatment with autologous CD5.CAR T cells demonstrated a safety profile similar to that observed after treatment with CD19.CAR T cells, including low-grade CRS, immune effector cell-associated neurotoxicity syndrome (ICANS) and prolonged cytopenias after 28 days.16 We did not observe selective T-cell aplasia (defined as an absolute CD4+ count <200 persisting beyond 8 weeks of infusion that was not present at baseline and was not related to subsequent cytotoxic therapy). Only 1 patient experienced viral reactivation requiring preemptive therapy, and no fungal or other opportunistic infections (including EBV-related lymphoproliferative disease) occurred. One patient experienced prolonged pancytopenia requiring hematopoietic stem cell rescue because of transfusion dependence and minimal response to growth factors.
CD5.CAR T-cell expansion occurred at all DLs, with the persistence of CD5.CAR T cells detected at 6.5 and 9 months in the 2 patients who achieved CR (but declined to proceed with HSCT), indicating persistence beyond 6 months, a duration that is comparable with that noted in trials of CAR T cells targeting other hematologic malignancies.14,17 Interestingly, expansion and persistence did not correlate with objective response and were not accompanied by prolonged T-cell aplasia or clinically significant infectious complications. CD5.CAR T cells were transplant-enabling in 1 patient who underwent allo-HSCT and remained in CR 41 months after. Thus, our early results suggest that although allo-HSCT should be recommended as a consolidative therapy for patients who achieve remission, it may not be an essential safeguard against prolonged T-cell aplasia. The continued persistence of endogenous circulating T cells in most patients suggests mechanisms that protect a population of normal T cells from CAR-mediated cytotoxicity. These findings are consistent with our previous in vitro observation in which CD5.CAR T cells spared a subset of autologous T cells upon prolonged coculture in conditions in which malignant cell lines were completely eliminated.8 The resistance of normal T cells to CD5-directed cytotoxicity coincides with the loss of detectable CD5 protein on the cell surface and increased expression of genes that mediate resistance to cytolysis and apoptosis.8 However, the mechanisms regulating the resistance of normal T cells to CD5.CAR T cells remain unclear. We observed no CD5 antigen loss in biopsy samples from patients who failed to respond or in those who relapsed. Thus, target antigen loss or escape was not a discernible mechanism of resistance. Continued studies in larger cohorts will help further elucidate the mechanisms of resistance.
CD5.CAR T cells trafficked to sites of active disease and were detected in available biopsy samples from the lymph nodes, bone marrow, and skin. Lymphodepleting chemotherapy followed by a single infusion of autologous CD5.CAR T cells induced objective clinical responses in 4 of 9 (44%) patients treated, including 2 CRs, 1 PR, and 1 mixed radiographic response. A repeat infusion of CD5.CAR T cells was safe in 2 patients but resulted in only transient CAR T-cell expansion and persistence.
The limited number of patients treated in this study precludes the powered statistical analyses of correlative studies. Nonetheless, all clinical responses were observed with CAR T products cryopreserved early (4-5 days) after transduction and shortened manufacture increased frequencies of minimally differentiated T-cell subsets expressing CCR7, CD62L, and CD27, which were otherwise depleted during longer ex vivo expansion due to tonic CD5.CAR signaling.8,18,19 Minimally differentiated naïve-like and central memory T cells have been associated with many of the desired properties for adoptive T-cell therapy, such as enhanced expansion and persistence and reduced exhaustion. A significant increase in the overall magnitude of expansion and initial persistence in the peripheral blood of CD5.CAR T cells manufactured using the shortened method suggest that minimally differentiated T-cell subsets were driving superior expansion. Furthermore, the high expression of the lymph node-homing receptors CCR7 and CD62L on CAR T cells may facilitate homing to lymphoma sites.20 Thus, our evidence supports the role of naïve-like and central memory T cells in enhancing systemic CD5.CAR-mediated antilymphoma activity additional studies with an expanded patient cohort are needed to validate this hypothesis.
For most patients, we were able to manufacture a CD5.CAR T-cell product within 4-5 days after transduction. However, 3 patients required longer manufacturing to reach the target cell dose, at least in part due to the reduced number of T cells in their starting material and possible poor T-cell fitness after multiple rounds of lymphotoxic chemotherapy.21 In these cases, extended CAR T manufacturing might have further reduced the antitumor potency of patients’ T cells, resulting in a lack of measurable clinical response. For these patients, procurement by leukapheresis may have increased the starting yield of PBMCs and reduced the time in culture needed for adequate expansion. The procurement of T cells earlier in the disease course may also improve autologous product fitness. Alternatively, for heavily pretreated patients or those who are unable to wait for the obligatory manufacture period, developing a CD5.CAR T-cell product premanufactured from a healthy cell source would help to overcome some of these obstacles.
Despite the challenges related to the timely treatment of this heavily pretreated population, our study supports the safety and feasibility of administering autologous CD5.CAR T cells as a therapeutic option for r/r mature TCLs. As we accumulate additional safety and efficacy data, we anticipate that, similar to CD19 CARs, their use will be considered in an earlier setting. This would likely allow both the patient and the autologous product to be more “fit.” Additionally, we are continuing to evaluate manufacture optimizations that will improve the potency of CD5 CAR T cells without increasing their toxicity. Although the highest DL (DL3) was found to be safe in the small cohort of patients with TCL reported here, the future dose for the phase 2 investigation will be based on composite data, including patients with mature TCL and T-cell acute lymphoblastic leukemia/lymphoblastic lymphoma. Additional data and long-term follow-up will be required to determine the true potential benefit of CD5.CAR T-cell therapy, including its impact on progression-free survival.
Acknowledgments
The authors thank all the medical staff at Houston Methodist Hospital for their care of the patients.
This study was supported (in part) by research funding from National Institutes of Health (NIH), National Cancer Institute (NCI) grant P50CA126752 and the Good Manufacturing Practices (GMP) facility was supported by NIH, NCI grant P30CA125123. Support was also provided by the National Gene Vector Biorepository at Indiana University, which is funded under NIH, NCI contract HSN261201500003I task order no. HHSN26100077.
Authorship
Contribution: R.H.R. and L.C.H. designed and supervised the clinical trial; H.Z., B.M., and N.L. oversaw the CAR T-cell production and quality control; Z.M. manufactured the clinical grade vector; B.G., H.E.H., and M.K.B. served as IND sponsors; L.C.H., C.A.R., and P.L. recruited and provided patient care; M.A. provided research coordination support; M.M. oversaw GMP manufacturing and clinical release of lines; L.C.H., M.M., M.J.W., and T.W. collected and/or analyzed clinical data and performed statistical analyses; T.S.S., L.Y., M.M., and M.S. performed release testing and correlative studies; L.C.H., R.H.R., and M.M. wrote the manuscript; L.C.H., R.H.R., H.E.H., M.K.B., and M.M. provided input on data interpretation and revised the manuscript; and all authors have approved the final version of the manuscript and are accountable for all aspects of the work.
Conflict-of-interest disclosure: M.M. and M.K.B. are the cofounders of March Biosciences. L.C.H., H.E.H., M.M., and M.K.B. served on the advisory board of the March Biosciences. M.K.B. and H.E.H. are cofounders and equity holders at AlloVir Inc and Marker Therapeutics. M.K.B. has equity in Tessa Therapeutics Ltd and March Biosciences, and serves on advisory boards for Marker Therapeutics, Allogene, Walking Fish, Abintus, Tessa Therapeutics, Athenex, Onk Therapeutics, Coya Therapeutics, Triumvira, Adaptimmune, Vor Therapeutics, and Tscan. H.E.H. has served on advisory boards for Gilead Biosciences, Novartis, PACT Pharma, Mesoblast, Kiadis, and Tessa Therapeutics and has received research support from Tessa Therapeutics and Kuur Therapeutics. M.M. serves on the scientific advisory board of NKILT Therapeutics and receives research support from Fate Therapeutics. R.H.R. has served as a consultant for Pfizer, received honoraria from Novartis, and research support from Tessa Therapeutics. B.G. owns QB Regulatory Consulting, which is in agreement with March Biosciences. C.A.R. has participated in advisory boards for Novartis, Genentech, and CRISPR Therapeutics, and has received research funding from Athenex and Tessa Therapeutics. P.L. has received clinical trial funding from Marker Therapeutics and AlloVir Inc. The remaining authors declare no competing financial interests.
Correspondence: LaQuisa C. Hill, Hematology Oncology, Center for Cell and Gene Therapy/Houston Methodist Hospital, 6565 Fannin St, Suite A6-080, Houston, TX 77030; email: laquisa.hill@bcm.edu.
References
Author notes
L.C.H. and R.H.R. contributed equally to this work.
Data are available upon reasonable request from the author, LaQuisa C. Hill (laquisa.hill@bcm.edu).
The online version of this article contains a data supplement.
There is a Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal