Abstract
Nonobese diabetic/severe combined immunodeficient (NOD/SCID) mice transplanted with human cord blood or adult marrow cells and injected 6 weeks posttransplant with 2 daily doses of transforming growth factor-β1 (TGF-β1), monocyte chemoattractant protein-1 (MCP-1), or a nonaggregating form of macrophage inflammatory protein-1 (MIP-1) showed unique patterns of inhibition of human progenitor proliferation 1 day later. TGF-β1 was active on long-term culture initiating cells (LTC-IC) and on primitive erythroid and granulopoietic colony-forming cells (HPP-CFC), but had no effect on mature CFC. MCP-1 inhibited the cycling of both types of HPP-CFC but not LTC-IC. MIP-1 did not inhibit either LTC-IC or granulopoietic HPP-CFC but was active on erythroid HPP-CFC and mature granulopoietic CFC. All of these responses were independent of the source of human cells transplanted. LTC-IC of either human cord blood or adult marrow origin continue to proliferate in NOD/SCID mice for many weeks, although the turnover of all types of human CFC in mice transplanted with adult human marrow (but not cord blood) is downregulated after 6 weeks. Interestingly, administration of either MIP-1β, an antagonist of both MIP-1 and MCP-1 or MCP-1(9-76), an antagonist of MCP-1 (and MCP-2 and MCP-3), into mice in which human marrow-derived CFC had become quiescent, caused the rapid reactivation of these progenitors in vivo. These results provide the first definition of stage-specific inhibitors of human hematopoietic progenitor cell cycling in vivo. In addition they show that endogenous chemokines can contribute to late graft failure, which can be reversed by the administration of specific antagonists.
SUSTAINED BLOOD CELL PRODUCTION depends on a small subset of pluripotent hematopoietic stem cells that retain an ability to balance self-renewal and differentiation divisions. Thus, to produce the billions of new blood cells required in humans every day, the process of their differentiation is normally spread out over many cell generations. In this way, each stem cell initially activated has the potential to produce a large clone of mature progeny. Regulation of the number of cells finally obtained can, thus, be highly complex, because opportunities for interaction with exogenous regulators occur at each successive cell generation and these may variably affect a variety of intrinsic pathways that ultimately control cell viability, cell-cycle progression, and differentiation. In addition, it is now known that even activation of the same receptor may elicit different responses depending on the differentiation or ontological status of the cell as well as the presence of concurrent synergistic or antagonistic signals.1-12
At least partial receptor profiles for various types of hematopoietic progenitors have now been described,13-19 and considerable progress in delineating the signaling pathways they may activate has also occurred. Nevertheless, it is still not possible to use this information to predict biologic outcomes. Therefore, the design of in vitro and in vivo models to address such questions remain of continued biological as well as clinical interest. We have previously shown how the long-term marrow culture (LTC) system can be used to identify both positive and negative factors that regulate the cell-cycle progression of primitive human erythroid and granulopoietic-restricted progenitors (capable of generating large colonies in semisolid media).20-22 These high proliferative potential colony-forming cells (HPP-CFC) are forced into a stable but reversible quiescent state when in contact with resting stromal feeder layers established from primary or subcultured human marrow. In contrast, related but more differentiated progenitors of the same lineages, distinguished by their lower proliferative potential (LPP) when assayed under the same conditions, do not respond to the inhibitors that downregulate HPP-CFC in LTC stromal layers. Hence the LPP-CFC also present remain continuously activated. The cycling activity of the HPP-CFC in the LTC system can, however, be manipulated both directly and indirectly. In the latter case, this includes strategies that simply neutralize the activity of specific endogenously produced inhibitors. Such studies have showed the operation within LTC stromal layers of a cooperative inhibitory mechanism. This is mediated by the dual and specific action on HPP-CFC of transforming growth factor-β1 (TGF-β1)23 and the -C-C- chemokine, monocyte chemoattractant protein-1 (MCP-1).22 Macrophage inflammatory protein-1α (MIP-1α), another -C-C-chemokine, can substitute for MCP-1 in this regard, but does not appear to be an active moiety in LTC adherent layers.22,24 The biologic relevance of these in vitro observations is supported by 2 lines of evidence. The first is the fact that normal HPP-CFC and LPP-CFC isolated directly from the marrow microenvironment in vivo show a similar difference in their proliferative activities.20 Second, in chronic myeloid leukemia (CML) there is a defect in responsiveness of the leukemic HPP-CFC to MCP-122 (and MIP-1α),24 but not to TGF-β1,25 which is associated with their deregulated proliferation both in LTC and in vivo.
More recently, we have shown that nonobese diabetic/severe combined immunodeficiency (NOD/SCID) mice engrafted with normal human hematopoietic cells of cord blood or adult marrow origin show distinct patterns of human CFC proliferation control after these progenitors have been regenerated to the maximum levels they attain within the mouse marrow microenvironment. Thus, even when the number of cells transplanted is matched to give a similar pace and magnitude of early human lymphomyeloid repopulation (0 to 6 weeks posttransplant), human marrow-derived grafts subsequently decline in association with a marked decrease in the rate of human CFC proliferation.26 In contrast, initially comparable grafts of human cord blood are sustained for at least 20 weeks, as is the cycling of the human CFC population they produce.27 These 2 models offered an opportunity to examine the potential role in an in vivo setting of both exogenously administered and endogenously produced inhibitors of human CFC activation and to possibly extend this approach to more primitive progenitors. In the present study, we provide the first evidence that the same inhibitors shown to be active in the LTC system are operative in the NOD/SCID model and that the cell-cycle progression of human LTC-initiating cells (LTC-IC) and HPP-CFC is inhibited by different regulators.
MATERIALS AND METHODS
Human cells.
Cord blood cells were obtained from normal infants delivered by cesarean section. Low density cells (<1.077 g/mL) were isolated by centrifugation over Ficoll-Paque (Pharmacia, Piscataway, NJ) and used directly or after cryopreservation at 135°C in 90% fetal calf serum (FCS; StemCell Technologies, Vancouver, British Columbia, Canada) and 10% dimethylsulfoxide (Sigma, St Louis, MO) as described.27 Normal adult human bone marrow was obtained as frozen material isolated from cadaveric donors (Northwest Tissue Centre, Seattle, WA) or was leftover from cells harvested from donors of allogeneic marrow transplants at our own center. Low-density cells were isolated before transplantation into NOD/SCID mice (see below). Unprocessed cells were used directly to initiate feeder layers for LTC experiments.28 All human cells were obtained with informed consent according to institutional guidelines.
Animals.
NOD/LtSz-scid/scid mice were bred and maintained in the animal facility of the British Columbia Cancer Research Center (Vancouver, British Columbia, Canada) from breeding pairs originally obtained from the Jackson Laboratory (Bar Harbor, ME). Animals were kept at all times under microisolation conditions and were provided exclusively with acidified water (pH = 3) and sterilized food ad libitum. Mice to be transplanted were irradiated at 6 to 8 weeks of age with 350 cGy of 137Cs γ-rays just prior to intravenous injection of 107 low-density human cord blood or 2 × 107 human adult marrow cells.
In vivo cytokine experiments.
Human TGF-β1 was obtained from R&D Systems (Minneapolis, MN). The human MIP-1α used in these experiments was a gift from British Biotechnology (Oxford, UK) and was a recombinant variant (BB-10100) specifically engineered to prevent aggregation.29 Human MCP-1, MCP-1(9-76),30 and MIP-1β were synthesized on an automated peptide synthesizer using tertiary Na-butyloxycarbonyl amino acid chemistry, as previously described.31,32 Cytokines diluted in medium, or an equal volume of medium (as a control), were always given as 2 intraperitoneal injections, 1 day apart, at the indicated doses and times posttransplant. One day after the second injection, the animals were sacrificed and cells flushed out of both tibias and femurs of each mouse into Hanks’ solution containing 2% FCS using a syringe and 22-gauge needle. Cells were then suspended, washed, counted, and diluted as required for phenotyping and progenitor measurements. Human TGF-β,33 BB10100,29 MCP-1, and MCP-1(9-76)30 are also known to be active on murine cells.
Flow cytometry.
Cells were pretreated with human serum and an anti-mouse IgG receptor antibody (2.4G2)34 at 107 cells/mL before incubation at 4°C for 20 minutes either with anti-CD34 (8G12),35 directly conjugated to cyanine-5 succinimidyl ester (Cy5) in combination with an anti-Thy-1 antibody labeled with phycoerythrin (PE) (both kindly provided by P. Lansdorp, Terry Fox Laboratory) and anti-CD19 labeled with fluorescein isothiocyanate (FITC) (Becton Dickinson, San Jose, CA) or with anti-CD34 (8G12)-PE in combination with anti-CD45-FITC (Hlel, Becton Dickinson) and anti-CD71-FITC (P. Lansdorp). Cells were washed once in 2% Hanks solution and then once again in the same medium containing 2 μg/mL propidium iodide (PI; Sigma Chemicals, St Louis, MO) to allow nonviable cells to be excluded. Cells were sorted on a FACStar Plus (Becton Dickinson) equipped with a 5-W argon and a 30-mW helium neon laser. Specific fluorescence of FITC, PE, and PI, excited at 488 (0.4 W) and 633 nm (30 mW), as well as forward and orthogonal light scatter signals, were used to establish sort windows. Controls consisted of staining additional aliquots of the same cells with irrelevant isotype-matched control antibodies directly labeled with the identical fluorochromes. All antibody-staining procedures were performed on marrow cells from untransplanted NOD/SCID to ensure nonreactivity of the reagents used with murine cells. The number of human cells present in a given suspension was determined from an analysis of 5,000 viable cells at settings excluding more than 99.9% of all negative controls with a minimum threshold of 0.1%. All of these procedures have been described in detail previously.27
Progenitor assays.
Progenitors of erythroid, granulopoietic, and mixed colonies that can achieve different sizes were assayed by plating cells in standard serum-containing methylcellulose cultures (Methocult 4230, StemCell) supplemented with the following purified human growth factors: 3 U/mL erythropoietin (Epo) (StemCell), 50 ng/mL Steel factor (SF, Terry Fox Laboratory), and 20 ng/mL each of interleukin-3 (IL-3), granulocyte-macrophage colony-stimulating factor (GM-CSF) (both from Novartis, Basel, Switzerland), G-CSF (StemCell), and IL-6 (Cangene, Mississauga, ON) and scoring the cultures 2 to 3 weeks later by using criteria we have employed for many years.20 To determine the number of LTC-IC present, other aliquots were seeded and suspended in myeloid LTC medium (Myelocult, StemCell) supplemented just prior to use with 10−6 mol/L hydrocortisone sodium hemisuccinate (Sigma) onto preestablished, irradiated feeder layers of mouse fibroblast cells genetically engineered to produce human SF, G-CSF, and IL-3. These cultures were then incubated for 6 weeks at 37°C with weekly half medium changes at which time the total content of CFC was assessed as described.36 To calculate the number of LTC-IC in the innoculum initially seeded into the primary cultures, the 6-week CFC content was divided by 18 for marrow-derived LTC-IC and 28 for cord blood–derived LTC-IC.36 All assays of human progenitors in suspensions obtained from engrafted NOD/SCID mice were performed on CD34+ cells isolated beforehand using a fluorescence-activated cell sorter (FACS) after staining with anti-human CD34-PE.
3H-thymidine suicide assays.
In vitro cytokine experiments.
In vitro inhibitor studies were performed in LTC initiated by seeding 4 × 106 low-density human cord blood cells or 8 × 106 human marrow cells in 2.5 mL of myeloid LTC medium onto semi-confluent, subcultured irradiated feeder layers of normal marrow adherent cells previously established in 35 mm tissue culture dishes.28 These cultures were then maintained at 33°C for 10 to 12 days before further treatment, as described. At the end of each experiment, the adherent layers were suspended and the cells then exposed to 3H-thymidine for 20 minutes before being plated in standard CFC assays.
Statistical analyses.
Values shown are the mean ± standard error of the mean (SEM). Significant differences between groups were evaluated using the Student’s two-tailed t-test and a P value of .05.
RESULTS
Selective effects of exogenously administered MIP-1α, MCP-1, and TGF-β1 on the cycling status of different human progenitor populations in NOD/SCID mice.
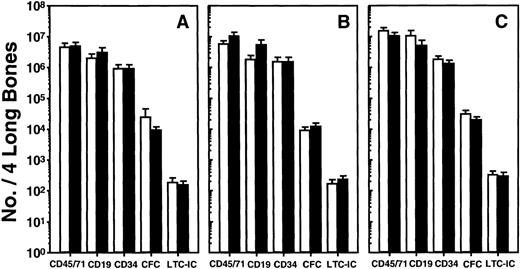
To examine the effects in vivo of cytokines previously shown to block human adult marrow-derived HPP-CFC proliferation in the LTC system, it was necessary to give these at times when the progenitors of interest were cycling. Based on previous data for both CFC and LTC-IC, experiments with adult human marrow-engrafted mice were, therefore, performed 6 weeks posttransplant (after which time, the cycling of the human CFC present decreases)26 and with human cord blood–engrafted mice between 6 and 10 weeks posttransplant (because the proliferative activity of the human progenitors present throughout this time remains high).27 In the first experiments, different groups of mice were given 2 daily injections of 1 μg of TGF-β1 or 2.5 to10 μg of MCP-1 or MIP-1α, or medium, and the next day phenotype, progenitor and cycling measurements were performed on cells from individual mice, and the data for similarly treated mice were then pooled. As illustrated in Fig 1 for the results averaged from all experiments, none of these cytokine treatments had significant immediate effects on any of 5 different parameters of human engraftment monitored (P > .05). This included an assessment of the size of the human CFC and LTC-IC populations detectable in the marrow harvested from both hind legs of the mice. As reported previously,27 the numbers of adult marrow and cord blood cells transplanted gave equivalent levels of engraftment at the times compared here and, since neither type of transplant was affected by any of the treatments within a day, the results shown in Fig 1 for both sources of cells were combined.
Lack of short-term effects of (A) TGF-β1, (B) MCP-1, or (C) MIP-1 on the number of different types of human cells present in NOD/SCID mice. Six to 8 weeks posttransplant animals were given 2 injections of 1 mg of TGF-β1 or 2.5 to 10 mg of MCP-1 or MIP-1, 1 day apart, and sacrificed 1 day after the second injection. Results for the cytokine-treated mice are shown by the solid bars and for the controls by the open bars. Values shown represent the mean ± SEM of the number of phenotypically or functionally defined human cells present in the 2 femurs and tibiae of individual mice. Data from a total of 20 experiments (7 with human bone marrow transplants, 13 with human cord blood transplants, 1 to 7 mice per group per experiment) have been pooled. No significant effects of the injected cytokines were observed in any group (P > .05).
Lack of short-term effects of (A) TGF-β1, (B) MCP-1, or (C) MIP-1 on the number of different types of human cells present in NOD/SCID mice. Six to 8 weeks posttransplant animals were given 2 injections of 1 mg of TGF-β1 or 2.5 to 10 mg of MCP-1 or MIP-1, 1 day apart, and sacrificed 1 day after the second injection. Results for the cytokine-treated mice are shown by the solid bars and for the controls by the open bars. Values shown represent the mean ± SEM of the number of phenotypically or functionally defined human cells present in the 2 femurs and tibiae of individual mice. Data from a total of 20 experiments (7 with human bone marrow transplants, 13 with human cord blood transplants, 1 to 7 mice per group per experiment) have been pooled. No significant effects of the injected cytokines were observed in any group (P > .05).
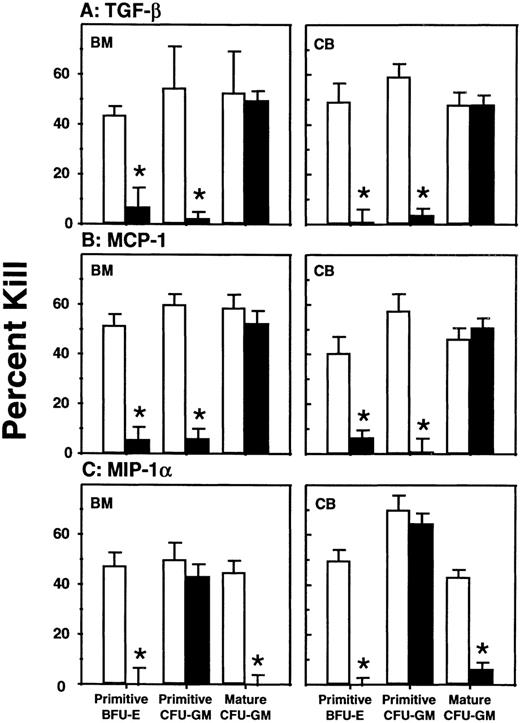
In contrast to the findings for human progenitor numbers, distinct and significant effects on their cycling status were revealed when3H-thymidine suicide assays were performed on the harvested cells just prior to progenitor determinations. As can be seen in Figs 2 and3 for measurements of CFC and LTC-IC proliferation, respectively, the TGF-β1 treatments markedly inhibited (P < .001) the proliferation of all primitive human progenitor populations. This included not only the HPP-CFC assessed by a 20-minute exposure to 3H-thymidine, but also the LTC-IC assessed by a 16-hour exposure in the presence of SF, IL-3, and G-CSF, suggesting that the majority of these cells had been induced to enter a profound, but eventually reversible, quiescent state. On the other hand, the mature human granulopoietic CFC obtained from the same TGF-β1–treated mice and exposed to3H-thymidine in the same 20-minute assays showed no alteration of their proliferative activity (Fig 2).
Comparison of the effect of cytokines on the cycling activity of various classes of human CFC present in the marrow of the same groups of mice depicted in Fig 1. Values shown represent the mean ± SEM of data obtained in the short-term (20 minutes)3H-thymidine suicide assay described in Materials and Methods. Data marked with asterisks were found to be significantly different from the corresponding control values (P < .05).
Comparison of the effect of cytokines on the cycling activity of various classes of human CFC present in the marrow of the same groups of mice depicted in Fig 1. Values shown represent the mean ± SEM of data obtained in the short-term (20 minutes)3H-thymidine suicide assay described in Materials and Methods. Data marked with asterisks were found to be significantly different from the corresponding control values (P < .05).
Comparison of the effect of cytokines on the cycling status of LTC-IC present in the marrow of the same groups of (A) bone marrow and (B) cord blood-transplanted mice shown in Fig 1. Values shown represent the mean ± SEM of data obtained from the overnight3H-thymidine suicide assay described in Materials and Methods. Only the addition of TGF-β1 (P < .001) was found to have a significant effect.
Comparison of the effect of cytokines on the cycling status of LTC-IC present in the marrow of the same groups of (A) bone marrow and (B) cord blood-transplanted mice shown in Fig 1. Values shown represent the mean ± SEM of data obtained from the overnight3H-thymidine suicide assay described in Materials and Methods. Only the addition of TGF-β1 (P < .001) was found to have a significant effect.
The effects of MCP-1 and MIP-1α were consistent for all doses of each tried and the results for the different dose groups have, therefore, been combined. MCP-1 showed the same ability for inhibiting HPP-CFC cycling in vivo as TGF-β1, but had no effect on human LTC-IC proliferation (P > .05). MIP-1α also significantly inhibited the proliferation of the primitive human erythroid progenitors (P < .001) but was unique in its ability to also inhibit mature (P < .001) but not primitive (P > .05) granulopoietic progenitors in vivo. Assessment of the effects of any of these inhibitors on mature human erythroid CFC in vivo was not possible because of their low numbers.
The data shown in Figs 2 and 3 also show that the same response patterns were exhibited by human progenitors derived from either cord blood or adult marrow sources. This result could not have been anticipated, as differences have been reported in both the types and concentrations of factors to which ontologically different populations of analogous human progenitors are responsive,1,6,10including 2 reports of subtle differences in responses to TGF-β and MIP-1α in liquid culture.11,12 However, effects on human cord blood cells in the LTC system have not been reported. The permanent cessation of human CFC proliferation in NOD/SCID mice repopulated with adult marrow cells26 by comparison with the continuous proliferation of their counterparts in recipients of cord blood cells27 also raised the possibility of intrinsic differences in inhibitory responses.
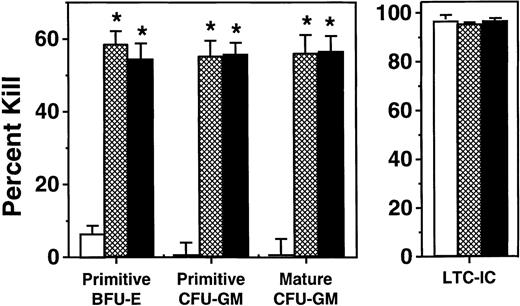
To first examine the effects of MIP-1α, MCP-1, and TGF-β1 on human cord blood-derived progenitors in the LTC system, a series of in vitro experiments similar to those already published for adult marrow progenitors were undertaken. In these, light density cord blood cells were seeded onto irradiated feeders subcultured from LTC adherent layers previously established from human marrow. Ten to 12 days later, half of the medium (and half of the nonadherent cells) was removed and replaced with fresh LTC medium, with or without added TGF-β1, MCP-1, or MIP-1α at concentrations previously shown to block the activation of marrow-derived HPP-CFC located in the adherent layer. As summarized in Table 1, cord blood–derived HPP-CFC (but not LPP-CFC; data not shown), which localize within marrow-derived stromal adherent layers in vitro, showed the same tendency as their marrow-derived counterparts to be maintained in a quiescent state in unperturbed LTC, which could then be rapidly (within 3 days) reversed by addition to the cultures of fresh medium. Similarly, TGF-β1, MCP-1, and MIP-1α were all able to block the activation of cord blood–derived HPP-CFC in LTC stimulated by a medium change.
Effect of Added Inhibitors on the Proliferation of Cord Blood–Derived Primitive Human CFC in Standard LTC
| Fresh Medium . | Inhibitor . | % Kill . | |
|---|---|---|---|
| Primitive BFU-E . | Primitive CFU-GM . | ||
| − | — | 1 ± 4 | 3 ± 2 |
| + | — | 43 ± 5 | 50 ± 4 |
| + | TGF-β1 | 0 ± 5 | 1 ± 3 |
| + | MCP-1 | 4 ± 5 | 2 ± 2 |
| + | MIP-1α | 2 ± 7 | 3 ± 3 |
| Fresh Medium . | Inhibitor . | % Kill . | |
|---|---|---|---|
| Primitive BFU-E . | Primitive CFU-GM . | ||
| − | — | 1 ± 4 | 3 ± 2 |
| + | — | 43 ± 5 | 50 ± 4 |
| + | TGF-β1 | 0 ± 5 | 1 ± 3 |
| + | MCP-1 | 4 ± 5 | 2 ± 2 |
| + | MIP-1α | 2 ± 7 | 3 ± 3 |
TGF-β1 at 5 ng/mL, MIP-1α and MCP-1 at 100 ng/mL. The effect of TGF-β1, MCP-1, or MIP-1α addition was significant (P < .01) when compared with no addition.
Abbreviations: BFU-E, burst-forming unit-erythroid; CFU-GM, colony-forming unit-granulocyte/macrophage.
Role of endogenous chemokines in regulating human progenitor proliferation in NOD/SCID mice engrafted with human marrow or cord blood cells.
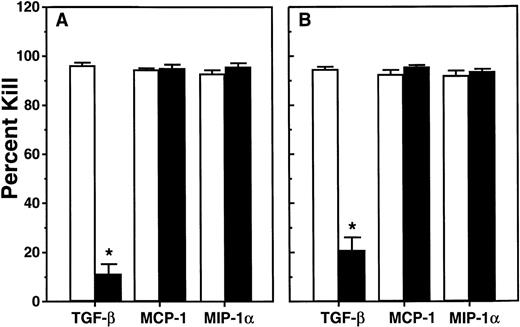
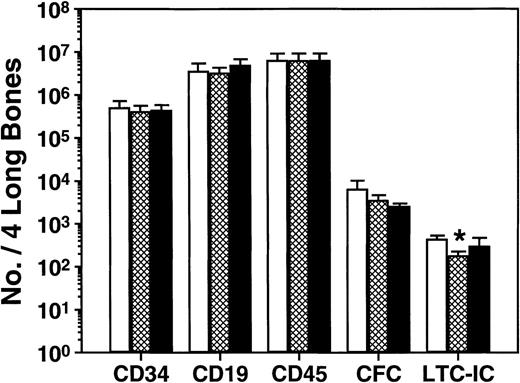
The fact that human CFC proliferation is shut down after 6 weeks in mice that are transplanted with adult human marrow cells26indicates the presence of local acting inhibitors. To determine whether MCP-1 might be one of these, we undertook a second series of in vivo experiments in which we asked whether 2 daily injections of 10 μg of either MIP-1β or a MCP-1 antagonist (a variant of MCP-1 lacking the first 8 amino acids of MCP-1, ie, MCP-1 [9-76]30) would alter the quiescent status of the human CFC present 10 to 12 weeks posttransplant. As shown in Fig 4, there was no effect (P > .05) 1 day later of either of these agents on any of the same 5 parameters of human cell engraftment studied in the previous in vivo experiments except for a marginal decrease in LTC-IC (P = .02) in mice given MCP-1 (9-76). Assessment of the cycling status of all 3 types of human CFC that could be evaluated confirmed their expected quiescent status in the control mice (Fig 5). However, injection of either MIP-1β or MCP-1(9-76) allowed the reactivation of these cells such that 1 day later all had become highly sensitive to a 20-minute exposure in vitro to 3H-thymidine. In 3 of these experiments, there were also sufficient human LTC-IC present to allow their cycling status to be assessed. Unexpectedly, these were found to be rapidly proliferating in the control groups, even though the human CFC harvested from the same mice were quiescent (Fig 5). This continued proliferation of human LTC-IC was also seen in the mice injected with MIP-1β or MCP-1(9-76).
Lack of short-term effects of chemokine antagonists on the number of different types of human cells present in NOD/SCID mice. Mice were transplanted with human bone marrow and 10 to 12 weeks later were given 2 injections of 10 μg of either agent (or medium), 1 day apart, and then sacrificed 1 day later. Results for MCP-1(9-76) are shown as the hatched bars, for MIP-1β as the solid bars, and for the controls as the open bars. Values shown represent the mean ± SEM of the total number of human cells, of the types shown, present in the 2 femurs and tibiae of a total of 24 mice from 4 experiments. Neither of the chemokine antagonists had a significant effect, although a slight effect of MCP-1(9-76) on LTC-IC numbers was noted (P = .02).
Lack of short-term effects of chemokine antagonists on the number of different types of human cells present in NOD/SCID mice. Mice were transplanted with human bone marrow and 10 to 12 weeks later were given 2 injections of 10 μg of either agent (or medium), 1 day apart, and then sacrificed 1 day later. Results for MCP-1(9-76) are shown as the hatched bars, for MIP-1β as the solid bars, and for the controls as the open bars. Values shown represent the mean ± SEM of the total number of human cells, of the types shown, present in the 2 femurs and tibiae of a total of 24 mice from 4 experiments. Neither of the chemokine antagonists had a significant effect, although a slight effect of MCP-1(9-76) on LTC-IC numbers was noted (P = .02).
Cycling activity of various types of primitive and mature human progenitors present in the marrow of the same mice described in Fig 4. Values shown represent the mean ± SEM of data obtained from both short-term (CFC) and overnight (LTC-IC) 3H-thymidine suicide assays. Data marked with an asterisk indicate differences from corresponding control values (P < .001).
Cycling activity of various types of primitive and mature human progenitors present in the marrow of the same mice described in Fig 4. Values shown represent the mean ± SEM of data obtained from both short-term (CFC) and overnight (LTC-IC) 3H-thymidine suicide assays. Data marked with an asterisk indicate differences from corresponding control values (P < .001).
These findings are consistent with a target cell specificity of MCP-1 that does not extend to more primitive human hematopoietic cells than those detectable as HPP-CFC (Figs 2 and 3). In addition they suggest that MCP-1 is likely to be a major cause of the decline in myelopoiesis seen after 6 weeks in NOD/SCID mice transplanted with adult human marrow. Accordingly, it would be expected that MCP-1(9-76) should also be an effective antagonist of exogenously added or endogenously produced MCP-1 in the LTC system. To test this, LTC of human marrow were initiated on preestablished marrow feeders and 10 to 12 days later fresh medium was added alone, with 100 ng/mL MCP-1 or 300 ng/mL MCP-1(9-76) or both. Assessment 2 to 3 days later of the cycling status of the HPP-CFC present in the adherent layers of these cultures showed that a 3-fold excess of the MCP-1 antagonist was sufficient to block the inhibitory action of simultaneously added MCP-1 (Table 2). Moreover, by delaying the addition of the MCP-1(9-76) until 3 days after a medium change and then assessing the cycling status of the HPP-CFC in the adherent layer another 4 days later, an ability of this antagonist to block the inhibitory activity of endogenously produced MCP-1 could be shown (Table 3).
The Ability of MCP-1 to Inhibit the Proliferation of Normal Hematopoietic Cells in Activated LTC Is Blocked by the Addition of MCP-1(9-76)
| Treatment . | % Kill After3H-thymidine . | |
|---|---|---|
| Primitive BFU-E . | Primitive CFU-GM . | |
| MCP-1 | 12 ± 4 | 0 ± 5 |
| None | 52 ± 4 | 50 ± 4 |
| MCP-1(9-76) | 37 ± 2 | 36 ± 13 |
| MCP-1 + MCP-1(9-76) | 52 ± 8 | 51 ± 9 |
| Treatment . | % Kill After3H-thymidine . | |
|---|---|---|
| Primitive BFU-E . | Primitive CFU-GM . | |
| MCP-1 | 12 ± 4 | 0 ± 5 |
| None | 52 ± 4 | 50 ± 4 |
| MCP-1(9-76) | 37 ± 2 | 36 ± 13 |
| MCP-1 + MCP-1(9-76) | 52 ± 8 | 51 ± 9 |
MCP-1 at 100 ng/mL and MCP-1(9-76) at 300 ng/mL. Results shown are the mean ± SEM of data obtained in 5 independent experiments. MCP-1 had a highly significant effect compared with no addition (P < .001), and no effect was seen with the addition of MCP-1(9-76) alone. A significant difference between MCP-1 and MCP-1 + MCP-1 (9-76) was also observed (P < .01).
Addition of MCP-1(9-76) to Previously Activated LTC of Normal Hematopoietic Cells Prevents Their Return to a Quiescent State 4 to 5 Days Later
| Addition . | % Kill After3H-thymidine . | |
|---|---|---|
| Primitive BFU-E . | Primitive CFU-GM . | |
| None | 4 ± 5 | 0 ± 3 |
| MCP-1(9-76) | 38 ± 5 | 35 ± 3 |
| Addition . | % Kill After3H-thymidine . | |
|---|---|---|
| Primitive BFU-E . | Primitive CFU-GM . | |
| None | 4 ± 5 | 0 ± 3 |
| MCP-1(9-76) | 38 ± 5 | 35 ± 3 |
MCP-1(9-76) at 300 ng/mL. Results shown are the mean ± SEM of data obtained in 6 independent experiments. The effect of MCP-1 (9-76) is highly significant (P < .001).
DISCUSSION
In the last decade, the ability of many cytokines to stimulate or inhibit the cycle progression of primitive hematopoietic cells has been shown in various in vitro systems. In a few cases, these have been supplemented by in vivo experiments showing corresponding effects of cytokine injections or predicted effects of their elimination by gene knock-out strategies. Such strategies have demonstrated the potent antiproliferative effect that exogenously administered TGF-β can have on murine CFU-S and CFC populations.38,39 They have also indicated that certain chemokines can have similar effects on these cells40-44 but are not effective in blocking the proliferation of murine cells with long-term in vivo repopulating activity.45
In humans, there is much less information to evaluate the physiologic relevance of these mechanisms and, hence, their importance to disease processes or potential for therapeutic exploitation. One line of evidence comes from the finding of an association in patients with CML of a loss of responsiveness of their leukemic HPP-CFC to MIP-1α and MCP-1,22,24 an increased proliferation activity of these cells in vivo,46 and a greater amplification of this population by comparison with more primitive leukemic cells detectable as LTC-IC.47 However, recent clinical studies of the effect of administering up to 100 mg of MIP-1α (BB 10010) per kilogram body weight have failed to provide consistent evidence of an antiproliferative effect of this chemokine in humans.48
An alternative approach was first suggested by the discovery that human hematopoietic stem cells (LTC-IC) could proliferate and differentiate in vitro in response to factors produced by murine fibroblasts49,50 and, in vivo, could home to the marrow of fetal sheep51 or genetically immunodeficient mice52,53and there be stimulated by factors within that environment. In a previous series of experiments, we documented the kinetics of engraftment of sublethally irradiated NOD/SCID mice after transplantation of initially comparable doses of human adult marrow or cord blood. Interestingly, at later times (beyond 6 weeks posttransplant), the cord blood grafts were relatively well sustained,27 whereas the adult marrow grafts declined.26 Moreover, this change in the overall size of the graft in the marrow-transplanted mice coincided with a selective decrease of the cycling activity of the human CFC population.
In the present studies, we used this xenograft model to examine the human progenitor target cell specificity of various candidate inhibitors in an in vivo setting and to evaluate their potential involvement in the endogenous regulation of human hematopoiesis within the microenvironment of the NOD/SCID marrow tissue. The results of the first experiments showed that TGF-β1, MCP-1, and MIP-1α could all inhibit human progenitor cycling in vivo at doses and within time frames where no effect on progenitor numbers was yet apparent. Thus, the results shown can be assumed to be representative of the initial response of each progenitor population and not confused by problems of selection (because of toxicity) or derivation from a previously affected different progenitor population. This careful choice of experimental design probably maximized our ability to show the different range of progenitor types affected by each of the inhibitors tested, regardless of their ontological origin. The failure of human LTC-IC proliferation to be inhibited by either MIP-1α or MCP-1 confirms and extends previous evidence of a selective failure of MIP-1α to inhibit the in vivo activation of murine stem cells with long-term reconstituting activity.45 This discrepancy in the effects of TGF-β1 and MCP-1 or MIP-1α on the most primitive types of hematopoietic cells (LTC-IC/in vivo repopulating cells) and their presumed immediate progeny (HPP-CFC)54suggests that chemokines may not be useful as myeloprotective agents in patients receiving repeated intensive doses of chemotherapy. On the other hand, it does provide a possible explanation for the simultaneous presence of cycling LTC-IC and quiescent HPP-CFC in the same mice 6 to 8 weeks after being transplanted with adult human marrow cells. The latter hypothesis is also supported by the effects obtained in mice injected with MIP-1β or MCP-1(9-76). MIP-1β is a -C-C- chemokine that has been shown to block the activity of MIP-1α55and possibly MCP-1 in vitro22 when present at molar excess, presumably because of competitive antagonism at the level of receptor binding. MCP-1(9-76) is a synthetic derivative of human MCP-1, which we have recently described and shown to specifically inhibit the activity of murine MCP-1, MCP-2, and MCP-3, but not other chemokines.30-32 The ability of these antagonists to selectively stimulate the proliferation of quiescent human CFC in the marrow of NOD/SCID mice clearly shows that endogenous chemokine production can contribute (either directly or indirectly) to the late shutdown of these progenitors without affecting their more primitive precursors in recipients of adult human marrow transplants. Accordingly, more prolonged administration of either of these antagonists might be expected to enhance more mature stages of human hematopoiesis in the NOD/SCID model and help restore engraftment levels. Similarly these agents might also have potential applications in patients exhibiting graft failure or other cytopenic conditions resulting from chemokine overproduction.
Finally, the present studies provide further and unique evidence for the general fidelity of the LTC system as an in vitro model of the in vivo marrow microenvironment. However, some unexplained disparities have also emerged from the limited in vivo studies that have been feasible to undertake. These include evidence of an activating effect in vivo of both antagonists tested (MCP-1[9-76] and MIP-1β) on a subset of human CFU-GM that did not appear sensitive to the exogenously administered inhibitors these agents are known to antagonize. Thus, MCP-1(9-76) allowed quiescent mature human CFU-GM to be reactivated even though injected MCP-1 did not inhibit these progenitors when they were already cycling. Similarly, MIP-1β allowed quiescent primitive human CFU-GM to be reactivated even though the proliferation of these cells was not inhibited by injections of MIP-1α. Whether these 2 examples of apparently discrepant results may be explained by different concentration or dose-scheduling effects, the presence of other agents able to modulate or counteract the effects of various chemokines (eg, specific cytokines25), or possible indirect effects is not possible to infer from the types of experiments performed here.
The present findings also raise the possibility that the conditions prevailing in a tolerant, xenogeneic host may be similar to those regulating hematopoiesis in the bone marrow of normal adults. Recently, it has been reported that various types of nonhematopoietic cells, including endothelial cells, may coengraft recipients of blood or marrow transplants.56-60 Therefore, it cannot be assumed that the endogenous regulators of the human hematopoiesis obtainable in NOD/SCID mice are exclusively, or even necessarily, of murine origin because human stromal cells as well as macrophages may be present in physiologically relevant numbers. However, both the in vitro (LTC) and in vivo (NOD/SCID mouse) models described here should be useful in addressing these unresolved, but important, questions.
ACKNOWLEDGMENT
The authors thank their colleagues in the Division of Hematology of the University of British Columbia and the Stem Cell Assay Service of the BC Cancer Agency for assistance in procuring and processing human cells and for providing the irradiated human marrow feeder layers. We also thank Gayle Thornbury and Giovanna Cameron for operating the FACS, Maya Sinclaire for technical assistance in the in vivo experiments, and Tara Palmater for typing the manuscript. The authors also acknowledge Dr P. Lansdorp (Terry Fox Laboratory) and British Biotech, Cangene, Novartis, and StemCell for generous gifts of reagents.
Supported by Novartis Canada, a grant from the National Cancer Institute of Canada (NCIC) with funds from the Terry Fox Run, and the Canadian Protein Engineering Network of the Centres of Excellence (PENCE). C.J.E. is a Terry Fox Cancer Research Scientist of the NCIC.
The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. section 1734 solely to indicate this fact.
REFERENCES
Author notes
Address reprint requests to C.J. Eaves, PhD, Terry Fox Laboratory, 601 W 10th Ave, Vancouver, British Columbia, Canada V5Z 1L3; e-mail:connie@terryfox.ubc.ca.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal