In this issue of Blood, Thorsson et al report a comprehensive single-cell multiomics analysis of 23 childhood B-cell progenitor acute lymphoblastic leukemia (BCP-ALL) cases, identifying subtype-specific maturation patterns and therapeutic targets, in particular the phosphatidylinositol 3-kinase (PI3K) pathway and CD371 (see figure) in the DUX4-rearranged (DUX4-r) subtype that offer potential opportunities for novel treatment approaches.1
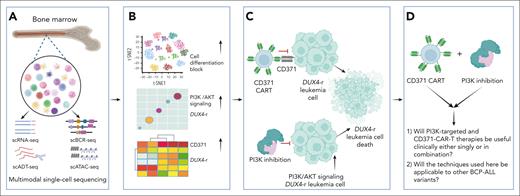
Molecular mechanisms and therapeutic research of BCP-ALL disease. (A) Single-cell sequencing analyzes pediatric BCP-ALL’s molecular mechanisms in leukemia. (B) BCP-ALL blocks cell differentiation and boosts PI3K/AKT and CD371 in DUX4-r. (C) New therapeutic research targeting PI3K and designing CD371 CAR T-cell therapy. (D) Outstanding questions. ADT, antibody-derived tag; ATAC, assay for transposase-accessible chromatin; BCR, B-cell receptor; sc, single cell; seq, sequencing.
Molecular mechanisms and therapeutic research of BCP-ALL disease. (A) Single-cell sequencing analyzes pediatric BCP-ALL’s molecular mechanisms in leukemia. (B) BCP-ALL blocks cell differentiation and boosts PI3K/AKT and CD371 in DUX4-r. (C) New therapeutic research targeting PI3K and designing CD371 CAR T-cell therapy. (D) Outstanding questions. ADT, antibody-derived tag; ATAC, assay for transposase-accessible chromatin; BCR, B-cell receptor; sc, single cell; seq, sequencing.
BCP-ALL, the leading cause of cancer in children, is triggered by genetic and epigenetic alterations.2-4 Though treatment outcomes are generally favorable, the associated toxicities and potential for long-term side effects are significant.5,6 Advances in kinase inhibitors and CD19-targeted chimeric antigen receptor (CAR) T-cell therapies have shown promise in patients with relapsed or refractory disease,7-9 but some patients fail to have a sustained response due to CD19 loss. Thus, novel, less toxic treatments for these patients are needed.
In this study, Thorsson et al explored cell differentiation states in different BCP-ALL subtypes through multiomics single-cell techniques. In particular, they focused on leukemia with DUX4 fusions, investigating PI3K inhibitors and the development of CD371 CAR T cells for the treatment of DUX4-fusion leukemia.
The authors employed multimodal single-cell sequencing to illuminate the genetic and epigenetic heterogeneity across BCP-ALLs, belonging to the BCR::ABL1-positive, ETV6::RUNX1-positive, high hyperdiploid, and DUX4-rearranged (DUX4-r) subtypes. Profiling over 188 000 cells revealed unique T-cell clusters in DUX4-r that activate pathways involved in nonlymphoid lineages and deregulate programs involved in lymphoid development. These findings enhance our understanding of BCP-ALL’s cellular complexity and might guide the development of more targeted therapies. Interestingly, the study found that CD4+ T cells exhibit abnormal expression in patients with DUX4, a finding that needs further exploration. The specific role of these abnormal CD4+ T cells, if any, in the development of DUX4 leukemia remains to be determined. Future research could isolate the abnormal CD4+ T cells for genomic and epigenetic sequencing to observe changes and coculture them with DUX4 leukemia cells for further mechanistic and pathogenic exploration.
Thorsson et al expanded on the normal bone marrow annotations to create a comprehensive next K-nearest-neighbor graphs, offering a detailed trajectory of B-cell development. This innovative approach allows for precise tracking of BCR rearrangements, and the mapping of BCP-ALL blast cells revealed a wide range of maturation stages. Subtypes like BCR::ABL1 and ETV6::RUNX1 were found to align with early B-cell stages, and high hyperdiploid cases showed a maturation path akin to normal B cells. In contrast, DUX4-r ALLs presented a more complex scenario, with cells displaying characteristics of both pro-B and mature B cells, suggesting a more advanced cellular transformation. The discovery of DUX4-r cells adopting nonlymphoid transcriptional features all demonstrated that there was lineage infidelity. This single-cell profiling enhances our understanding of BCP-ALL’s cellular heterogeneity and highlights new directions for therapeutic intervention. Although the potential of DUX4-r ALL cells to transdifferentiate into multiple cell lineages has been identified, the specific molecular mechanisms remain unclear. Future research can delve deeper into the molecular mechanisms behind this phenomenon.
The authors assessed the differential gene expression analysis in DUX4-r ALL blast cells, which revealed the overexpression of 871 genes, including AGAP1, GATA3, S100A16, and MCAM, associated with open chromatin. This finding indicates that there is activation of nonlymphoid lineage pathways and deregulation of lymphoid development programs in DUX4-r cells. Notably, the PI3K/AKT pathway was upregulated. Encouragingly, the DUX4-r cell line NALM6 demonstrated heightened sensitivity to the PI3K inhibitors alpelisib and copanlisib, as evidenced both by in vitro assays and within a patient-derived xenograft (PDX) model. The study suggests the potential of PI3K inhibitor treatment for the DUX4-r subtype, but given the short duration of PDX models, typically ∼21 days, the potential development of drug resistance with long-term use was not addressed.
In this study, the reclassification of DUX4-r blast cells into homogeneous and heterogeneous subgroups helped to uncover unique gene signatures and immunophenotypes in leukemia cells. Heterogeneous blasts, resembling the DUX4-b class, showed myeloid potential. The enforced expression of CEBPA in the DUX4-r-positive NALM6 cell induced a monocytic phenotype, evidenced by increased expression of CD371 and decreased CD19 that might help explain relapsed and refractory DUX4-r leukemia. All cases displayed signs of monocytic and T-cell lineage potential. The current study conducted a bioinformatics analysis of gene expression. This investigation has pinpointed a multitude of cell surface markers, opening avenues for future studies to delve into their functional significance and their implications in the trajectory of disease progression.
Finally, the authors identified CD371 as a potential therapeutic target in DUX4-r due to its expression on both lymphoid and monocytic blast cells,10 a characteristic of lineage infidelity in DUX4-r cases. The authors developed second-generation CAR T cells targeting CD371 and demonstrated their efficacy against the CD371-positive cell line NALM6 both in vitro and in vivo using PDX models. The results showed that CD371 CAR T cells effectively reduced leukemic cells, comparable to CD19 CAR T cells, indicating the promise of CD371 as a target for CAR T-cell therapy in this BCP-ALL subtype. It is well known that failure of CAR T-cell therapy targeting CD19 may be due to the loss of CD19 expression in leukemia cells. Whether the CD371 CAR T-cell therapy proposed in the study will also encounter similar issues needs further exploration.
In conclusion, this important study by Thorsson et al demonstrates that the multimodal single-cell analysis of BCP-ALL provided detailed insights into the differentiation block in common subtypes and revealed distinct maturation patterns and lineage infidelity in DUX4-r. It also identified the sensitivity of DUX4-r to PI3K inhibitors and CD371-targeted CAR T cells. The methodology will undoubtedly aid in finding more precise, less toxic BCP-ALL treatments beyond DUX4-type.
Conflict-of-interest disclosure: The authors declare no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal