Abstract
To clarify the role of γN-terminal Gly, γ5 Glu, and γ143 Ser in 2, 3-biphosphosphoglycerate (BPG) binding to fetal hemoglobin (Hb F ), we engineered and produced normal human Hb F and two Hb F variants (Hb F γG1V, γS143H, and Hb F γG1V, γE5P, γS143H) using a yeast expression system and then compared their oxygen-binding properties with those of native human Hb F and adult Hb (Hb A). Oxygen affinity of Hb F γG1V, γS143H in the absence of 2, 3-BPG was slightly higher than that of normal Hb F. The decrease in oxygen affinities for Hb F γG1V, γS143H with increasing 2, 3-BPG concentrations was larger than that of normal Hb F, but significantly less than that of Hb A. In contrast, oxygen affinities of Hb F γG1V, γE5P, γS143H in the absence and presence of 2, 3-BPG were much lower than those of Hb F γG1V, γS143H and were similar to those of Hb A. These results indicate that differences between Pro and Glu at the A2 position in the A helix in Hb A and Hb F, respectively, are critical for reduced binding of 2, 3-BPG to Hb F, even though β5 Pro does not interact directly with 2, 3-BPG in Hb A. Hb F variants such as Hb F γG1V, γE5P, γS143H, which exhibit reduced oxygen affinity, should facilitate design of efficient antisickling fetal Hb variants for potential use in gene therapy for sickle cell disease.
OXYGEN AFFINITY of fetal blood is higher than that of maternal blood and may help to provide the fetus with an adequate oxygen supply during pregnancy. In mammals, 2, 3-biphosphosphoglycerate (BPG) mediates intracellular hemoglobin function by lowering oxygen affinity. Fetal red blood cells have higher affinity for oxygen because of a smaller effect of 2, 3-BPG on oxygen affinity of fetal hemoglobin (Hb F ) compared with adult Hb (Hb A).1-5 Hb A is much more sensitive to 2, 3-BPG, so that fetal red blood cells that contain Hb F and normal levels of 2, 3-BPG have significantly higher oxygen affinity than adult red blood cells containing Hb A. The higher affinity is in part due to decreased binding of 2, 3-BPG to Hb F, which results in higher oxygen affinity than Hb A in red blood cells, even though the affinity of purified Hb F for oxygen in the absence of 2, 3-BPG is slightly lower than that of Hb A.1,2,4,5 The phosphate groups of 2, 3-BPG form salt bonds with the β N-terminus and the β2 and β143 His imidazoles in addition to binding of the carboxyl group of 2, 3-BPG to the ε-amino group of β82 Lys in Hb A.6 Differences between the β and γ chains at amino acids 1 and 143 include β1 Val versus γ1 Gly and β143 His versus γ143 Ser and are thought to contribute to the reduced binding of 2, 3-BPG.1,5 In addition, the stereo-chemical basis of this decreased interaction in Hb F has been explained by x-ray analysis and may be the result of the increased distance between the phosphate groups of 2, 3-BPG and γ2 His in Hb F.6 X-ray diffraction studies also showed that structural differences between the γ and β chains do not cause any gross functional perturbations7; however, differences in primary structure of the γ and β chains might help explain the lower binding to 2, 3-BPG of Hb F. We can now assess reasons for differences between Hb A and Hb F by preparing Hb A and Hb F variants and studying their oxygen-binding properties using recombinant DNA technology. Because Hb F inhibits polymerization of Hb S, these basic studies should define the best antisickling Hb F variants with desired oxygen affinity, stability, and thermodynamic characteristics for potential use in gene therapy for sickle cell disease. In this report, we define amino acids responsible for the higher oxygen affinity of Hb F in the presence of 2, 3-BPG compared with Hb A.
MATERIALS AND METHODS
The Gγ-globin cDNA (0.44 kb) was obtained from a cDNA library made from mRNA isolated from K562 cells. The shuttle vector, pGS 200-γ, containing full-length human Gγ-globin cDNA, was created by overlap polymerase chain reaction (PCR) from pGS189, which contains human β-globin cDNA, by exchanging the Xho I β-globin cDNA fragment with the Gγ-globin cDNA (1.4 kb) region.8 Plasmids containing Val-γ1, His-γ143 or Val-γ1, Pro-γ5, His-γ143 γ-globin cDNAs were constructed using PCR mutagenesis and subcloning, as described previously.8,9 The complete coding sequence of the wild-type and mutated Gγ-globin cDNAs flanked by the GGAP promoter and MFα3′-UN regions was determined using site-specific primers and fluorescently tagged terminators in a cycle sequencing reaction in which extension products were analyzed on an automated laser-activated fluorescence-emission DNA sequencer.9 The entire γ-globin cDNA region was excised by Xho I digestion and substituted for the β-globin cDNA region by subcloning into the Xho I site of the expression vector pGS389.9 The resultant vector contains full-length human cDNAs for α and Gγ-globin variants under transcriptional control of dual pGGAP promoters, as well as a partially functional yeast LEU2d gene and the URA3 gene for plasmid amplification and selection in yeast.8
Expression in yeast and isolation of the Hb F variants were as described previously.8,9 The purified Hb F variants were subjected to electrospray mass analysis (Fisons Instruments, VG Biotech, Altricham, UK) using the multiply charged ion peaks from the α-globin chain (molecular weight = 15,126.4 Daltons) as an external reference for mass scale calibrations.10 Absorption spectra of the purified Hb variants were recorded using a Hitachi U-2000 spectrophotometer (Hitachi Instruments, Inc, Danbury, CT). Circular dichroism spectra of the variants were recorded using an Aviv-Model 62 DS instrument (Varian Analytical Instruments, San Fernando, CA) using a 0.1-cm light path cuvette at approximately 10 μmol/L Hb concentrations. Methods for determination of kinetics of polymerization of deoxyhemoglobins in 1.8 mol/L phosphate buffers using the temperature-jump method were as reported previously.11 Oxygen dissociation curves were determined in 50 mmol/L Bis-Tris containing 5 mmol/L EDTA, pH 7.2, at 20°C using a Hemox Analyzer (TCS Med Co, Huntington Valley, PA).12
RESULTS AND DISCUSSION
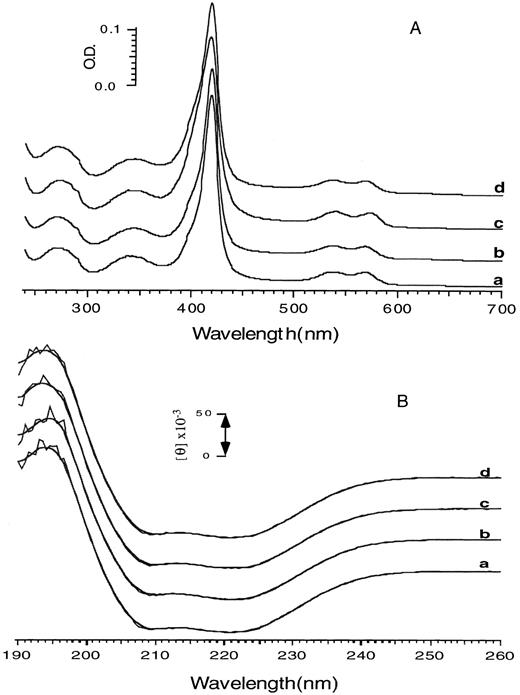
To clarify the role of γN-terminal Gly, γ5 Glu, and γ143 Ser in 2, 3-BPG binding to Hb F, we engineered and produced normal human Hb F and two Hb F variants (Hb F γG1V, γS143H, and Hb F γG1V, γE5P, γS143H) using a yeast expression system and then compared their oxygen-binding properties with those of native human Hb F and Hb A.8,9 Purified Hb F γG1V, γS143H showed similar migration on cellulose acetate electrophoresis to normal human Hb F made in yeast or isolated from human cord blood (Fig 1). In contrast, cellulose acetate electrophoresis showed, as expected, that surface charge of the triple variant Hb F γG1V, γE5P, γS143H at pH 8.6 was more positive compared with Hb F and Hb F γG1V, γS143H (Fig 1). Small amounts of abnormal forms of recombinant hemoglobins are produced in yeast, including sulphaem-containing hemoglobin and/or misfolded hemoglobins.13 14 These will affect oxygen-binding properties of recombinant hemoglobins and, therefore, were removed by chromatographic purification. Lack of sulphaem-containing contaminants was confirmed by measuring absorption spectra (Fig 2A). Mass spectral analysis using electrospray ionization mass spectrometry (ES-MS) showed expected Gγ-globin chain molecular masses of 15,993 Daltons for the Gγ chain purified from yeast, 16,088 Daltons for the Gγ1Val, 143His- and 16,056 Daltons for the Gγ1Val, 5Pro, 143Hisglobin chains in addition to 15,126 Daltons for the α-globin chain. Absorption spectra of the CO forms of the two Hb F variants were the same as those of native and recombinant Hb F (Fig 2A). In addition, circular dichroism spectra in the region from 190 to 290 nm for recombinant Hb F and the two Hb F variants were similar to that of native Hb F (Fig 2B), indicating that these substitutions do not significantly affect globin folding and/or the overall secondary structure of Hb tetramers.
Cellulose acetate electrophoresis of Hb F variants. Mobilities after cellulose acetate electrophoresis of Hb F purified from cord blood (lane 3) and yeast (lane 4) and the two Hb F variants (Hb F γG1V, γS143H [lane 5] Hb F γG1V, γE5P, γS143H [lane 6]) were compared with native Hb A (lane 2) and Hb S (lane 1).
Cellulose acetate electrophoresis of Hb F variants. Mobilities after cellulose acetate electrophoresis of Hb F purified from cord blood (lane 3) and yeast (lane 4) and the two Hb F variants (Hb F γG1V, γS143H [lane 5] Hb F γG1V, γE5P, γS143H [lane 6]) were compared with native Hb A (lane 2) and Hb S (lane 1).
Absorption and circular dichroism spectra of Hb F and the two Hb F variants. Absorption (A) and CD (B) spectra were recorded for the CO forms of human and recombinant Hb F and the two Hb F variants in 0.1 mol/L phosphate buffer, pH 7.0, at 5°C. Results were corrected for small differences in protein concentration. a, b, c, and d represent Hb F purified from cord blood, yeast, and the two purified Hb F variants (Hb F γG1V, γS143H and Hb F γG1V, γE5P, γS143H), respectively.
Absorption and circular dichroism spectra of Hb F and the two Hb F variants. Absorption (A) and CD (B) spectra were recorded for the CO forms of human and recombinant Hb F and the two Hb F variants in 0.1 mol/L phosphate buffer, pH 7.0, at 5°C. Results were corrected for small differences in protein concentration. a, b, c, and d represent Hb F purified from cord blood, yeast, and the two purified Hb F variants (Hb F γG1V, γS143H and Hb F γG1V, γE5P, γS143H), respectively.
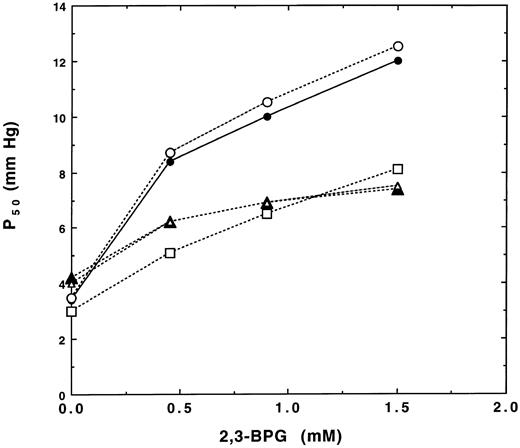
Oxygen affinities of Hb F and the Hb F variants were measured in the absence and presence of 2, 3-BPG in 50 mmol/L Bis-Tris buffer, pH 7.4, containing 0.1 mol/L NaCl at 20°C, and P50 values (the partial oxygen pressure required to give 50% saturation of hemoglobin) are shown in Fig 3. Oxygen affinities of Hb F made in yeast or purified from human cord blood were identical in the absence of 2, 3-BPG and were slightly lower than that of normal human Hb A, as indicated by their slightly higher P50 values (4.1 v 3.4 for Hb A). P50 values of Hb A and Hb F increased with increasing 2, 3-BPG; however, effects of 2, 3-BPG on Hb F were much less than that of Hb A. The P50 value of Hb F γG1V, γS143H in the absence of 2, 3-BPG was slightly lower than that of normal Hb F (3.0 v 4.0 for Hb F ). The increases in P50 for Hb F γG1V, γS143H with increasing 2, 3-BPG concentrations were larger at greater than 0.5 mmol/L 2, 3-BPG than that of normal Hb F (Fig 3). Effects of 2, 3-BPG on increases in P50 for this variant were still significantly less than that of Hb A. These results clearly indicate that amino acids other than the N-terminal and 143 positions in the γ- and β-globin chains are responsible for differences in 2, 3-BPG effects on oxygen affinity of Hb A and Hb F. Therefore, we engineered an additional substitution of Pro for Glu at the γ5 (A2) position and expressed Hb F γG1V, γE5P, γS143H in yeast to examine the role of γ5 Glu in decreased 2, 3-BPG binding to Hb F.
Effects of 2, 3-BPG on oxygen affinity of the Hb F variants. Oxygen affinities of recombinant Hb F (▵), Hb F γG1V, γS143H (□), and Hb F γG1V, γE5P, γS143H (○) were measured in 50 mmol/L Bis-Tris, pH 7.2, containing 5 mmol/L EDTA and 0.1 mol/L NaCl at 20°C as a function of various 2, 3-BPG concentrations, and results were compared with those of Hb A (•) and Hb F (▴) isolated from human adult and cord red blood cells, respectively.
Effects of 2, 3-BPG on oxygen affinity of the Hb F variants. Oxygen affinities of recombinant Hb F (▵), Hb F γG1V, γS143H (□), and Hb F γG1V, γE5P, γS143H (○) were measured in 50 mmol/L Bis-Tris, pH 7.2, containing 5 mmol/L EDTA and 0.1 mol/L NaCl at 20°C as a function of various 2, 3-BPG concentrations, and results were compared with those of Hb A (•) and Hb F (▴) isolated from human adult and cord red blood cells, respectively.
P50 values of the triple variant Hb F γG1V, γE5P, γS143H in the absence and presence of 2, 3-BPG were much higher than those of Hb F γG1V, γS143H and were similar to those of Hb A (Fig 3). These results clearly show that the difference between Pro and Glu at the A2 position in the A helix in Hb A and Hb F, respectively, is critical for reduced binding of 2, 3-BPG to Hb F, even though β5 Pro does not interact directly with 2, 3-BPG in Hb A. Negatively charged Glu (A2) at this position may affect the size, charge, and/or shape of the cavity as well as the position of γ2 His, in comparison with Pro (A2) of Hb A. These differences might then result in decreased 2, 3-BPG binding to Hb F, which results in higher oxygen affinity for Hb F in the presence of 2, 3-BPG compared with Hb A.
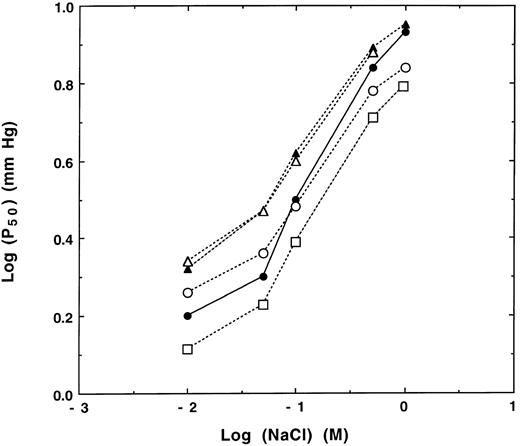
In addition to 2, 3-BPG, inorganic ions, protons, CO2 , and organic phosphates have marked effects on oxygenation of hemoglobin.1,4,15,16 For example, oxygen affinity is lowered in the presence of increasing chloride ion concentrations (Fig 4).1,4 Binding sites for chloride in deoxyhemoglobin have been identified by x-ray analysis and include the N-terminal amino group of the β chain as well as specific sites in the α chain.17 Previous studies showed that chloride effects on oxygen affinities of purified Hb F and Hb A are slightly different (see Bunn and Forget1 and Bunn and Briel4 and see Fig 4). At low chloride ion concentrations, Hb F has a significantly lower oxygen affinity (higher log P50 ) than Hb A, as shown in Fig 4. It is likely that, at low chloride concentrations, the deoxy or T quaternary structure of Hb F is more stable than that of Hb A, because Hb F may have fewer repelling positive charges in its central cavity.18,19 In contrast, at near physiologic concentrations of chloride, Hb F and Hb A have similar oxygen affinities. Effects of chloride ion on oxygen affinity of Hb F were less than that of Hb A as previously reported (see Bunn and Forget1 and Bunn and Briel4 and see Fig 4). Chloride ion binding to Hb A occurs at the 2, 3-BPG binding site, and therefore may have less effect on oxygenation of Hb F than on that of Hb A.16,18 The alkaline Bohr effect of Hb F (Δlog P50 / ΔpH between pH 6.5 and 8.5), like that of Hb A, is markedly dependent on chloride ion concentration. At low chloride concentrations (eg, 0.005 mol/L), both Hb A and Hb F have identical Bohr effects; whereas at physiologic chloride ion concentrations, the Bohr effect of Hb F is about 20% higher than that of Hb A.1 18 It is likely that the smaller Bohr effect of Hb A is due to the presence of β143 His instead of γ143 Ser.
Effects of chloride ion on oxygen affinity of the Hb F variants. Oxygen affinities of recombinant Hb F (▵), Hb F γG1V, γS143H (□), and Hb F γG1V, γE5P, γS143H (○) were measured in 50 mmol/L Bis-Tris, pH 7.2, containing 5 mmol/L EDTA at 20°C as a function of different chloride ion concentrations and were compared with those of Hb A (•) and Hb F (▴) isolated from human adult and cord red blood cells, respectively.
Effects of chloride ion on oxygen affinity of the Hb F variants. Oxygen affinities of recombinant Hb F (▵), Hb F γG1V, γS143H (□), and Hb F γG1V, γE5P, γS143H (○) were measured in 50 mmol/L Bis-Tris, pH 7.2, containing 5 mmol/L EDTA at 20°C as a function of different chloride ion concentrations and were compared with those of Hb A (•) and Hb F (▴) isolated from human adult and cord red blood cells, respectively.
Oxygen affinities of Hb F γG1V, γS143H in the absence and presence of chloride ion were higher than those of Hb F (Fig 4). Effects of chloride on oxygen affinity of Hb F γG1V, γS143H were similar to those of Hb F. Substitution of Pro for Glu at the γ5 (A2) position in Hb F γG1V, γS143H increased P50 values in the presence or absence of chloride, but P50 values were lower than those of normal Hb F. Effects of chloride on oxygen affinity of Hb F γG1V, γE5P, γS143H were also similar to those of Hb F γG1V, γS143H and normal Hb F. These results indicate that the N-terminal Gly and/or γ143 Ser instead of Val and His, respectively, in Hb F increases oxygen affinity, whereas the Glu to Pro change at the A2 position in Hb F decreases oxygen affinity and increases 2, 3-BPG binding. There may be no differences between the two Hb F variants when comparing effects of chloride ion on oxygen affinity because of the smaller size and, therefore, ease of entry of chloride into the cavity in contrast to the larger size of 2, 3-BPG.
Proline plays an important role in the native structure of proteins and usually does not participate in direct formation of α helices. Introduction of proline in the middle of a helical segment usually leads to subunit destabilization, as evidenced by several naturally occurring Hb variants such as Hb Saki (β14 Leu → Pro), Hb Crete (β129 Ala → Pro), Hb Bicetre (β63 His → Pro), and Hb Atlanta (β75 Leu → Pro).20-22 In contrast, x-ray analysis indicated that Pro at the second position of the A helix in the β chain is not conserved, suggesting that Pro at this position is not critical for structure. In fact, Hb Warwickshire (β5 Pro → Arg) showed no difference in oxygen-binding properties in comparison with Hb A at physiologic chloride ion concentrations.20 In addition, this substitution caused mild instability and a slight decrease rather than increase in oxygen affinity in the presence of 2, 3-BPG in comparison with Hb A (11.0 v 9.96 mm Hg of P50 for Hb A). These results indicate that there is an increase in 2, 3-BPG binding to hemoglobin as a result of the β5 Pro → Arg change.23 However, the recombinant Hb A variant (β5 Pro → Ala) showed increased oxygen affinity (0.68 for the P50 ratio of Hb A βP5A/Hb A) without any affects on heat stability.24 Effects of 2, 3-BPG on oxygen affinity of this variant were less than those of Hb A in the presence of 0.1 mol/L NaCl. In addition, it is interesting to note that the decreased senstivity to 2, 3-BPG of horse Hb compared with human Hb A was suggested to be due to perturbations induced by amino acid differences in the A-helix at positions β-A1 (β4 Thr v Ser) and β-A2 (β5 Pro v Gly) in addition to the amino acid replacement at position β-NA2 (β2 His v Gln).25
X-ray diffraction analysis of crystals of Hb F showed that the only detectable difference in tertiary structure of the γ and β chains in Hb F and Hb A, respectively, is in the N-terminal regions.6 7 Differences in primary structure of γ and β chains near the N-terminal region in the A helix (eg, Gly [NA1], Phe [NA3] Glu [A2], Asp [A4] Ala [A6], and Ser [A10] in γ v Val, Leu, Pro, Glu, Ser, and Ala in β) may decrease interaction of Hb F with 2, 3-BPG. Our results also show that Glu instead of Pro at the A2 position in the γ chain slightly increased oxygen affinity. These findings suggest that amino acids at the A2 position may influence the first turn of the A helix, which may affect interactions between the A and EF helices in the absence of 2, 3-BPG. Differences in flexibility of the first turn of the A helix in Hb A versus Hb F may be affected by Pro in Hb A and the negative charge of Glu in Hb F at the A2 position, and these differences could also modify γ2 His binding to 2, 3-BPG.
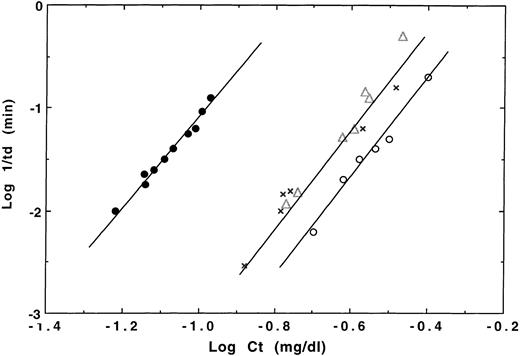
It is well known that Hb F and Hb A2 inhibit polymerization of Hb S in vitro and that high levels of Hb F in red blood cells of patients with sickle cell disease have been frequently associated with reduced disease severity.1,26 Therefore, there is considerable interest in finding ways to increase synthesis of Hb F in adults for the purpose of ameliorating sickle cell disease.26 The net effect of Hb F or Hb A2 on Hb S polymerization is to dilute Hb S by exclusion of FS or A2S hybrids (α2βsγ or α2βsδ) from deoxy Hb S polymerization that results in inhibition of polymerization.1,8,27,28 We compared polymerization of Hb S/Hb F γG1V, γE5P, γS143H versus Hb S/Hb F and Hb S/Hb A2 mixtures to assess inhibitory effects of Hb F γG1V, γE5P, γS143H on Hb S polymerization. Polymerization of 1:1 mixtures of Hb S/Hb F γG1V, γE5P, γS143H was much delayed compared with Hb S alone, just like Hb S/Hb F and Hb S/Hb A2 mixtures, and also showed a delay time before polymerization. Polymerization rates for Hb S/Hb F γG1V, γE5P, γS143H mixtures were similar to those of Hb S/Hb A2 mixtures and slightly faster than those for Hb S/Hb F mixtures at the same Hb concentrations.28 Logarithmic plots of delay time versus concentration for Hb S/Hb F γG1V, γE5P, γS143H showed a straight line like Hb S/Hb A2 mixtures; this line was shifted right 0.44 U from the line for Hb S and slightly left from the line for Hb S/Hb F mixtures (Fig 5). In addition, the critical concentration required for polymerization of Hb S/Hb F γG1V, γE5P, γS143H mixtures was the same as that of Hb S/Hb A2 mixtures. These results indicate that Hb F γG1V, γE5P, γS143H inhibits Hb S polymerization like Hb A2 and that Hb F γG1V, γE5P, γS143H/S hybrids are excluded like Hb A2/S hybrids from the initiation of polymerization with Hb S.28
Relationship between log of reciprocal delay time and Hb concentration for polymerization of Hb F γG1V, γE5P, γS143H/Hb S mixtures. Kinetics of polymerization of the deoxy-forms of 1:1 mixtures of Hb F γG1V, γE5P, γS143H/Hb S compared with Hb F/Hb S or Hb A2 /Hb S mixtures were performed in 1.8 mol/L phosphate buffer, pH 7.4, at 30°C using the temperature-jump method. ▵, ○, and X represent Hb F γG1V, γE5P, γS143H/S, F/S, and A2 /S mixtures, respectively, whereas • represents HbS.
Relationship between log of reciprocal delay time and Hb concentration for polymerization of Hb F γG1V, γE5P, γS143H/Hb S mixtures. Kinetics of polymerization of the deoxy-forms of 1:1 mixtures of Hb F γG1V, γE5P, γS143H/Hb S compared with Hb F/Hb S or Hb A2 /Hb S mixtures were performed in 1.8 mol/L phosphate buffer, pH 7.4, at 30°C using the temperature-jump method. ▵, ○, and X represent Hb F γG1V, γE5P, γS143H/S, F/S, and A2 /S mixtures, respectively, whereas • represents HbS.
One approach to treatment of sickle cell disease is gene therapy that might involve viral transfer of the human γ-globin or variant gene into self-renewing hematopoietic stem cells in the marrow, hopefully resulting in long-term expression at high levels in adult blood cells after transplantation of the virally transduced bone mallow cells. Oxygen affinities of mixtures of Hb S and Hb F show higher oxygen affinity than that of Hb S or Hb A alone, which is caused by different affinities for 2, 3-BPG. This results in oxygen being first removed from Hb S in FS mixtures under low oxygen pressure. Therefore, reduction in oxygen affinity of Hb F in red blood cells containing Hb S and Hb F would be beneficial to patients by allowing more Hb S in these mixtures to be oxygenated under low oxygen tension and, therefore, significantly reduce sickling of red blood cells. The presence of Hb F variants with lower oxygen affinity than native Hb F in Hb S-containing red blood cells would then delay polymerization of Hb S. Therefore, studies aimed at production of more efficient Hb F variants for use as antisickling agents for gene therapy are critical. This is also important because there are limitations on expression levels of Hb F with viral vectors in cells; therefore, design of the most efficient Hb variant could be rate-limiting. Construction of Hb F variants like Hb F γG1V, γE5P, γS143H with reduced oxygen affinity should facilitate optimal selection of Hbs for potential use in gene therapy for sickle cell disease and thalassemia in the future. Furthermore, the growing knowledge of hematopoetic stem cell biology will also help efforts to enhance gene transfer efficiency and expression of vectors containing Hb F variants.
ACKNOWLEDGMENT
The authors thank Dr Eric Rappaport and members of the Nucleic Acid/Protein Core at The Children's Hospital of Philadelphia for automated DNA sequence analysis. We are grateful to Dr H.E. Witkowska for mass spectral analysis of the β-chain variants performed at the Children's Hospital Mass Spectrometry Facility (Oakland, CA; Dr C. Shackleton, Director).
Supported in part by grants from the National Institutes of Health (No. P60 HL38632 and DK 16691), the American Heart Association, and the March of Dimes Birth Defects Foundation and by the Nemours Foundation.
Address reprint requests to Kazuhiko Adachi, PhD, Division of Hematology, The Children's Hospital of Philadelphia, 34th St and Civic Center Blvd, Philadelphia, PA 19104.

![Fig. 1. Cellulose acetate electrophoresis of Hb F variants. Mobilities after cellulose acetate electrophoresis of Hb F purified from cord blood (lane 3) and yeast (lane 4) and the two Hb F variants (Hb F γG1V, γS143H [lane 5] Hb F γG1V, γE5P, γS143H [lane 6]) were compared with native Hb A (lane 2) and Hb S (lane 1).](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/90/8/10.1182_blood.v90.8.2916/4/m_bl_0050f1.jpeg?Expires=1767756498&Signature=Vy7MZJZaUuNptyW~e~KEx~B6L1UBlQTU~-F69WsLBM~Cq8a1dfrJvYAjPfIhsXDH0H3MWLAYfsHQCEAe8~jinqmbiP~e0Q2vn8EkFXeK0eq1r4Rhq2UJaYC-Qc9OUqGg1e78xQzRwxw7FDXuz1l53TOMCmHd9G1NLPLpDhwlHIAyo0j3gxao9YUrAujgDSCzheAw2UxAnVdNkH79H91dk9L1xPsIibwsL58JOD3yn0rqrWx7U9GQb8XzqCHSR3tIkMqhhQGprXvGaAIPMbSmbD1hFY-uZfFvIkXvLddZlJes0VYrwnGI91P6hQLekJFRyS0jLfj0ZP2QU~QCVgElRQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal