In this issue of Blood, Chen et al provide new insight into the progression of acute kidney injury (AKI) to chronic kidney disease (CKD) using a mouse model of sickle cell disease (SCD).1 The authors report a progressive loss of kidney function over time in sickle mice. Moreover, AKI could be induced in young sickle mice with repeated doses of heme. The kidneys from both aged and heme-treated young sickle mice had reduced expression of endothelial protein C receptor (EPCR) and increased soluble EPCR (sEPCR) in the plasma. Notably, overexpression of EPCR partially protected young SCD mice from heme-mediated kidney injury, suggesting that preserving EPCR levels in the kidney may protect against the development of CKD.
SCD is a hematologic disorder caused by a point mutation in the β-globin gene, generating sickle hemoglobin (HbS). HbS polymerizes in low oxygen conditions, causing red blood cell sickling, hemolysis, and aggregation, resulting in vaso-occlusive events. Individuals with SCD also develop cumulative end-organ damage. Sickle nephropathy is a progressive condition characterized by persistent albuminuria, which is present in 30% of pediatric patients2 and up to 60% of adult patients, leading to progressive decline in kidney function and irreversible kidney damage.3 CKD is a consistent predictor of early mortality in this population.4 Both hemodialysis and peritoneal dialysis can be initiated when glomerular filtration rate (GFR) drops significantly, yet poor peripheral venous access can limit the utility of these therapies. Renal transplant is reserved for the most advanced cases of end-stage renal disease due to risk of graft failure or sepsis; recently, however, posttransplant survival has improved and is now comparable with that seen in patients with diabetes. Specific CKD-directed therapies being investigated in SCD animal models and clinical trials in SCD patients include angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, mineralocorticoid receptor antagonists, endothelin receptor antagonists, and pyruvate kinase activators. Preliminary results from the studies are still inconclusive.3 The data presented by Chen et al represent a step toward identifying novel mechanisms and therapeutic targets for CKD in SCD.
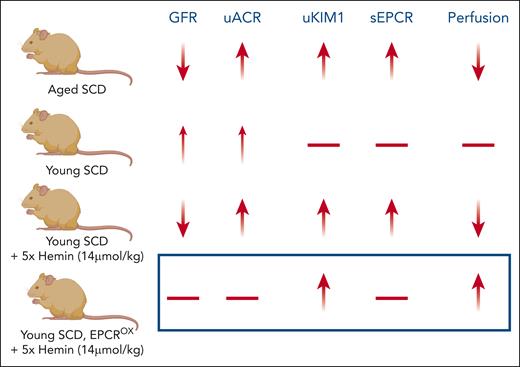
The Townes mouse model of SCD exhibits a similar timeline of CKD, presenting with early elevated GFR (hyperfiltration) that steadily decreases with age, while developing concomitant proteinuria, as seen in humans.5 Chen et al confirm this finding and also demonstrate age-dependent decreases in vascular density in the cortex and corticomedullary regions, measured by super-resolution contrast imaging. In previous work, the authors demonstrated that hemolysis and extracellular heme trigger renal vascular congestion.6 In the current work, they demonstrate that repeat dosing of heme decreases GFR and increases albuminuria, even in young sickle mice. The novel observation presented is that the renal vasculature of heme-infused young and aged sickle mice has decreased expression of the endothelial protein C receptor (EPCR), and elevated levels of soluble EPCR (sEPCR) in the plasma, suggesting shedding of EPCR from the renal endothelium.
EPCR regulates vascular homeostasis by facilitating the activation of protein C to activated protein C (APC), enabling APC to mediate anti-inflammatory and barrier protective signaling through biased agonism of protease-activated receptor 1 (PAR1).7 Recent evidence from our lab implicates a protective role for EPCR-APC-PAR1–biased signaling against thromboinflammation and vascular congestion in the liver and kidneys of sickle mice.8 Several recent studies have implicated the loss of cellular EPCR expression in inflammation, endothelial dysfunction, renal injury, and cardiovascular disease in models of diabetes9 and cardiac ischemia-reperfusion injury.10
Chen et al tested the hypothesis that stabilizing endothelial EPCR levels will preserve renal function in heme-treated young sickle mice. Sickle cell mice with EPCR overexpression were generated by bone marrow transplantation and challenged with heme. This approach not only reduced the amount of EPCR shedding but also preserved renal vascular density and kidney function (see figure). Interestingly, a cohort of sickle patients with CKD had elevated sEPCR levels and total plasma heme compared with sickle patients without CKD. The authors suggest that elevated sEPCR levels could be a predictive biomarker for CKD, and that preserving endothelial EPCR expression may be a beneficial therapeutic target to prevent or treat kidney injury in SCD patients.
Although Chen et al suggest a contribution of a new pathway to the development of CKD in SCD, several questions remain unanswered. For instance, the exact mechanism by which heme initiates the shedding of EPCR from the renal vasculature is unclear. In addition, it is not known whether all circulating sEPCR originates from the kidneys or if other organs contribute to its levels. The connection between EPCR loss and renal injury also needs further exploration. One hypothesis is that EPCR shedding leads to insufficient APC generation, thereby attenuating the endogenous anticoagulation system. Another possibility is that reduced EPCR levels diminish the anti-inflammatory and cytoprotective signaling of APC-PAR1. Accordingly, our laboratory has demonstrated that inhibiting APC exacerbates thrombin generation and inflammation in Townes sickle mice, and that endogenous APC-PAR1–biased signaling attenuates thromboinflammation and vascular congestion in these mice.8 In addition, although preserving EPCR expression has been shown to maintain GFR, it had no impact on tubular epithelial injury, raising the question of whether this preservation alone is sufficient to prevent kidney failure. These gaps highlight the need for further research to fully understand the role of EPCR and its shedding in the pathogenesis of CKD in SCD.
The recent approval of 2 gene therapy strategies is a welcome addition to the limited therapeutic options available to individuals with SCD, yet their effect on CKD and multiorgan failure is currently unknown.3 It is crucial to identify therapeutic strategies to prevent organ failure in sickle patients who are awaiting or ineligible for transformative treatments such as gene therapy and bone marrow transplantation. In their work, Chen et al advance our understanding of CKD mechanisms in SCD and points toward novel therapeutic targets to mitigate kidney injury in these patients. Future research should focus on translating these findings into clinical practice and exploring the long-term benefits of these therapeutic interventions in the broader sickle cell patient population.
A summary of the key results from Chen et al, in which kidney function was assessed in 10-month-old (Aged) mice with SCD as a decrease in glomerular filtration rate (GFR), increased uACR, increased uKIM1 levels, and decreased renal perfusion. Kidney injury was associated with increased sEPCR in plasma. In contrast, 4- to 6-week-old (Young) SCD mice had modest kidney injury, which was exacerbated by repeat doses of hemin. EPCROX reversed some markers of heme-induced kidney injury in young SCD mice. EPCROX, overexpression of endothelial protein C receptor; sEPCR, soluble endothelial protein C receptor; SCD, sickle cell disease; uACR, urinary albumin:creatinine ratio; uKIM1, urinary kidney injury molecule 1. Figure created with BioRender.com.
A summary of the key results from Chen et al, in which kidney function was assessed in 10-month-old (Aged) mice with SCD as a decrease in glomerular filtration rate (GFR), increased uACR, increased uKIM1 levels, and decreased renal perfusion. Kidney injury was associated with increased sEPCR in plasma. In contrast, 4- to 6-week-old (Young) SCD mice had modest kidney injury, which was exacerbated by repeat doses of hemin. EPCROX reversed some markers of heme-induced kidney injury in young SCD mice. EPCROX, overexpression of endothelial protein C receptor; sEPCR, soluble endothelial protein C receptor; SCD, sickle cell disease; uACR, urinary albumin:creatinine ratio; uKIM1, urinary kidney injury molecule 1. Figure created with BioRender.com.
Conflict-of-interest disclosure: E.M.S. declares no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal