Abstract
We describe a tolerance-based stem cell transplantation protocol that combines sublethal radiation with transient blockade of the CD40-CD154 costimulatory pathway using an anti-CD154 antibody. With this protocol, we established hematopoietic chimerism in BALB/c mice transplanted with fully allogeneic C57BL/6 bone marrow. The percentage of donor-origin mononuclear cells in recipients was more than 99%. In addition, all chimeric mice treated with anti-CD154 antibody remained free of graft-versus-host disease (GVHD) and accepted donor-origin but not third-party skin allografts. It was similarly possible to create allogeneic hematopoietic chimerism in NOD/Lt mice with spontaneous autoimmune diabetes. Pancreatic islet allografts transplanted into chimeric NOD/Lt mice were resistant not only to allorejection but also to recurrence of autoimmunity. We conclude that it is possible to establish robust allogeneic hematopoietic chimerism in sublethally irradiated mice without subsequent GVHD by blocking the CD40-CD154 costimulatory pathway using as few as 2 injections of anti-CD154 antibody. We also conclude that chimerism created in this way generates donor-specific allograft tolerance and reverses the predisposition to recurrent autoimmune diabetes in NOD/Lt mice, enabling them to accept curative islet allografts.
Stem cell transplantation leading to allogeneic hematopoietic chimerism has been proposed as a method for the generation of transplantation tolerance1 and the treatment of autoimmune diseases.2 Implementation of clinical trials has in part been impeded by the toxicity of conditioning procedures and by the development of acute and chronic graft-versus-host disease (GVHD).3-6
Recently, it has been hypothesized that procedures that induce tolerance to hematopoietic allografts may be able to obviate any requirement for lethal myeloablative preparation and to prevent GVHD.1 One class of promising tolerance induction protocols is based on blockade of the CD40-CD154 (CD40-CD40 ligand) and B7-CD28 T cell costimulatory pathways.1,7,8 In vivo blockade of the CD154 pathway using anti-CD154 monoclonal antibody (mAb) has been shown to prevent GVHD9 by blocking CD4 help and aborting CD8 alloresponsiveness.10,11 In addition, ex vivo treatment of allogeneic cells using anti-CD154 mAb before adoptive transfer prevents GVHD despite the fact that the cells retain their ability to respond to antigen.12 Allogeneic stem cell transplantation has been performed with partial success in leukemia patients using ex vivo treatment with CTLA4-Ig to block the B7-CD28 costimulation pathway and prevent GVHD.13,14 It has also been shown that simultaneous blockade of both the CD40-CD154 and B7-CD28 pathways of costimulation permits the generation of allogeneic bone marrow chimerism in sublethally conditioned C57BL/6 mice transplanted with B10.A stem cells.15 In that study, the mixed allogeneic chimeras were free of GVHD.
Tolerance-based stem cell transplantation protocols of this kind are likely to find applicability in the transplantation of other allografts because allogeneic chimerism leads to donor-specific tolerance. This has been recognized since 1945, when Owen discovered that transplantation tolerance was generated by the natural intrauterine hematopoietic chimerism that occurs in freemartin cattle.16Subsequently, it was demonstrated that neonatal tolerance induced by stem cell transplantation without conditioning also leads to tolerance and permits donor-specific allotransplantation.17,18Consistent with these observations, Wekerle et al have shown that their adult chimeric stem cell–transplanted mice accept donor-specific skin allografts.15
Stem cell transplantation has been studied not only as a method for inducing chimerism and allograft tolerance but also as a treatment for autoimmune diseases.2,4,5,19 Lymphohemopoietic cells with abnormal function have been implicated in this class of disorders, and their replacement by cells derived from a new population of stem cells is a rational therapeutic approach. Case reports of stem cell transplantation for treatment of autoimmunity have been published,2,4,5 19 and clinical trials of stem cell therapy in systemic lupus erythematosus, multiple sclerosis, rheumatoid arthritis, and scleroderma are underway. The reversal of these autoimmunediatheses by stem cell transplantation is likely to be associated with some degree of recovery in affected organ systems.
In other autoimmune disorders, notably type 1 diabetes mellitus, the target tissue appears to have little in vivo capacity for regeneration and is irreversibly destroyed by the autoimmune process. In such instances, even syngeneic transplantation of replacement tissue is unsuccessful due to recurrent autoimmunity.19,20 Cure of diseases in this category will require both cure of the autoimmune process and the generation of an allotolerant state that permits transplantation of a replacement for the damaged organ—pancreatic islets in the case of diabetes. This problem is frequently studied in the nonobese diabetic (NOD) mouse model of type 1 diabetes.21 In the absence of immunosuppression, NOD mice reject both allogeneic22,23a and syngeneic islet grafts.22
We now report that it is possible to generate hematopoietic chimerism in mice transplanted with fully allogeneic bone marrow using the combination of sublethal irradiation and a brief course of anti-CD154 mAb. Chimeric mice conditioned with this protocol show little evidence of GVHD despite the presence of a peripheral lymphoid compartment almost entirely of donor origin, and they accept donor-origin skin allografts. Finally, we show that chimeric NOD/Lt mice with autoimmune diabetes mellitus are resistant not only to allorejection, but also to recurrence of autoimmunity in islet allografts, and are cured of their hyperglycemia.
Materials and methods
Animals
NOD/Lt (H2g7) mice were obtained from The Jackson Laboratory (Bar Harbor, ME). C57BL/6 (H2b), BALB/c (H2d), and CBA/JCR (H2k) mice were obtained from the National Cancer Institute, Frederick, MD. All animals were certified to be free of Sendai virus, pneumonia virus of mice, murine hepatitis virus, minute virus of mice, ectromelia, lactate dehydrogenase–elevating virus, GD7 virus, reo-3 virus, mouse adenovirus, lymphocytic choriomeningitis virus, polyoma, Mycoplasma pulmonis, and Encephalitozoon cuniculi. All animals were housed in microisolator cages and given ad libitum access to autoclaved food and acidified water. They were maintained in accordance with the guidelines of the Institutional Animal Care and Use Committee of the University of Massachusetts Medical School and recommendations in theGuide for the Care and Use of Laboratory Animals.23b
Antibodies and flow cytometry
MR1 hamster anti-mouse CD154 mAb (immunoglobulin G) was obtained from unpurified ascites and diluted in phosphate-buffered saline to a final concentration of 1 mg/mL for injection as described.24-26 The ascites was collected into sterile tubes and cooled immediately to 4°C. Ascites from several animals was pooled and kept frozen at −70°C until used. The specific pools of MR1 mAb used in these studies were not assayed for endotoxin levels. The MR1 hybridoma was produced by the fusion of NS-1 myeloma cells with spleen cells derived from hamsters immunized with mouse T cells.27,28 Fluorescein isothiocyanate–conjugated anti-H2-Kb mAb (antidonor) and phycoerythrin-conjugated anti-H2-Kd mAb (antihost) were obtained from Pharmingen (San Diego, CA). Flow microfluorometry was performed as described.29-31 Briefly, single cell suspensions were labeled with antibody, rinsed, fixed in 1% paraformaldehyde, and analyzed on a FACScan (Becton Dickinson, Sunnyvale, CA). Forward and side scatter properties were used to distinguish lymphocytes, monocytes, and granulocytes. Dead cells and erythrocytes were excluded by electronic gating. At least 10 000 events were analyzed for each sample.
The relative percentages of host- and donor-origin cells in the BALB/c (H2-Kd+) and NOD/Lt (H2-Kd+) recipients of C57BL/6 (H2-Kb+) bone marrow were determined by flow microfluorometry. The percentage of peripheral blood mononuclear cells (PBMCs) in chimeric mice expressing major histocompatibility complex class I was determined by dual labeling with anti-H2-Kb(donor) and anti-H2-Kd (recipient) antibodies. The percentage of class I–positive PBMC mice averaged 91.5% ± 8.7% in chimeric NOD/Lt mice (n=35) and 89.4% ± 15.1% in chimeric BALB/c mice (n=46). Because fewer than 100% of hematopoietic cells expressed major histocompatibility complex class I antigen, the relative percentage of donor-origin cells (H2-Kb+) in chimeric recipients was calculated as follows: [%H2-Kb+ ÷ (%H2-Kb+ + %H2-Kd+)] × 100. In preliminary experiments, known mixtures of BALB/c and C57BL/6 PBMCs were analyzed, and it was determined that the lower limit of sensitivity of the assay for detecting donor (H2-Kb+) or host (H2-Kd+) cells was 0.5%.
Bone marrow transplantation
Marrow donors were female C57BL/6 mice. Donor femurs and tibias were flushed with RPMI medium using a syringe with a 24-gauge needle. Recovered cells were filtered through sterile nylon mesh (70 μm, Becton Dickinson, Franklin Lakes, NJ), counted, and resuspended in RPMI. Recipients were BALB/c or NOD/Lt mice at least 6 weeks of age. In a preliminary experiment, the doses of radiation that produced 100% mortality in untreated control BALB/c and NOD/Lt mice were determined to be 700 rad (n=5) and 1000 rad (n=5). Doses of 600 rad (n=5) and 900 rad (n=5) or less were uniformly nonfatal. All NOD/Lt recipients were spontaneously diabetic (plasma glucose concentration >250 mg/dL) at the time of bone marrow transplantation.
Before transplantation, recipients were randomized to receive anti-CD154 mAb plus radiation or radiation alone. All injections of mAb were given intraperitoneally. Whole-body irradiation was administered using a 137Cs source (Gammacell 40, Atomic Energy of Canada, Ottawa, ON, Canada). All recipients received a single intravenous injection of 25 × 106 donor bone marrow cells in a volume of 0.5 to 1.0 mL via the lateral tail vein within 1 to 5 hours of irradiation. The doses and timing of the injections, the doses of radiation, and the timing of injections of mAb in relation to irradiation varied in different experiments and are described in detail in “Results.” Blood samples were obtained from all bone marrow recipients 4 to 6 weeks after transplantation for determination of the percentages of donor and host cells by flow microfluorometry. Additional blood samples were obtained periodically as described in “Results.” The presence of hematopoietic chimerism was defined as the presence of at least 1% donor-origin PBMCs.
Diagnosis of diabetes and pancreatic islet cell transplantation
NOD/Lt mice were monitored twice weekly for the presence of glycosuria. Diabetes in glycosuric mice was diagnosed on the basis of 2 plasma glucose concentrations at least 250 mg/dL on different days (Glucose Analyzer2, Beckman Instruments, Fullerton, CA). Prior to islet transplantation, diabetic NOD/Lt mice were treated with time-release insulin pellets (1 pellet per animal; Linplant, Linshin, Searborough, ON, Canada) implanted subcutaneously. Pellets were removed on the day of islet transplantation.
Pancreatic islets were harvested from C57BL/6 donor mice by collagenase digestion as described.24 32 Islets (20 per gram of body weight) were transplanted into the renal subcapsular space of diabetic NOD/Lt recipients, which were monitored twice weekly thereafter for the presence of glycosuria. Graft failure was defined as the presence of a plasma glucose concentration of at least 250 mg/dL on 2 successive days. To confirm graft function in the case of recipients that were normoglycemic at the conclusion of an experimental protocol, nephrectomy of the kidney containing the islet graft was performed and the animal allowed to recover. The graft was scored as having been functional if hyperglycemia recurred (at least 250 mg/dL).
Skin transplantation
Full-thickness skin grafts about 1 cm in diameter were obtained from shaved donors, scraped to remove muscle, and grafted without suturing onto prepared sites on the flanks of anesthetized recipients as described.25 Skin grafts were dressed with Vaseline-impregnated gauze and an adhesive bandage for the first week after surgery. Thereafter, skin grafts were assessed 3 times weekly, and rejection was defined as the first day on which the entire graft surface appeared necrotic.25
Histology
Samples of transplanted skin, host skin, small intestine, large intestine, and liver were recovered from selected experimental mice, fixed, stored in 10% buffered formalin solution, and then embedded in paraffin and processed for light microscopy. Kidneys into which islet grafts had been transplanted were processed in the same way. Sections for routine light microscopy were stained with hematoxylin and eosin. Sections of islet graft-bearing kidneys were stained immunohistochemically for the presence of insulin and glucagon as described.33 Portions of islet graft-bearing kidneys were also snap-frozen and processed for immunohistochemistry as described.34 Sections were reacted with antibodies directed against CD3, CD4, CD8, and CD19. Histologic analyses were performed by a qualified pathologist (B.A.W.) who was unaware of the treatment status of specimen donors.
Statistical analysis
Parametric data are presented as the arithmetic mean ± 1 SD. Average duration of graft survival is presented as the median survival time (MST). Graft survival among groups was compared using the method of Kaplan and Meier35; the equality of allograft survival distributions for animals in different treatment groups was tested using the log-rank statistic.36Pvalues <.05 were considered statistically significant.
Results
Blockade of CD154 permits establishment of C57BL/6 hematopoietic chimerism in BALB/c mice without lethal conditioning
We first established that it is possible to generate hematopoietic chimerism in BALB/c (H2d) mice transplanted with fully allogeneic C57BL/6 (H2b) bone marrow using the combination of sublethal irradiation and a brief course of anti-CD154 mAb. As shown in Table 1, chimerism could be established using as little as 400 rad of conditioning radiation and as few as 2 doses of anti-CD154 mAb. The percentage of successful transplantation procedures yielding detectable chimerism using 400 rad varied as a function of the dose and frequency of treatment with anti-CD154 mAb. Treatment with only 2 doses of 0.5 mg of mAb was optimal, yielding chimerism in 100% of 9 mice in 2 independent trials. Chimerism could also be established without blockade of CD154 in recipients given 500 rad but not 400 rad of conditioning radiation.
Hematopoietic chimerism in BALB/c mice given varying sublethal doses of radiation and C57BL/6 bone marrow in the presence or absence of anti-CD154 monoclonal antibody
| Radiation (rad) . | Anti-CD154 mAb . | Number of Chimeric Mice (%) . | Percentage of Donor-Origin PBMCs in Chimeric Mice . | |
|---|---|---|---|---|
| Number of Injections . | Dose . | |||
| 300 | 0 | — | 0/10 (0%) | — |
| 4 | 0.25 mg | 0/5 (0%) | — | |
| 14 | 0.25 mg | 0/5 (0%) | — | |
| 2 | 0.5 mg | 0/5 (0%) | — | |
| 400 | 0 | — | 0/10 (0%) | — |
| 4 | 0.25 mg | 2/4 (50%) | Both >99% | |
| 14 | 0.25 mg | 4/9 (44%) | All >99% | |
| 2 | 0.5 mg | 9/9 (100%) | All >99% | |
| 500 | 0 | — | 10/10 (100%) | All >99% |
| 4 | 0.25 mg | 4/4 (100%) | All >99% | |
| 14 | 0.25 mg | 8/9 (89%) | All >99% | |
| 2 | 0.5 mg | 9/9 (100%) | All >99% | |
| Radiation (rad) . | Anti-CD154 mAb . | Number of Chimeric Mice (%) . | Percentage of Donor-Origin PBMCs in Chimeric Mice . | |
|---|---|---|---|---|
| Number of Injections . | Dose . | |||
| 300 | 0 | — | 0/10 (0%) | — |
| 4 | 0.25 mg | 0/5 (0%) | — | |
| 14 | 0.25 mg | 0/5 (0%) | — | |
| 2 | 0.5 mg | 0/5 (0%) | — | |
| 400 | 0 | — | 0/10 (0%) | — |
| 4 | 0.25 mg | 2/4 (50%) | Both >99% | |
| 14 | 0.25 mg | 4/9 (44%) | All >99% | |
| 2 | 0.5 mg | 9/9 (100%) | All >99% | |
| 500 | 0 | — | 10/10 (100%) | All >99% |
| 4 | 0.25 mg | 4/4 (100%) | All >99% | |
| 14 | 0.25 mg | 8/9 (89%) | All >99% | |
| 2 | 0.5 mg | 9/9 (100%) | All >99% | |
BALB/c (H2d) mice were irradiated, transfused intravenously with 25 × 106 C57BL/6 (H2b) bone marrow cells, and injected intraperitoneally with anti-CD154 mAb as indicated. The schedule of anti-CD154 mAb (relative to irradiation and bone marrow transplantation on day 0) was as follows. Recipients given 2 injections were treated on days 0 and +3; recipients of 4 injections on days −3, 0, +3, +7; and recipients of 14 injections on days −3, 0, and twice weekly thereafter. The percentage of H2b donor-origin PBMCs was determined by flow cytometry 4 to 6 weeks after irradiation and bone marrow transplantation. The presence of chimerism was defined as at least 1% donor-origin cells.
The data shown in Table 1 were obtained 4 to 6 weeks after transplantation. All mice documented to be chimeric at this point were subsequently reanalyzed at varying intervals 49 to 338 days after transplantation. Analysis of PBMC demonstrated that, without exception, they remained chimeric to the same degree as indicated in Table 1.
Mice were ultimately killed at varying intervals 27 to 48 weeks after transplantation for histologic studies and flow microfluorometric analyses of spleen, thymus, and bone marrow. In mice that were given 400 or 500 rad of conditioning radiation and 4 or 14 doses of anti-CD154 mAb, more than 99% of splenic B cells, CD4+ T cells, and CD8+ T cells detected were of donor origin (n=9). In contrast, the percentage of donor-origin thymocytes in chimeric recipients was generally much smaller, 57% ± 27% (range, 17% to > 99%; n=9). Bone marrow cells were more than 99% donor-origin.
We also analyzed 2 chimeric mice that had received only 500 rad of conditioning radiation and no anti-CD154 mAb. In these mice, more than 99% of B cells, CD4+ T cells, and CD8+ T cells were of donor origin. The percentages of donor-origin thymocytes in these 2 mice were more than 99% and 60%. Bone marrow cells were more than 99% donor-origin in both.
The diagnosis of GVHD was made clinically or histologically. Throughout the period of observation (up to 338 days), there was no clinical evidence of GVHD in any chimeric bone marrow recipient that had received any dose of radiation plus any of the anti-CD154 mAb regimens (n=36; Table 1). This was true in the case of mice that were not further manipulated (n=22) and mice that were given donor-specific skin allografts (n=14; described below). The absence of GVHD was confirmed by histologic study of skin, liver, and small and large intestine in a subset of the animals that were skin graft recipients (n=9). Two of the 9 mice showed rare apoptotic bodies in intestinal crypts, but cryptitis was absent. Similarly, the livers of 2 of 9 mice showed scattered peripheral lymphocytes, but bile duct inflammation was not evident. Skin biopsies were unremarkable.
Among the 10 chimeric BALB/c recipients that had been given 500 rad of conditioning radiation but no anti-CD154 mAb, 7 survived more than 10 weeks. The other 3 exhibited clinical signs of GVHD before death; these included weight loss, loss of hair, hunched posture, and erythematous encrusted ears. The remaining 7 animals were confirmed still to be chimeric 7 to 21 weeks after bone marrow transplantation and given skin allografts as described in the next section. Among these, 5 subsequently died with clinical evidence of GVHD. Two surviving mice in this group were killed on day 112 after skin grafting for flow cytometric and histologic analyses. Both were documented to have remained chimeric, and neither showed histologic evidence of GVHD.
Donor-specific skin allografts uniformly survive in chimeric mice that receive anti-CD154 antibody
We next measured the survival of donor-origin skin allografts in a subset of the bone marrow–transplanted mice presented in Table 1. Selected mice from among those tested for chimerism 4 to 6 weeks after bone marrow transplantation were given C57BL/6 skin grafts 3 to 15 weeks later.
Donor-specific (C57BL/6) skin allograft survival in chimeric mice that were given any of the anti-CD154 mAb treatment regimens plus 400 rad (n=6) or 500 rad (n=8) was significantly prolonged (MST, >183 days and >112 days, respectively). Graft survival in these 2 groups was statistically similar, and the groups were combined in subsequent analyses (Figure 1, group 1). Median skin allograft survival was statistically significantly longer in mice that had received both radiation and anti-CD154 mAb (MST, 166 days; n=14; Figure 1, group 1) than in chimeric mice that had received only 500 rad of radiation (MST, 9 days; n=7; Figure 1, group 2). As the figure shows, skin graft survival in 3 of the 7 chimeric animals that had received radiation alone was prolonged. One of these 3 mice had clinical signs of GVHD and died with its graft intact. The other 2 were killed for histologic study 112 days after skin grafting when 1 of them became ill. As noted above, both of these mice were chimeric, and neither showed histologic evidence of GVHD. The skin graft from 1 animal showed scattered mononuclear cells around hair follicles and sweat glands and, also, rare apoptotic keratinocytes and intradermal mononuclear cells. The other graft was unremarkable, with no evidence of mononuclear infiltration.
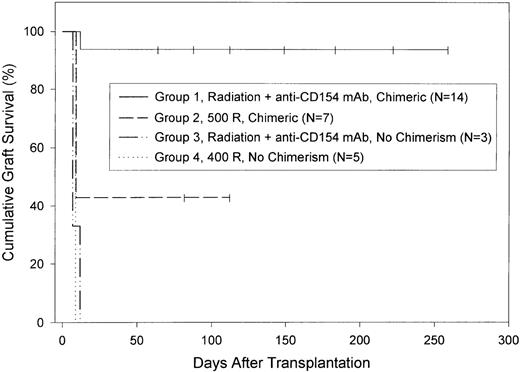
C57BL/6 skin allograft survival in BALB/c recipients of C57BL/6 bone marrow.
Both chimeric and nonchimeric BALB/c mice from among those described in Table 1 were selected at random and transplanted with a C57BL/6 skin graft 7 to 19 weeks after bone marrow transplantation as described in “Materials and Methods.” For purposes of analysis, mice were divided into 4 groups. Mice in group 1 had received 400 or 500 rad of conditioning radiation plus anti-CD154 mAb and were chimeric. MST of grafts in this group was more than 166 days. Mice in group 2 had received 500 rad of conditioning radiation but no anti-CD154 mAb and were chimeric. MST of grafts in this group was 9 days. Mice in group 3 had received 400 or 500 rad of radiation plus anti-CD154 mAb but were not chimeric. Mice in group 4 were given 400 rad of radiation but no anti-CD154 mAb and were not chimeric. MST of grafts in both group 3 and group 4 was 9 days. Graft survival in group 1 was significantly longer than in any other group (P<.0001). Censored data—mice that died or were killed for histologic study or were still alive at the end of the experiment—are indicated by vertical lines.
C57BL/6 skin allograft survival in BALB/c recipients of C57BL/6 bone marrow.
Both chimeric and nonchimeric BALB/c mice from among those described in Table 1 were selected at random and transplanted with a C57BL/6 skin graft 7 to 19 weeks after bone marrow transplantation as described in “Materials and Methods.” For purposes of analysis, mice were divided into 4 groups. Mice in group 1 had received 400 or 500 rad of conditioning radiation plus anti-CD154 mAb and were chimeric. MST of grafts in this group was more than 166 days. Mice in group 2 had received 500 rad of conditioning radiation but no anti-CD154 mAb and were chimeric. MST of grafts in this group was 9 days. Mice in group 3 had received 400 or 500 rad of radiation plus anti-CD154 mAb but were not chimeric. Mice in group 4 were given 400 rad of radiation but no anti-CD154 mAb and were not chimeric. MST of grafts in both group 3 and group 4 was 9 days. Graft survival in group 1 was significantly longer than in any other group (P<.0001). Censored data—mice that died or were killed for histologic study or were still alive at the end of the experiment—are indicated by vertical lines.
Skin allograft survival in irradiated mice that had failed to become chimeric (<1% donor cells) was uniformly brief regardless of whether they had received anti-CD154 mAb (MST, 9 days for both groups; Figure1, groups 3 and 4).
We next determined if skin allograft survival in chimeric BALB/c recipients of C57BL/6 bone marrow was donor-specific. A subset of mice that were documented to be chimeric after treatment with both conditioning irradiation (400 or 500 rad) and anti-CD154 mAb (2 doses) was selected at random 14 to 15 weeks after bone marrow transplantation. They were given either C57BL/6 (H2b, donor-specific) or CBA/JCR (H2k, third-party) skin allografts. As expected, donor-specific (C57BL/6) skin allograft survival in chimeric mice was significantly prolonged (MST, >65 days; n=3). In contrast, CBA/JCR skin allografts survived only briefly (MST, 15 days; n=4).
Histologic analysis of transplanted skin was performed in a subset of 11 mice with intact grafts that had survived 64 to 259 days. These included 9 chimeric recipients that had received radiation plus anti-CD154 mAb and 2 that had received radiation alone. In all instances, there was no evidence of inflammation suggestive of graft rejection.
Blockade of CD154 permits establishment of C57BL/6 hematopoietic chimerism in sublethally conditioned NOD/Lt mice
We next established that it is possible to generate hematopoietic chimerism in NOD/Lt (H2g7) mice transplanted with allogeneic C57BL/6 (H2b) bone marrow using the combination of sublethal irradiation and a brief course of anti-CD154 mAb. In this experiment, all NOD/Lt recipients had developed spontaneous autoimmune diabetes prior to conditioning and bone marrow transplantation. Consistent with previous reports,37 a preliminary experiment demonstrated that NOD/Lt mice are relatively radioresistant; the lethal radiation dose was 1000 rad. We tested for the generation of chimerism in NOD/Lt mice using 600 to 900 rad of conditioning radiation.
As shown in Table 2, hematopoietic chimerism in NOD/Lt mice could be generated using as little as 600 rad of conditioning radiation provided that the mice were also treated with anti-CD154 mAb. As few as 2 doses of anti-CD154 mAb were sufficient. Chimerism could also be established without blockade of CD154, but required a minimum of 700 rad of conditioning radiation.
Hematopoietic chimerism in NOD/Lt mice given varying sublethal doses of radiation and C57BL/6 bone marrow in the presence or absence of anti-CD154 monoclonal antibody
| Radiation (rad) . | Anti-CD154 mAb . | Number of Chimeric Mice (%) . | Percentage of Donor-Origin PBMCs in Chimeric Mice . | |
|---|---|---|---|---|
| Number of Injections . | Dose . | |||
| 600 | 0 | — | 0/6 (0%) | — |
| 14 | 0.25 mg | 3/6 (50%) | 24%, 93%, >99% | |
| 2 | 0.5 mg | 8/12 (67%) | 95%, 97%, Six >99% | |
| 700 | 0 | — | 4/4 (100%) | All >99% |
| 14 | 0.25 mg | 1/3 (33%) | All >99% | |
| 2 | 0.5 mg | 5/6 (83%) | All >99% | |
| 800 | 0 | — | 2/2 (100%) | Both >99% |
| 14 | 0.25 mg | 0/2 (0%) | — | |
| 2 | 0.5 mg | 3/3 (100%) | All >99% | |
| 900 | 14 | 0.25 mg | 6/6 (100%) | All >99% |
| 2 | 0.5 mg | 3/3 (100%) | All >99% | |
| Radiation (rad) . | Anti-CD154 mAb . | Number of Chimeric Mice (%) . | Percentage of Donor-Origin PBMCs in Chimeric Mice . | |
|---|---|---|---|---|
| Number of Injections . | Dose . | |||
| 600 | 0 | — | 0/6 (0%) | — |
| 14 | 0.25 mg | 3/6 (50%) | 24%, 93%, >99% | |
| 2 | 0.5 mg | 8/12 (67%) | 95%, 97%, Six >99% | |
| 700 | 0 | — | 4/4 (100%) | All >99% |
| 14 | 0.25 mg | 1/3 (33%) | All >99% | |
| 2 | 0.5 mg | 5/6 (83%) | All >99% | |
| 800 | 0 | — | 2/2 (100%) | Both >99% |
| 14 | 0.25 mg | 0/2 (0%) | — | |
| 2 | 0.5 mg | 3/3 (100%) | All >99% | |
| 900 | 14 | 0.25 mg | 6/6 (100%) | All >99% |
| 2 | 0.5 mg | 3/3 (100%) | All >99% | |
Spontaneously diabetic NOD/Lt mice (H2g7) were irradiated, transfused intravenously with 25 × 106C57BL/6 (H2b) bone marrow cells, and injected intraperitoneally with anti-CD154 mAb as indicated. The schedule of anti-CD154 mAb (relative to irradiation and bone marrow transplantation on day 0) was as follows. Recipients given 2 injections were treated on days 0 and +3, and recipients of 14 injections on days −3, 0, and twice weekly thereafter. The percentage of H2bdonor-origin PBMCs was determined by flow cytometry 4 to 6 weeks after irradiation and bone marrow transplantation. The presence of chimerism was defined as at least 1% donor-origin cells.
The percentage of transplantation procedures yielding detectable chimerism in mice given both radiation and anti-CD154 mAb varied with the dose and frequency of treatment with the mAb. As was true in the case of BALB/c recipients of C57BL/6 bone marrow, treatment with 2 doses of 0.5 mg of mAb was optimal at each dose of radiation.
The data in Table 2 were obtained 4 to 6 weeks after transplantation. Mice initially observed to be chimeric at this point were subsequently reanalyzed at varying intervals 49 to 331 days after transplantation. With the exception of 1 mouse, they were all documented to have remained chimeric to the same extent as indicated in Table 2. In the 1 exceptional mouse, the percentage of donor-origin cells at 4 weeks was atypically low (24%) and no donor-origin cells could be detected at 7 weeks.
Mice were ultimately killed at varying intervals 16 to 42 weeks after transplantation for histologic studies and flow microfluorometric analyses of spleen, thymus, and bone marrow. Except for the 1 animal described above, more than 99% of splenic B cells, CD4+T cells, and CD8+ T cells detected were of donor origin. In the 1 exceptional case, no donor-origin cells could be detected in blood, spleen, bone marrow, or thymus 20 weeks after transplantation. As was also true of the BALB/c recipients of C57BL/6 bone marrow (Table 1), the average percentage of donor-origin thymocytes in chimeric NOD/Lt recipients (Table 2) was lower, 61% ± 37% (range, 25%-99%; n=5).
The diagnosis of GVHD in these mice was made clinically or histologically. Throughout the period of observation (up to 331 days), there was no clinical evidence of GVHD in any chimeric NOD/Lt bone marrow recipient that had received any dose of radiation plus any of the anti-CD154 mAb regimens (n=29; Table 2). This was true in the case of mice that were not further manipulated (n=10) and mice that were given donor-specific allografts (n=19; described below). The absence of GVHD was confirmed by histologic study of skin, liver, and small and large intestine in a subset of the animals that were allograft recipients (n=11). Five of 11 mice showed scattered periportal lymphocytes. The skin and intestine were unremarkable.
Among the 6 chimeric NOD/Lt recipients that had been given 700 or 800 rad of conditioning radiation but no anti-CD154 mAb, 3 were found dead within 6 weeks of bone marrow transplantation. These mice died before they could receive islet grafts, and the causes of death are not known. The 3 remaining mice were given islet grafts as described in the next section. Two mice survived more than 23 weeks after bone marrow transplantation without clinical evidence of GVHD. The third died with clinical evidence of GVHD shortly after islet grafting.
Donor-specific islet allografts uniformly survive in chimeric NOD/Lt mice and reverse autoimmune diabetes
We next measured the survival of donor-origin islet allografts in a subset of the bone marrow–transplanted NOD/Lt mice presented in Table 2. Selected mice from among those tested for chimerism 4 to 6 weeks after bone marrow transplantation were given C57BL/6 islet allografts 1 week later. These mice had spontaneously developed autoimmune diabetes before bone marrow transplantation. They were treated with slow-release insulin pellets until the day of islet transplantation, at which time the pellets were removed.
Islet allograft survival in chimeric diabetic NOD/Lt mice given any of the anti-CD154 mAb treatment regimens plus any dose of conditioning radiation was significantly prolonged. Through the end of the experiment, none of these 19 islet recipients rejected its graft (MST, >120 days; Figure 2, group 1). One mouse died unexpectedly 12 days after islet transplantation but was normoglycemic 2 days before death. The mouse did not exhibit signs of GVHD, and the cause of death is not known. To confirm graft function, 11 of the 18 long-term islet allograft recipients underwent unilateral nephrectomy of the islet graft-bearing kidney 75 to 276 days after islet transplantation. Hyperglycemia recurred in all nephrectomized mice, confirming the prolonged survival and function of the transplanted islets.
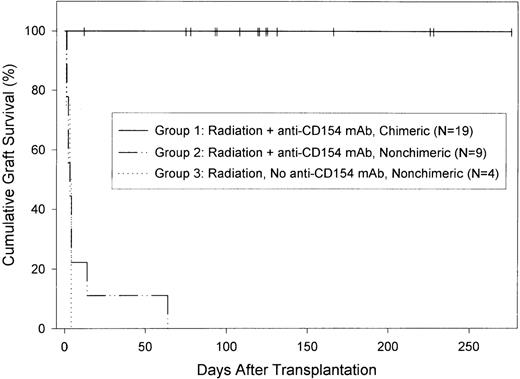
C57BL/6 islet allograft survival in diabetic NOD/Lt recipients of C57BL/6 bone marrow.
Both chimeric and nonchimeric NOD/Lt mice from among those described in Table 2 were selected at random and transplanted with a C57BL/6 islet graft 5 to 7 weeks after bone marrow transplantation as described in “Materials and Methods.” All mice were diabetic at the time of both bone marrow and islet transplantation; their diabetes was treated with sustained-release insulin pellets until islet transplantation, at which time the pellets were removed. For purposes of analysis, mice were divided into 3 groups. Mice in group 1 had received 600 to 900 rad of conditioning radiation plus anti-CD154 mAb and were chimeric. MST of islet grafts in this group was more than 120 days. Mice in group 2 had received 600 to 800 rad of conditioning radiation and anti-CD154 mAb and were nonchimeric. Mice in group 3 had received 600 rad of radiation but no anti-CD154 mAb and were nonchimeric. MST of grafts in groups 2 and 3 was 3 days. Graft survival in group 1 was significantly longer than in the other groups (P<.0001). Graft survival in groups 2 and 3 was statistically similar. Censored data—mice that died or were nephrectomized at the end of the experiment—are indicted by vertical lines.
C57BL/6 islet allograft survival in diabetic NOD/Lt recipients of C57BL/6 bone marrow.
Both chimeric and nonchimeric NOD/Lt mice from among those described in Table 2 were selected at random and transplanted with a C57BL/6 islet graft 5 to 7 weeks after bone marrow transplantation as described in “Materials and Methods.” All mice were diabetic at the time of both bone marrow and islet transplantation; their diabetes was treated with sustained-release insulin pellets until islet transplantation, at which time the pellets were removed. For purposes of analysis, mice were divided into 3 groups. Mice in group 1 had received 600 to 900 rad of conditioning radiation plus anti-CD154 mAb and were chimeric. MST of islet grafts in this group was more than 120 days. Mice in group 2 had received 600 to 800 rad of conditioning radiation and anti-CD154 mAb and were nonchimeric. Mice in group 3 had received 600 rad of radiation but no anti-CD154 mAb and were nonchimeric. MST of grafts in groups 2 and 3 was 3 days. Graft survival in group 1 was significantly longer than in the other groups (P<.0001). Graft survival in groups 2 and 3 was statistically similar. Censored data—mice that died or were nephrectomized at the end of the experiment—are indicted by vertical lines.
As noted at the end of the previous section, only 3 NOD/Lt mice that had become chimeric after irradiation alone survived long enough to receive islet grafts. Two received NOD/SCID (severe combined immunodeficiency) islet grafts; 1 received a C57BL/6 islet graft. The NOD/SCID graft recipients died 40 and 185 days after transplantation. Both were normoglycemic immediately before death, but the mouse that died on day 40 showed evidence of GVHD. The single recipient of C57BL/6 islets remained normoglycemic and clinically well until the conclusion of the experiment 260 days after islet grafting.
Islet allograft survival in irradiated mice that had failed to become chimeric (<l% donor cells) was uniformly brief. This was true both for recipients that had received anti-CD154 mAb (MST, 3 days; n=9; Figure 2, group 2) and for those that had not (MST, 3 days; n=4; Figure2, group 3).
Histologic analysis of transplanted islets was performed on the 11 nephrectomy specimens described above (Figure3). In some cases, light microscopy revealed a very sparse mononuclear cell infiltrate in the renal capsule or adjacent to the islet graft, but no mononuclear cell infiltrates were observed in the grafts themselves. Immunohistochemical analyses using antibodies specific for CD3, CD4, CD8, and CD19 confirmed that few or no lymphoid cells were present in the transplanted tissue. Consistent with the observation that hyperglycemia recurred after nephrectomy, immunohistochemical staining demonstrated that insulin was present in each specimen. Immunoreactive glucagon was also detectable in each specimen.
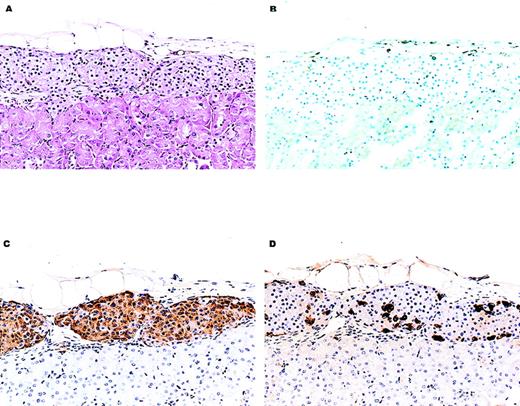
Histology of a representative C57BL/6 islet allograft from a chimeric NOD/Lt recipient.
Shown are histologic sections of C57BL/6 islet tissue that had been transplanted into the renal subcapsular space of a chimeric NOD/Lt mouse. The NOD/Lt recipient had developed spontaneous autoimmune diabetes and had been treated with radiation and anti-CD154 mAb and C57BL/6 bone marrow about 4 weeks before islet transplantation (see “Materials and Methods”). This recipient was normoglycemic for 124 days after transplantation and became hyperglycemic immediately after recovery of the kidney bearing the islet tissue shown here. (A) Hematoxylin and eosin–stained section showing normal appearing islet tissue. An inflammatory infiltrate (× 150) was not evident. (B) A frozen section stained immunohistochemically for the presence of the CD3+ lymphocytes. There is no appreciable CD3+ T-cell infiltrate (× 150). (C, D) Sections stained immunohistochemically for the presence of insulin and glucagon, respectively. Abundant hormone-containing cells are visible (× 150).
Histology of a representative C57BL/6 islet allograft from a chimeric NOD/Lt recipient.
Shown are histologic sections of C57BL/6 islet tissue that had been transplanted into the renal subcapsular space of a chimeric NOD/Lt mouse. The NOD/Lt recipient had developed spontaneous autoimmune diabetes and had been treated with radiation and anti-CD154 mAb and C57BL/6 bone marrow about 4 weeks before islet transplantation (see “Materials and Methods”). This recipient was normoglycemic for 124 days after transplantation and became hyperglycemic immediately after recovery of the kidney bearing the islet tissue shown here. (A) Hematoxylin and eosin–stained section showing normal appearing islet tissue. An inflammatory infiltrate (× 150) was not evident. (B) A frozen section stained immunohistochemically for the presence of the CD3+ lymphocytes. There is no appreciable CD3+ T-cell infiltrate (× 150). (C, D) Sections stained immunohistochemically for the presence of insulin and glucagon, respectively. Abundant hormone-containing cells are visible (× 150).
Skin allograft tolerance is donor-specific in allogeneic NOD/Lt hematopoietic chimeras bearing functional donor-specific islet allografts
We next determined whether the state of transplantation tolerance in chimeric NOD/Lt recipients of C57BL/6 bone marrow was donor-specific, as was the case for the chimeric BALB/c recipients described above. A subset of NOD/Lt mice that had been treated with conditioning radiation plus anti-CD154 mAb and were normoglycemic more than 100 days after transplantation of C57BL/6 islets were selected at random. They were given either a C57BL/6 (H2b, donor-specific) or a CBA/JCR (H2k, third-party) skin allograft. Survival of C57BL/6 skin allografts was significantly prolonged (MST, >128 days; n=3). In contrast, CBA/JCR skin allografts survived only briefly (MST, 15 days; n=3; Table 3). None of the islet allografts failed in either group during the period of observation, and recurrence of hyperglycemia after unilateral nephrectomy was observed in all mice. Histologic analysis of the donor-specific C57BL/6 skin grafts obtained at the conclusion of the experiment showed no evidence of inflammation suggestive of graft rejection.
Skin allograft survival in chimeric NOD/Lt mice bearing donor-specific islet allografts
| Bone Marrow Donor . | Islet Allograft Donor . | Skin Allograft Donor . | Skin Allograft Survival (Days) . |
|---|---|---|---|
| C57BL/6 | C57BL/6 | C57BL/6 | >26, >128, >128 |
| C57BL/6 | C57BL/6 | CBA/JCr | 13, 15, 23 |
| Bone Marrow Donor . | Islet Allograft Donor . | Skin Allograft Donor . | Skin Allograft Survival (Days) . |
|---|---|---|---|
| C57BL/6 | C57BL/6 | C57BL/6 | >26, >128, >128 |
| C57BL/6 | C57BL/6 | CBA/JCr | 13, 15, 23 |
Chimeric NOD/Lt mice bearing C57BL/6 islet allografts were selected at random from among those presented in Figure 2 (group 1). They were given either a C57BL/6 (H2b, donor-specific) or a CBA/JCr (H2k, third-party) skin allograft as described in “Materials and Methods.” None of the islet allografts failed in either group during the period of observation.
Discussion
These results demonstrate that it is possible to generate hematopoietic chimerism in mice transplanted with fully allogeneic bone marrow using the combination of sublethal irradiation and a brief course of anti-CD154 mAb comprising as few as 2 injections. Chimeric mice treated according to this protocol showed no evidence of GVHD despite the presence of a peripheral lymphoid compartment almost entirely of donor origin. Chimeric mice also accepted donor-origin skin and islet allografts.
The minimum dose of radiation required to generate chimerism differed between the BALB/c and NOD/Lt strains. Chimerism could be generated with as little as 400 rad in BALB/c mice, whereas 600 rad appeared to be the threshold in NOD/Lt mice. These differences may reflect intrinsic differences between these strains with respect to overall radioresistance. The thresholds for chimerism in BALB/c and NOD/Lt mice were roughly proportional to the respective lethal radiation doses of 700 and 1000 rad. The radiation doses required for chimerism may simply be those that achieve adequate, but not lethal, degrees of elimination of host hematopoietic cells.
The costimulatory blockade produced by treatment with anti-CD154 mAb exerted 2 important effects. First, it lowered the dose of radiation required to achieve chimerism in both of the strain combinations tested. The effect was to some extent dependent on the overall dose of anti-CD154 mAb administered, but the data indicate clearly that prolonged treatment is not required. Administration of antibody on the day of transplantation, just before the transfusion of cells, and again 3 days later was sufficient to lead to hematopoietic chimerism.
The second major effect of costimulatory blockade was on the occurrence of GVHD. Treatment with anti-CD154 mAb was clearly associated with prevention of GVHD in successful chimeras. With few exceptions, chimeras generated using irradiation alone showed clinical characteristics of GVHD. In contrast, chimeras generated with the same dose of radiation but with the addition of anti-CD154 mAb showed no evidence of this common complication, either acutely or chronically, for up to about 11 months after bone marrow transplantation. These observations are consistent with previous reports documenting that treatment with anti-CD154 mAb can prevent GVHD in a model system based on the transfer of alloreactive T cells.9-11 These earlier studies demonstrated that anti-CD154 mAb can block the graft-versus-host activity of transferred alloreactive CD8+T cells. The present study extends these earlier reports by demonstrating that donor T cells that continue to develop in a fully reconstituted bone marrow allograft recipient are tolerant of the host in the absence of additional costimulatory blockade.
The absence of GVHD in mice treated with both sublethal irradiation and anti-CD154 mAb is particularly remarkable in view of the extent and durability of the chimerism achieved. The percentage of donor-origin mononuclear cells in both bone marrow and the periphery of recipients was more than 99%. With the exception of a single mouse, chimerism—when established—appeared to be permanent. The sole exception was the 1 mouse that had less than 99% (actually 24%) donor-origin cells present 4 weeks after transplantation. This exception highlights 1 unexpected characteristic of the chimeras generated with our protocol: If chimerism was present at all, it was virtually total. To the limit of our ability to detect it by flow microfluorometry, mixed chimerism occurred in only the 1 exceptional mouse, and in that animal it was not permanent.
The establishment of essentially complete hematopoietic chimerism using sublethal doses of radiation is not readily explained. It could be argued that extensive chimerism resulted from the induction of GVHD intense enough to eliminate host hematopoietic cells, but anti-CD154 mAb is known to prevent GVHD.9Alternatively, the extent of chimerism could be a cell dose effect. Our observations contrast with those of Wekerle et al, whose bone marrow transplantation protocol based on sublethal conditioning and both anti-CD154 mAb and CTLA4-Ig generated many chimeras that circulated only 70% to 80% donor-origin PBMCs. Those investigators, however, injected 15 × 106 donor cells, whereas we injected 25 × 106 cells. Interestingly, Wekerle et al also reported that their recipients treated with sublethal irradiation and anti-CD154 mAb monotherapy became chimeric, but the extent of chimerism was lower and declined over time. In preliminary experiments using our protocol, we have observed that the injection of 15 × 106 donor cells leads to permanent mixed allogeneic chimeras that are 60% to 90% donor-origin (D.L.G., unpublished data). It may be that the dose of radiation, the dose of transplanted donor bone marrow, and the precise regimen of costimulatory blockade exert independent effects on the extent and durability of chimerism achievable using this approach.
An additional interesting finding in our chimeric mice was the relatively low percentage of donor-origin mononuclear cells present in the thymus. Mice with more than 99% donor-origin cells in bone marrow, spleen, and peripheral blood in some instances had as few as 17% donor-origin thymocytes. Based on the high level of chimerism achieved in the bone marrow of these recipients and the normal turnover of cells in the thymus, this observation is surprising and unexplained. There is, however, a precedent for this observation. The extent of chimerism in the thymus was reportedly different from that in other tissues in hematopoietic chimeras that received high doses of Ly5 congenic marrow without any host pretreatment.38 The reason for the persistence of host-origin intrathymic progenitors is under investigation in our laboratory using an adult thymic organ culture system.39
The ability to generate fully allogeneic chimeric mice free of GVHD provided us with the opportunity to perform additional studies in the areas of transplantation and autoimmunity. As expected, based on the work of Owen16 and many others,40-42 allogeneic chimeric mice were able to accept donor-origin allografts while retaining the ability to reject third-party grafts. Because skin contains a large number of professional antigen-presenting cells, skin allografts represent a robust stimulus for rejection. Our observation of prolonged skin allograft survival in mice treated with irradiation and anti-CD154 mAb suggests that the tolerant state induced by our protocol is robust and may support the transplantation of other vascular organs. It is important to point out that treatment with anti-CD154 mAb was required for uniform donor skin graft tolerance in chimeric mice. The survival of skin allografts placed on animals that had been rendered chimeric by irradiation alone tended to be short, and in the cases in which skin grafts were not immediately rejected, GVHD appeared to be present. Several mice with GVHD and skin allografts died.
Our observation that it was possible to generate hematopoietic chimerism in NOD/Lt mice using sublethal irradiation and costimulatory blockade was somewhat surprising. We have reported that there may be a general defect in tolerance mechanisms in NOD/Lt mice.33 We observed that NOD/Lt mice are resistant to the induction of transplantation tolerance based on the administration of donor-specific transfusion and anti-CD154 mAb33—a procedure that induces transplantation tolerance in a variety of normal mice.25,43 44 The present data suggest that this defect in tolerance mechanisms in NOD/Lt mice can be overcome by stem cell transplantation.
It has previously been shown that creation of mixed allogeneic chimerism without costimulatory blockade is sufficient to prevent the development of spontaneous diabetes in young NOD mice.45-47The present data suggest that stem cell transplantation using sublethal conditioning and anti-CD154 mAb should be investigated as a potential method for prevention of autoimmune diseases in which determinants of susceptibility reside in bone marrow.19 It will be of interest to determine if the reversal of autoimmune diatheses by stem cell transplantation is associated with some degree of recovery in affected organ systems.
In the case of type 1 diabetes, which occurs in the NOD mouse, the tissue affected by autoimmunity is destroyed. Replacement of the affected tissue in these instances requires procedures that overcome not only allorejection but also recurrent autoimmune destruction of the transplanted tissue. Diabetic NOD mice reject syngeneic islet grafts,33 and only lethal irradiation followed by allogeneic bone marrow transplantation has prevented recurrent autoimmunity in donor-origin islet allografts.37We now show that it is possible to cure autoimmune diabetes in NOD/Lt mice by first generating hematopoietic chimerism using sublethal irradiation plus a brief course of anti-CD154 mAb and then transplanting donor-specific islets of Langerhans. These grafts restored normoglycemia, and none failed during the period of experimental observation. The data suggest that the generation of allogeneic hematopoietic chimerism based on sublethal irradiation and costimulatory blockade can prevent both allograft rejection and recurrent autoimmunity.
The use of costimulatory blockade to facilitate transplantation tolerance induction in primates has recently been reported.48,49 Stem cell transplantation has not been adapted to the clinic for transplantation tolerance induction due to the significant morbidity and mortality still inherent in current treatment protocols.1 19 It is reasonable to suggest that protocols for murine stem cell transplantation based on sublethal conditioning and blockade of costimulation may be amenable to translation for use in clinical medicine.
Acknowledgments
The authors thank Linda Paquin, Elaine Norowski, Linda Leehy, and Shouying Wang for their technical assistance.
Supported in part by program project DKAI53006 and center grant 5PODK32520 from the National Institutes of Health and by a grant from the Hood Foundation. The contents of this publication are solely the responsibility of the authors and do not necessarily represent the official views of the National Institutes of Health.
Reprints:Dale L. Greiner, Diabetes Division, University of Massachusetts Medical School, Two Biotech, 373 Plantation St, Suite 218, Worcester, MA 01605; e-mail: dale.greiner@umassmed.edu.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal