Key Points
Disease relapse is reduced after DUCBT if the LU and the patient share the same HLA mismatch with the WU.
Visual Abstract
During double umbilical cord blood transplant (DUCBT), the winning unit (WU) rejects the losing unit (LU) because of WU T cells directed against the LU mismatched HLA. This immune response might protect against relapse, especially when the patient and LU (PT-LU) share the same mismatch with the WU. To validate this hypothesis, a retrospective Eurocord study, conducted on 383 DUCBTs, focused on posttransplant relapse and HLA mismatches between PT-LU and the WU. A PT-LU HLA-A mismatch shared with the WU was associated with a lower 7-year relapse incidence (16% vs 28%; P = .048). In addition, multiple PT-LU shared HLA mismatches were also associated with a lower relapse risk (7% vs 29%; P = .003). In DUCBT with ≥2 HLA mismatches between patient and WU, the number of HLA mismatches between those did not significantly affect relapse incidence, whereas multiple PT-LU shared HLA mismatches remained associated with a lower relapse risk (7% vs 29%; P = .0038). Finally, considering patients who did not develop either grade 2 to 4 acute graft-versus-host disease or chronic graft-versus-host disease, a PT-LU shared HLA-A mismatch as well as multiple PT-LU HLA mismatches shared with the WU remained associated with a significantly lower 7-year relapse incidence. In multivariate adjusted analyses multiple PT-LU shared HLA mismatches remained associated with a significantly reduced 7-year posttransplant relapse risk. Our analysis indicates that, during DUCBT, PT-LU shared HLA mismatches prime an immune response of the WU against leukemia, reducing the long-term risk of posttransplant relapse, and that DCBT has particular utility in those with high-risk leukemia.
Introduction
Hematopoietic stem cell transplant has the potential to cure hematologic malignancies that would not have been cured with chemotherapy alone. Reducing disease relapse is primarily mediated by a graft-versus-leukemia (GVL) effect, with engrafted donor immune cells, including donor T cells, recognizing residual recipient leukemia as foreign histoincompatible cells.1 Optimizing GVL is critical and must be managed in combination with strategies that reduce graft-versus-host disease (GVHD). However, graft T-cell depletion and intensified pretransplant and posttransplant immune suppression are themselves associated with increased disease relapse.
Matching for HLA between recipient and donor contributes significantly to successful graft outcome, but its involvement in the GVL effect is not as well characterized. The critical role of HLA in the pathogenesis of posttransplant leukemia relapse is evidenced by the frequent downregulated expression or deletion of one of the HLA genes at relapse, with residual leukemia cells subsequently escaping the GVL immune response.2 Furthermore, after haplo-identical transplant, it is the mismatched HLA haplotype that is preferentially downregulated, suggesting that major HLA mismatch might itself be a GVL target and prevent disease relapse.3
There are institutional and registry studies reporting improved outcomes using cord blood (CB) as a donor cell source compared with other cell sources, especially in those with residual measurable disease at transplant.4-6 Such benefit, likely, reflects that CB transplant is more often performed both T-cell replete and HLA mismatched compared with other stem cell sources, thus promoting a more efficient and sustained GVL effect. Increased donor-recipient HLA mismatch has been reported to be associated with reduced relapse risk after transplant7 and CB selection guidelines even advise against selection of a fully matched unit in disease at high risk of posttransplant relapse.8
The cell dose of the cryopreserved CB unit is an important determinant of transplant outcomes9 in patients with high body weight, and double umbilical CB transplant (DUCBT) is often performed to improve cell dose and limit the risk of nonengraftment. During DUCBT, 1 unit predominantly engrafts, usually called the “winning unit” (WU), whereas the other unit is lost, usually called the “losing unit” (LU).10
Others have reported HLA-specific responses mediated by WU T cells directed at the HLA mismatches between the WU and the LU.11 We have hypothesized that such an HLA-mediated response might be associated with a reduced relapse risk if the patient and the LU (PT-LU) share the same HLA mismatch with the WU, because HLA-restricted recognition of the PT-LU mismatch shared by the WU immune system might promote rejection of the leukemic cells.
To test our hypothesis, we evaluated the impact of HLA mismatches between WU, LU, and recipient on relapse risk after transplant for hematologic malignancies in 383 consecutive DUCBTs performed in European Society for Blood and Marrow Transplantation (EBMT) centers between 2005 and 2022.
Methods
Study design
This is a multicenter retrospective registry-based study on DUCBT performed between 2005 and 2022 in adult or pediatric patients with acute leukemia or myelodysplastic syndrome (MDS), which were reported to Eurocord/EBMT. The Eurocord registry collects data on DUCBT performed in Europe and other participating countries. This study was performed in collaboration with the Cellular Therapy and Immunobiology Working Party of the EBMT.
Inclusion and exclusion criteria
Patients were eligible for this study if they met the following criteria: diagnosis of acute myeloid leukemia, acute lymphoblastic leukemia, or MDS; DUCBT performed as first allogeneic transplant at EBMT centers; having HLA typing available at antigen level (low-resolution typing) for HLA-A, HLA-B, and HLA-C, and high-resolution typing for HLA-DRB1 for the recipient and both CB units; and documented engraftment and chimerism data at day +100 after transplant evaluation (100-day registry follow-up data) or at later follow-up when chimerism at day +100 evaluation was not available. Patients who had received T-cell depleting serotherapy during the conditioning regimen were excluded. Patients lacking data on relapse status or on T-cell depleting serotherapy were also excluded. All patients or legal guardians provided written informed consent for transplant and data collection. The study was approved by the institutional review board of the Eurocord scientific committee and performed according to the Declaration of Helsinki.
Definitions and end points
The WU was defined as the CB unit representing >50% of blood chimerism at day +100 (100-day registry follow-up data) or at later follow-up when chimerism at the day +100 evaluation was not available after allogeneic DUCBT.
The influence of HLA mismatch on DUCBT outcomes considered classical loci (HLA-A and HLA-B at low-resolution typing, and HLA-DRB1 at high-resolution typing) as well as HLA-C at low-resolution typing; and focused on the recipient and LU matched at an HLA determinant that was mismatched with the WU, as exemplified in Table 1.
Myeloablative conditioning regimen was defined as a regimen containing either a total body irradiation with a dose of >6 Grays or a total dose of busulfan of at least 8 mg/kg given orally, or 6.4 mg/kg given IV.
Disease status at transplant was reported by each transplant center and classified as either first or second remission, greater than second remission, or active disease. Transplant-related mortality (TRM) was defined as death without relapse.
The primary end point was relapse at 7 years from DUCBT. Secondary end point was TRM at 7 years from DUCBT.
Statistical methods
To describe the study population, percentages were reported for categorical covariates and median value with ranges for continuous covariates. Estimates of the 7-year cumulative incidence of relapse (CIR) and TRM as well as their 95% confidence interval (95% CI) were calculated using the cmprsk library for S-Plus. Univariate comparative analyses were performed using Gray test for the 7-year CIR and TRM. TRM was considered as the competing risk for relapse, when studying relapse. Relapse was considered as the competing risk for TRM, when studying TRM. Multivariate analyses based on the Fine and Gray model were used to assess the effect of a given covariate assessing HLA sharing adjusted on the covariates significantly affecting the considered end points, that is, relapse or TRM. Results of the statistical tests were based on 2-sided P values, with type I error fixed at .05 for statistical significance. Statistical analyses were performed using S-Plus 8.0 Enterprise Developer software (Insightful Corp).
Results
Characteristics of the patients and transplant procedures
A total of 383 patients met the eligibility criteria. Characteristics of these patients as well as of the transplant procedures are summarized in Table 2. The median year of DUCBT was 2012. Briefly, most patients had acute leukemia in complete remission at the time of CBT. Of note, cyclophosphamide combined with fludarabine and total body irradiation was the most commonly used conditioning regimen whereas cyclosporine combined to mycophenolate mofetil was the most frequently used GVHD prophylaxis. Median follow-up time for the 204 patients alive at last contact was 6 years (range, 0.3-16.2). Among 383 patients, 191 (50%) shared an HLA specificity with the LU that mismatched the WU. These PT-LU shared mismatches involved HLA-A in 68 transplants (18%), HLA-B in 58 (15%), HLA-C in 77 (20%), and HLA-DRB1 in 47 transplants (12%). In addition, these PT-LU shared mismatches occurred at 2 and 3 HLA loci simultaneously in 41 (11%) and 9 (2%) transplants, respectively. In those combined shared mismatches, HLA-A, HLA-B, HLA-C, and HLA-DRB1 were involved in 26, 29, 38, and 16 cases, respectively.
For the whole group of patients, the 7-year overall and event-free survivals were 52% (95% CI, 47-57) and 48% (95% CI, 43-54), respectively. Seven-year posttransplant CIR and TRM, which are the main focus of this study, are detailed hereafter as well as analyses on acute and chronic GVHD.
CIR
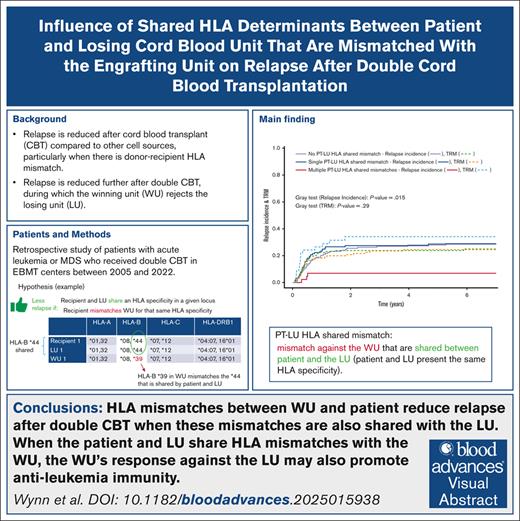
The 7-year CIR was 26% (95% CI, 21-31) for the overall group of patients. In univariate analysis, the CIR was significantly lower when there was a PT-LU HLA-A shared mismatch against the WU (Figure 1A; Table 3). A trend toward a reduced relapse risk was also observed when considering PT-LU HLA-B shared mismatches (Figure 1B; Table 3). No significant difference was observed for HLA-C and HLA-DRB1 (Figure 1C-D; Table 3).
The 7-year CIR and TRM (competing risk) according to the presence of a PT-LU shared HLA mismatch against the WU. (A) CIR and TRM according to the presence of a PT-LU HLA-A shared mismatch against the WU. With no PT-LU HLA-A shared mismatch (n = 315) the CIR was 28% (95% CI, 23-34) vs 16% (95% CI, 7-25) with a PT-LU HLA-A shared mismatch (n = 68). With no PT-LU HLA-A shared mismatch, the TRM was 25% (95% CI, 20-30) vs 26% (95% CI, 15-37) with a PT-LU HLA-A shared mismatch. (B) CIR and TRM according to the presence of a PT-LU HLA-B shared mismatch against the WU. With no PT-LU HLA-B shared mismatch (n = 325) the CIR was 28% (95% CI, 23-33) vs 15% (95% CI, 5-24) with a PT-LU HLA-B shared mismatch (n = 58). With no PT-LU HLA-A shared mismatch the TRM was 25% (95% CI, 20-30) vs 30% (95% CI, 17-42) with a PT-LU HLA-B shared mismatch. (C) CIR and TRM according to the presence of a PT-LU shared HLA-C mismatch against the WU. With no PT-LU HLA-C shared mismatch (n = 306) the CIR was 27% (95% CI, 22-32) vs 22% (95% CI, 12-31) with a PT-LU HLA-C shared mismatch (n = 77). With no PT-LU HLA-C shared mismatch the TRM was 26% (95% CI, 21-31) vs 24% (95% CI, 14-34) with a PT-LU HLA-C shared mismatch. (D) CIR and TRM according to the presence of a PT-LU shared HLA-DRB1 mismatch against the WU. With no PT-LU HLA-DRB1 shared mismatch (n = 335) the CIR was 26% (95% CI, 21-31) vs 28% (95% CI, 13-42) with a PT-LU HLA-DRB1 shared mismatch (n = 47). With no PT-LU HLA-DRB1 shared mismatch the TRM was 24% (95% CI, 20-29) vs 34% (95% CI, 20-48) with a PT-LU HLA-DRB1 shared mismatch.
The 7-year CIR and TRM (competing risk) according to the presence of a PT-LU shared HLA mismatch against the WU. (A) CIR and TRM according to the presence of a PT-LU HLA-A shared mismatch against the WU. With no PT-LU HLA-A shared mismatch (n = 315) the CIR was 28% (95% CI, 23-34) vs 16% (95% CI, 7-25) with a PT-LU HLA-A shared mismatch (n = 68). With no PT-LU HLA-A shared mismatch, the TRM was 25% (95% CI, 20-30) vs 26% (95% CI, 15-37) with a PT-LU HLA-A shared mismatch. (B) CIR and TRM according to the presence of a PT-LU HLA-B shared mismatch against the WU. With no PT-LU HLA-B shared mismatch (n = 325) the CIR was 28% (95% CI, 23-33) vs 15% (95% CI, 5-24) with a PT-LU HLA-B shared mismatch (n = 58). With no PT-LU HLA-A shared mismatch the TRM was 25% (95% CI, 20-30) vs 30% (95% CI, 17-42) with a PT-LU HLA-B shared mismatch. (C) CIR and TRM according to the presence of a PT-LU shared HLA-C mismatch against the WU. With no PT-LU HLA-C shared mismatch (n = 306) the CIR was 27% (95% CI, 22-32) vs 22% (95% CI, 12-31) with a PT-LU HLA-C shared mismatch (n = 77). With no PT-LU HLA-C shared mismatch the TRM was 26% (95% CI, 21-31) vs 24% (95% CI, 14-34) with a PT-LU HLA-C shared mismatch. (D) CIR and TRM according to the presence of a PT-LU shared HLA-DRB1 mismatch against the WU. With no PT-LU HLA-DRB1 shared mismatch (n = 335) the CIR was 26% (95% CI, 21-31) vs 28% (95% CI, 13-42) with a PT-LU HLA-DRB1 shared mismatch (n = 47). With no PT-LU HLA-DRB1 shared mismatch the TRM was 24% (95% CI, 20-29) vs 34% (95% CI, 20-48) with a PT-LU HLA-DRB1 shared mismatch.
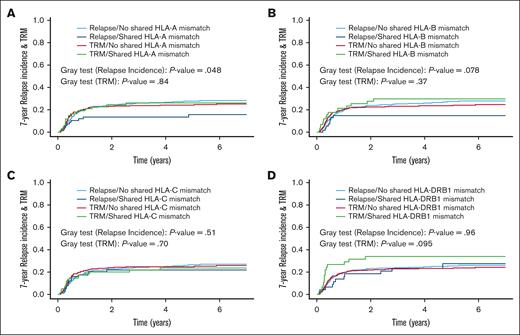
Moreover, compared with none or a single PT-LU HLA shared mismatch, the 7-year CIR was significantly lower when there were multiple PT-LU HLA shared mismatches against the WU (Figure 2; Table 3).
The 7-year CIR and TRM (competing risk) according to the number of PT-LU HLA locus shared mismatches against the WU. With no or a single PT-LU HLA shared mismatch against the WU (n = 334) CIR was 29% (95% CI, 24-34) vs 7% (95% CI, 0-14) with multiple PT-LU HLA shared mismatches (n = 48). With no or a single PT-LU HLA shared mismatch against the WU the TRM was 24% (95% CI, 19-29) vs 35% (95% CI, 21-49) with multiple PT-LU HLA shared mismatches.
The 7-year CIR and TRM (competing risk) according to the number of PT-LU HLA locus shared mismatches against the WU. With no or a single PT-LU HLA shared mismatch against the WU (n = 334) CIR was 29% (95% CI, 24-34) vs 7% (95% CI, 0-14) with multiple PT-LU HLA shared mismatches (n = 48). With no or a single PT-LU HLA shared mismatch against the WU the TRM was 24% (95% CI, 19-29) vs 35% (95% CI, 21-49) with multiple PT-LU HLA shared mismatches.
To more precisely define the influence of PT-LU shared mismatches at each HLA locus, 3 groups were compared: group 1 had no PT-LU shared mismatch for the considered HLA locus; group 2 had a single PT-LU shared mismatch only for this considered locus; and group 3 had a PT-LU shared mismatch in the considered locus as well as in at least 1 additional HLA locus. Except when HLA-B locus was considered as the starting point, the 7-year CIR was significantly lower when multiple PT-LU shared mismatches were observed as compared with none or a single PT-LU shared mismatch for HLA-A, HLA-C, and HLA-DRB1 (Table 3).
Although the maximum number of HLA mismatches between the recipient and the WU was not significantly affecting the relapse risk in univariate analysis (relative risk [RR], 1.05; 95% CI, 0.87-1.28; P = .59), a last group of univariate analyses was performed focusing on 320 patients (84% of the overall group) who had at least 2 HLA mismatches with the WU. In those patients, multiple PT-LU HLA shared mismatches against the WU were associated with a significantly lower 7-year CIR when compared with those who have none or only a single shared mismatch without a significant increase of the 7-year TRM (Figure 3). When considering PT-LU HLA-A shared mismatch combined to an additional shared HLA locus mismatch against the WU, the 7-year CIR was also significantly reduced (combined PT-LU HLA-A shared mismatch, 0% vs none or a single PT-LU HLA-A shared mismatch, 28% [95% CI, 21-35; P = .0033]) without a significant increase of the 7-year TRM. Similar findings were observed for HLA-C (combined PT-LU HLA-C shared mismatch, 9% [95% CI, 0-18] vs none or a single PT-LU HLA-C shared mismatch, 30% [95% CI, 23-37]; P = .014).
The 7-year CIR and TRM (competing risk) according to the number of PT-LU HLA locus shared mismatches against the WU and considering patients with at least 2 HLA mismatches with the WU. With no PT-LU HLA shared mismatch against the WU (n = 146), the CIR was 29% (95% CI, 21-37) vs 29% (95% CI, 20-37) with a single PT-LU HLA shared mismatch (n = 127) vs 7% (95% CI, 0-15) with multiple PT-LU HLA shared mismatches (n = 46). With no PT-LU HLA shared mismatch against the WU, the TRM was 25% (95% CI, 18-32) vs 24% (95% CI, 17-32) with a single PT-LU HLA shared mismatch vs 34% (95% CI, 20-49) with multiple PT-LU HLA shared mismatches.
The 7-year CIR and TRM (competing risk) according to the number of PT-LU HLA locus shared mismatches against the WU and considering patients with at least 2 HLA mismatches with the WU. With no PT-LU HLA shared mismatch against the WU (n = 146), the CIR was 29% (95% CI, 21-37) vs 29% (95% CI, 20-37) with a single PT-LU HLA shared mismatch (n = 127) vs 7% (95% CI, 0-15) with multiple PT-LU HLA shared mismatches (n = 46). With no PT-LU HLA shared mismatch against the WU, the TRM was 25% (95% CI, 18-32) vs 24% (95% CI, 17-32) with a single PT-LU HLA shared mismatch vs 34% (95% CI, 20-49) with multiple PT-LU HLA shared mismatches.
Regarding posttransplant covariates, grade 2 to 4 acute GVHD was associated with a significant reduction of the 7-year CIR (Table 3). Grade 3 to 4 acute GVHD as well as chronic GVHD were also associated with a lower CIR, but the difference did not reach statistical significance (Table 3).
In multivariate adjusted analyses, multiple PT-LU HLA shared mismatches remained significantly associated with a lower relapse risk, whether considering only pretransplant covariates that significantly influenced the 7-year CIR in univariate analysis (Table 4) or adding other covariates such as cytogenetics and posttransplant covariates grade 2 to 4 acute GVHD (Table 4) or chronic GVHD (Table 4). When replacing the number of PT-LU HLA shared mismatches against the WU in the first multivariate model by the presence of a PT-LU shared HLA-A mismatch, a trend toward a lower relapse risk was observed in patients with such a mismatch (RR, 0.55; 95% CI, 0.28-1.07; P = .076). This trend became significant in the second and third multivariate models that considered cytogenetics and either grade 2 to 4 acute GVHD (RR, 0.48; 95% CI, 0.23-0.98; P = .045) or chronic GVHD (RR, 0.43; 95% CI, 0.19-0.94; P = .035). This finding was mainly because of the patients with a PT-LU shared HLA-A mismatch combined with an additional shared mismatch at another HLA locus (no relapse for those patients). For HLA-B, HLA-C, or HLA-DRB1 loci, a PT-LU shared mismatch against the WU, either alone or combined with another shared HLA locus mismatch, had no significant impact of the relapse risk in multivariate analysis.
Acute GVHD
The cumulative incidences of grade 2 to 4 and grade 3 to 4 acute GVHD were 51% (95% CI, 46-56) and 19% (95% CI, 15-23), respectively, for the overall group of patients.
Neither any of the PT-LU HLA shared mismatches against the WU, nor the number of PT-LU HLA shared mismatches, nor the other covariate assessing the HLA compatibility between the recipient and the CB units significantly affected grade 2 to 4 acute GVHD incidence.
Regarding grade 3 to 4 acute GVHD incidence, a PT-LU HLA-A shared mismatch against the WU was associated with a significantly lower incidence of this complication (no PT-LU HLA-A shared mismatch, 21% [95% CI, 16-26] vs a PT-LU HLA-A shared mismatch, 9% [95% CI, 2-15]; P = .019). Similar results were observed when comparing patients with no or a single PT-LU HLA-A shared mismatch (21%; 95% CI, 15-26) and those with a shared HLA-A mismatch combined with another shared HLA locus mismatch (4%; 95% CI, 0-11; P = .04). No other HLA-related covariate significantly affected grade 3 to 4 acute GVHD incidence.
Chronic GVHD
The cumulative incidences of chronic GVHD and extensive chronic GVHD were 46% (95% CI, 40-52) and 21% (95% CI, 16-26), respectively.
Although not reaching statistical significance, a PT-LU HLA-DRB1 shared mismatch against the WU was associated with a lower incidence of chronic GVHD (no PT-LU HLA-DRB1 shared mismatch, 48% [95% CI, 42-54] vs a PT-LU HLA-DRB1 shared mismatch, 31% [95% CI, 16-47]; P = .052), with a significant increase of the TRM unrelated to this complication (no PT-LU HLA-DRB1 shared mismatch, 13% [95% CI, 8-17] vs a PT-LU HLA-DRB1 shared mismatch, 25% [95% CI, 10-40]; P = .014). Patients with multiple PT-LU HLA shared mismatches against the WU also had a lower chronic GVHD incidence (none or a single PT-LU HLA shared mismatch, 48% [95% CI, 42-54] vs multiple PT-LU HLA shared mismatches, 37% [95% CI, 19-55]; P = .076), without any significant difference in terms of the TRM unrelated to this complication (P = .52). No other HLA-related covariates significantly affected chronic GVHD incidence.
Regarding extensive chronic GVHD, patients with a PT-LU HLA-A shared mismatch combined with another PT-LU shared HLA locus mismatch against the WU had a significantly lower extensive chronic GVHD incidence (none or a single PT-LU HLA-A shared mismatch, 24% [95% CI, 18-30)] vs a combined PT-LU HLA-A shared mismatch, 5% [95% CI, 0-14]; P = .044), without a significant difference in terms of TRM unrelated to this complication (P = .78). Similarly, although not reaching statistical significance, patients with a PT-LU HLA-C shared mismatch combined with another PT-LU shared HLA locus mismatch against the WU had a lower extensive chronic GVHD incidence (none or a single PT-LU HLA-C shared mismatch, 23% [95% CI, 17-29] vs a combined PT-LU HLA-C shared mismatch, 6% [95% CI, 0-15]; P = .052), with a lower TRM unrelated to this complication (none or a single PT-LU HLA-C shared mismatch, 44% [95% CI, 36-52] vs a combined PT-LU HLA-C shared mismatch, 25% [95% CI, 6-43]; P = .059).
Moreover, a PT-LU HLA-DRB1 shared mismatch against the WU was also associated with a lower extensive chronic GVHD incidence (no PT-LU HLA-DRB1 shared mismatch, 23% [95% CI, 17-28] vs a PT-LU HLA-DRB1 shared mismatch, 9% [95% CI, 0-19; P = .068]), without a significant difference in terms of TRM unrelated to this complication (P = .44).
Finally, multiple PT-LU HLA shared mismatches against the WU were associated with a significantly lower extensive chronic GVHD incidence (none or a single PT-LU HLA shared mismatch, 23% [95% CI, 18-28] vs multiple PT-LU HLA shared mismatches, 8% [95% CI, 0-17]; P = .042), without any significant difference in terms of TRM unrelated to this complication (P = .66). No other HLA-related covariates significantly affected extensive chronic GVHD incidence.
TRM
The 7-year TRM was 28% (95% CI, 22-32) for the overall group of patients. In univariate analysis, a trend toward a higher 7-year TRM was observed when multiple PT-LU shared mismatches were observed (none or 1 PT-LU shared mismatch, 24% [95% CI, 19-29]; multiple PT-LU shared mismatches, 35% [95% CI, 21-49]; P = .067; Figure 2). Focusing on each HLA locus, a trend toward an increased 7-year TRM was observed when a PT-LU shared mismatch HLA-DRB1 (TRM, 34% [95% CI, 20-48] vs 24% [95% CI, 20-29] with no PT-LU shared mismatch affecting HLA-DRB1; P = .095; Figure 1D). This finding was because of the effect of multiple PT-LU shared mismatches, of which, 1 was in HLA-DRB1 (TRM, 55% [95% CI, 27-82]; none or a single PT-LU shared mismatch in HLA-DRB1, 25% [95% CI, 19-30]; P = .004). No statistically significant influence on the 7-year TRM was observed for HLA-A, HLA-B, or HLA-C PT-LU shared mismatches (Figure 1A-C).
Discussion
The improved outcome of allogeneic stem cell transplants, especially in older patients, has mainly been because of a TRM reduction with better donor selections, high-resolution HLA typing, and the enhanced ability to manage HLA-mismatched transplants using either haplotype-mismatched family donors,12-15 mismatched unrelated adult donors, or CB donors. Advances in conditioning regimens, GVHD prevention such as posttransplant cyclophosphamide, and new cytomegalovirus prophylaxis with letermovir,16 have also contributed to reduce TRM in HLA-mismatched transplants.
However, relapse remains the major cause of death in patients who have received transplantation for acute leukemia or MDS. For instance, in adult patients with acute myeloid leukemia, the CIR has only decreased from 30% to 20% before and after 2000, whereas in adult patients with acute lymphoblastic leukemia, the CIR has not significantly changed over time (EBMT registry reports).17
Disease subtype and status at transplant, type and intensity of the conditioning regimen, as well as the complex interplay of immunobiological processes between the recipient and the donor(s) contribute to disease relapse. Graft immunological reactivity plays a critical role in controlling the disappearance of residual leukemic cells. However, this protective GVL effect is strongly counterbalanced by the occurrence of acute and/or chronic GVHD and by the immunosuppressive treatment required to manage these reactions.
At relapse, HLA coding genes can be either targeted by chromosomal deletion that lead to their partial or complete loss,3 or their expression on leukemic cells can be downregulated after transplantation. In haplo-identical transplants, the mismatched HLA is frequently lost, suggesting that the mismatched HLA, itself, is a target of the GVL effect.18 Recipient/donor HLA mismatch is critical in controlling disease relapse, especially after CB transplants. As reported in registry-based CB studies, relapse is likely to be more frequent when the recipient and CB unit are closely matched.14,15 In DUCBT, HLA mismatches between the patient and both CB units have been shown to influence transplant outcomes, a high level of HLA mismatches being associated with a lower CIR.19,20
In solid organ transplant, recipient T-cell and B-cell immune responses against the HLA-mismatched solid organ are critical for transplant rejection. Those immune responses can be stimulated through exposure to the mismatched HLA molecules encountered during blood cell transfusion, so that graft loss is accelerated if the transfusion and the transplanted organ share the same HLA mismatch with the recipient.21
In DUCBT, a similar process has been suggested, during which, LU leucocytes stimulate a T-cell response of the WU directed against the HLA mismatches between the 2 CB units.11,22 WU T cells, specifically directed at the HLA mismatch, have been associated with LU rejection. After the first retrospective observations,11 a prospective phase 2 study performed by the cooperative HOVON group validated these findings with particular emphasis on CD4+ T cells with HLA class II single-allele specificity, that could readily be detected early after DUCBT.22
Our study supports the hypothesis that if the recipient and the LU share the same HLA mismatch with the WU, then the immune response of WU against LU might be extended to the recipient malignant cells, hence reducing posttransplant CIR. Furthermore, we demonstrate that the reduced relapse associated with HLA mismatch during DCBT is only when that mismatched HLA is shared between patient and LU, and with the WU. Additionally, and importantly, such shared HLA mismatch between LU and patient is associated with neither increased GVHD nor with increased TRM. Of note, our adjusted analyses showed that multiple PT-LU HLA shared mismatches remained significantly associated with a lower relapse risk independent of important pretransplant factors such as cytogenetic risk. Nevertheless, these results need to be viewed with caution because they were based on a small number of patients in the high-risk group and with different diagnoses.
Our data suggest that the primary HLA molecule involved in this tripartite GVL process is HLA-A and the shared HLA-A–mismatch effect on CIR becomes pronounced when combined with other PT-LU shared mismatches affecting other HLA loci. A potential explanation for the central involvement of HLA-A is the close proximity and possible linkage with nonclassical HLA molecules including HLA-E. HLA-E is a nonclassical HLA molecule that binds and displays peptides from the signal peptides of HLA-A, -B, or -C on the surface of all cells. Bridging the innate and adaptive immune systems, HLA-E is a ligand of natural killer (NK) group 2 family of receptor A (NKG2A) and NKG2C NK-cell receptors that plays a pivotal role in modulating NK-cell inhibitory/activating properties, with a major role in antileukemic processes.23 In combination, this interaction can also create an equilibrium between GVHD and the GVL effect.24 Polymorphic variants within HLA-E, and within the signal peptides of class I molecules have been shown to affect the amount of HLA-E expression, which, in turn, may alter the dynamics of DUCBT engraftment. This appears to be reinforced by our analysis showing a significantly lower CIR when the WU has 2 or 3 HLA shared mismatches with the LU and recipient pair.
PT-LU HLA-DRB1 shared mismatches against the WU were associated with an increased risk of early TRM after DUCBT (Figure 1D). HLA class II antigens do not interact with HLA-E and therefore are unlikely to direct the equilibrium between GVHD and the GVL effect, as opposed to HLA-A antigens.
Altogether, our study suggests that, although relapse seems to be controlled by HLA class I molecules, an immune response mediated by HLA class II variants may, at least in part, govern TRM. Our results suggest that the role of HLA class I antigens affecting rejection/relapse risk should be distinguished from the role of HLA class II antigens that are more likely to drive TRM and possibly GVHD. Nevertheless, the role of class II antigens in transplant outcome requires further investigation.
The major limitations of our study were the relatively small number of studied patients, the heterogeneity of included diseases, and the retrospective nature of our data. We were unable to independently study the impact of mismatches at HLA-DQ and HLA-DP, which have been shown to be associated with the mechanism of graft predominance in DUCBT11 and to be associated with a low relapse risk. Tight linkage disequilibrium between HLA-DRB1 and DQB1 would predict that mismatching at HLA-DRB1 in our data would include a high likelihood of additional mismatching at DQB1 and that the effects observed with DRB1 are representative of mismatching at more than 1 class II locus. These limitations and that it is difficult to predict WU and LU before DUCBT mean that our results do not yet influence clinical practice, including the selection of CB units for transplant. We encourage future investigations, with the collaboration of additional transplant groups, in order to extend our study to a larger scale to validate these results and to better characterize the relationship between recipient and CB units HLA mismatching and the GVL effect.
Despite these limitations, our data are consistent with other scientific reports showing that relapse is reduced in DUCBT transplant,25 that HLA mismatch is associated with a further reduction of relapse risk,19,20 and that T cells have been isolated and characterized as being directed at the LU HLA mismatched to the WU during the establishment of unit dominance.11
Taken together, our results support the continuing use of DUCBT, particularly for patients with acute leukemia or MDS at high risk of relapse. Furthermore, we, have reported recently that T cells may be expanded during CB transplant after exposure to mismatched leukocytes and with a phenotypic switch, suggesting antigen-specific responses.26 These cell sources may present mismatched HLA that is shared with the recipient, in the same way as the LU does. The importance of the CB transplant platform is that it is a T-cell replete graft, and that specific T-cell responses can be induced early, including against HLA mismatches, as described in our study. Those responses might explain the reduced relapse incidence after CB transplant. Our findings lay the groundwork to better understanding disease relapse after allogeneic transplantation that remains a significant limitation on patient outcomes after transplant.
Authorship
Contribution: R.W., F.V., S.S., and E.G. formulated the hypothesis and designed the research design; R.W., S.S., K.P.K., E.G., F.V., and P.G. analyzed the data and wrote earlier drafts of the manuscript; F.V. and P.G. performed expert statistical analyses; F.V., G.S., C.K., B.C., A.R., and E.G. identified suitable patients and gathered data from the Eurocord database; R.T., V.R., C.A., R.P.d.l.T., K.C., A.C., M.S., P.A., M.B., and J.J.C. contributed specific patient data to allow registered patients to be eligible for this particular study; and all authors read and approved the final manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Robert Wynn, Blood and Marrow Transplant Program, Royal Manchester Children’s Hospital, Oxford Rd, Manchester M13 9WL, United Kingdom; email: robert.wynn@mft.nhs.uk.
References
Author notes
Data are available on request from the author, Fernanda Volt (fernanda.volt-ext@aphp.fr).