Key Points
The DRCI predicts overall survival of patients undergoing alloHCT.
The DRCI is a prognostic scoring system for evaluating patients eligible for alloHCT.
Abstract
Allogeneic hematologic cell transplantation (alloHCT) is the only curative therapy for many adults with hematological malignancies. However, it can be associated with substantial risks of morbidity and mortality that are dependent on patient comorbidity– or disease risk–related factors. Several pretransplantation prognostic scoring systems have been developed to estimate survival of patients undergoing alloHCT; however, there is significant interstudy variability in the predictive capacity of these assessment tools. We tested the prognostic capability of a composite scoring system including the disease risk index and HCT comorbidity index (DRCI). The DRCI scoring system was applied pretransplantation to determine whether it predicted clinical outcomes of 959 adult patients with hematological malignancies undergoing alloHCT from 2000 to 2013 at the University of Minnesota. The DRCI score categorized patients into 6 risk groups, with 2-year overall survival ranging between 74% for the very low-risk DRCI group and 34% for the very high-risk DRCI group. In multiple regression analyses adjusted for patient age and donor type, the risk of overall mortality independently increased as the DRCI score increased. Additionally, the DRCI score independently predicted risk of relapse, disease-free survival, and graft-versus-host disease–free/relapse–free survival. Our data demonstrate that the pretransplantation DRCI scoring system predicts outcomes after alloHCT and can be used to guide clinical decision making for patients considering alloHCT.
Introduction
Allogeneic hematopoietic cell transplantation (alloHCT) is frequently used as a potentially curative therapy for many adults with various hematological malignancies. Survival of patients after alloHCT, however, is largely dependent on many disease- and patient-related factors. The refined disease risk index (DRI) considers disease type and disease status at the time of transplantation, and it has recently been identified as a powerful prognostic indicator for overall survival (OS) of patients undergoing alloHCT.1,2 DRI only accounts for disease-related factors at the time of transplantation1,2 ; it does not consider patient-related factors, such as comorbidities that have been consistently shown to affect risk of mortality after alloHCT.3-6 The HCT comorbidity index (HCT-CI), in contrast, accounts for only concurrent comorbidities at the time of transplantation and does not address characteristics of the underlying diagnosis. Although it too has been associated with risk of treatment-related mortality (TRM) and OS regardless of diagnosis, patient age, or conditioning regimen intensity,3,7 it does not consider disease type, disease stage, or disease phenotype, all of which have been shown to be powerful risk factors for relapse and mortality after transplantation.8-15 To determine which patients are likely to benefit from alloHCT, a unified comprehensive pretransplantation prognostic tool that accounts for all previously validated patient- and disease-related risk factors would be of direct clinical value. Despite the existence of several pretransplantation prognostic scoring systems, the predictive capacity of these tools varies.6,16-21 Moreover, existing scoring systems do not consider all components of the refined DRI and HCT-CI.2,5 Therefore, we developed a combined risk assessment using both the HCT-CI and disease-related factors (DRCI) to more precisely predict the clinical outcome of each patient being considered for alloHCT. In this report, we examine how the DRCI predicts clinical outcomes of adult patients undergoing alloHCT for hematological malignancies.
Methods
Study population
All consecutive adult (age ≥18 years) patients with hematological malignancies who received myeloablative or reduced-intensity conditioning (RIC) alloHCT between 2000 and 2013 were included in this study. Data were extracted from the University of Minnesota Blood and Marrow Transplant database and supplemented by review of individual medical records. HCT-CI, DRI, and Karnofsky performance score were assessed before alloHCT. Patients received peripheral blood or bone marrow from 8/8 HLA-matched sibling or unrelated volunteer donors or 4-6/6 HLA matched umbilical cord blood (UCB). Patients were excluded if the graft was autologous or from a haploidentical donor.
Myeloablative conditioning for a majority of adult donor recipients included cyclophosphamide and total-body irradiation (TBI) followed by busulfan and cyclophosphamide or a fludarabine-based regimen.22 Myeloablative conditioning for UCB recipients included fludarabine, cyclophosphamide, and TBI.23 Cyclophosphamide, fludarabine, and low-dose TBI was used for those undergoing RIC alloHCT regardless of donor type.24-27 Patients without immunosuppressive chemotherapy within 3 months of alloHCT or no prior autologous transplantation within 12 months of alloHCT also received equine antithymocyte globulin as part of their conditioning. Graft-versus-host disease (GVHD) prophylaxis after myeloablative conditioning generally consisted of calcineurin inhibitor and methotrexate for adult donor recipients and cyclosporine and mycophenolate mofetil (MMF) for UCB recipients.23,27 A majority of patients after RIC alloHCT received calcineurin inhibitor and MMF for GVHD prophylaxis, except those who underwent UCB transplantation after 2012 and received sirolimus and MMF.28 All transplantation treatment protocols were approved by the University of Minnesota Institutional Review Board, and all patients provided written consent before alloHCT.
Definitions and end points
The primary end point was OS at 2 years after alloHCT. Secondary end points included 2-year disease-free survival (DFS; defined as being alive with no evidence of disease relapse), TRM, incidence of relapse, grade 2 to 4 acute GVHD at day 180, 2-year chronic GVHD, and GVHD-free/relapse–free survival (GRFS). DRI was defined as reported by Armand et al2 and the Center for Blood and Marrow Transplant Research Acute Leukemia Working Committee. HCT-CI was defined as reported by Sorror et al.3,5 The intensity of conditioning was defined by using the Center for Blood and Marrow Transplant Research functional definitions.29 Consensus criteria and the Minnesota modification were used to grade acute and chronic GVHD.30-32 GRFS was defined as reported by Holtan et al.33
Statistical analysis
In this observational retrospective study, we examined the effect of the composite DRCI score on clinical outcomes of alloHCT. We report patient-, disease-, and transplantation-related characteristics descriptively. The Kaplan-Meier method was used to estimate OS and DFS probabilities, and the cumulative incidence estimator was used to calculate probabilities of TRM and relapse. Prognostic factors associated with OS were estimated using the Cox proportional hazards regression model. In this analysis, DRI was classified as low risk (LR) vs intermediate risk (IR) vs high or very high risk (HR/VHR), with the HR and VHR groups combined because of the small number of patients within the VHR group. HCT-CI was classified as a score of 0 to 2 vs ≥3, based on similar OS estimates observed in a univariable analysis for patients with HCT-CI scores of 0 to 2 vs ≥3. DRI (IR; hazard ratio, 1.81) and HR/VHR (hazard ratio, 2.89) were significantly different than LR (P < .01). Similarly, HCT-CI ≥3 (hazard ratio, 1.40) was significantly different than HCT-CI of 0 to 2 (P < .01). A multivariable OS model confirmed the significance of DRI and HCT-CI for OS. We then evaluated statistical interaction between DRI and HCT-CI, which was not significant (P = .93). After testing DRI and HCT-CI separately and in a multivariate model, a composite score of DRCI was then developed that considered both DRI and HCT-CI scores of patients at transplantation by dividing each DRI risk group into 2 subgroups based on HCT-CI score (0-2 vs ≥3), resulting in 6 subgroups: very LR (VLR) DRCI (LR DRI and 0-2 HCT-CI), LR DRCI (LR DRI and ≥3 HCT-CI), IR-1 risk DRCI (IR DRI and 0-2 HCT-CI), IR-2 DRCI (IR DRI and ≥3 HCT-CI), HR DRCI (HR DRI and 0-2 HCT-CI), and VHR DRCI (HR DRI and ≥3 HCT-CI). Patient-, disease-, and transplantation-related variables were considered in multivariate models using forward stepwise selection. The DRCI groups, regardless of level of significance, were included in all steps of model building (reference group, VLR). Adjusted OS and DFS probabilities were estimated using the Cox regression model. Because patients undergoing alloHCT with an HLA-matched adult donor might have had different survival outcomes as compared with those receiving UCB, we also performed the DRCI model selection analysis of OS separately for the adult donor cohort and UCB cohort. This separate analysis showed that the model is the same for each donor type and for the entire study population with donor types combined. Statistical analysis was computed using SAS software (version 9.3; SAS Institute, Cary, NC).
Results
Patient characteristics
We identified 959 adult patients with hematological malignancies who underwent alloHCT between 2000 and 2013 (Table 1). On the basis of DRCI risk stratification of patients at transplantation, 117 (12%) were VLR, 378 (39%) LR, 95 (10%) IR-1, 59 (6%) IR-2, 236 (25%) HR, and 74 (8%) VHR. Median age at transplantation was 50 years (range, 18-75 years). At the time of transplantation, 38% of patients had a high comorbidity score (≥3 HCT-CI), and 54% were cytomegalovirus (CMV) seropositive. The most frequent indications for alloHCT were leukemia (67%) and lymphoma (27%). DRI was either HR or VHR in ∼20% of the entire study cohort. AlloHCT used an adult donor (matched sibling donor, n = 419; matched unrelated donor, n = 54) in 46% and UCB in 54% of patients. RIC regimens (64%) were commonly used. Median follow-up of survivors was 4.9 years (range, 1.0-14.2 years).
Patient characteristics (N = 959)
| Variable . | n (%) . |
|---|---|
| DRCI | |
| VLR | 117 (12) |
| LR | 59 (6) |
| IR-1 | 378 (39) |
| IR-2 | 236 (25) |
| HR | 95 (10) |
| VHR | 74 (8) |
| DRI | |
| LR | 176 (18) |
| IR | 614 (64) |
| HR/VHR | 169 (18) |
| HCT-CI | |
| 0 | 297 (31) |
| 1-2 | 293 (31) |
| ≥3 | 369 (38) |
| Age, y | |
| Median (range) | 50 (18-75) |
| 18-49 | 485 (51) |
| ≥50 | 474 (49) |
| Sex | |
| Male | 567 (59) |
| Female | 392 (41) |
| Diagnosis | |
| Leukemia | 636 (66) |
| Lymphoma | 258 (27) |
| Other | 65 (7) |
| Donor type | |
| Adult donor | 444 (46) |
| UCB | 515 (54) |
| Conditioning | |
| Myeloablative | 344 (36) |
| RIC | 615 (64) |
| CMV serostatus | |
| Positive | 519 (54) |
| Negative | 438 (46) |
| Missing | 2 (0) |
| Year of HCT | |
| 2000-2006 | 442 (46) |
| 2007-2013 | 517 (54) |
| Median (range) follow-up of 425 survivors | 4.9 (1.0-14.2) |
| Variable . | n (%) . |
|---|---|
| DRCI | |
| VLR | 117 (12) |
| LR | 59 (6) |
| IR-1 | 378 (39) |
| IR-2 | 236 (25) |
| HR | 95 (10) |
| VHR | 74 (8) |
| DRI | |
| LR | 176 (18) |
| IR | 614 (64) |
| HR/VHR | 169 (18) |
| HCT-CI | |
| 0 | 297 (31) |
| 1-2 | 293 (31) |
| ≥3 | 369 (38) |
| Age, y | |
| Median (range) | 50 (18-75) |
| 18-49 | 485 (51) |
| ≥50 | 474 (49) |
| Sex | |
| Male | 567 (59) |
| Female | 392 (41) |
| Diagnosis | |
| Leukemia | 636 (66) |
| Lymphoma | 258 (27) |
| Other | 65 (7) |
| Donor type | |
| Adult donor | 444 (46) |
| UCB | 515 (54) |
| Conditioning | |
| Myeloablative | 344 (36) |
| RIC | 615 (64) |
| CMV serostatus | |
| Positive | 519 (54) |
| Negative | 438 (46) |
| Missing | 2 (0) |
| Year of HCT | |
| 2000-2006 | 442 (46) |
| 2007-2013 | 517 (54) |
| Median (range) follow-up of 425 survivors | 4.9 (1.0-14.2) |
OS
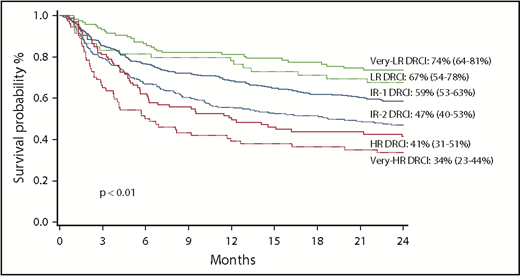
The probability of OS at 2 years for the entire cohort was 54% (95% confidence interval [CI], 51%-58%). For DRCI groups, OS at 2 years was 74% for VLR, 67% for LR, 59% for IR-1, 47% for IR-2, 41% for HR, and 34% for VHR (P < .01; Figure 1). In multiple regression analysis, after adjusting for patient age and donor type, DRCI was identified as the strongest independent predictor of OS among patients undergoing alloHCT (Table 2). None of the other factors examined, including patient age, donor type, and conditioning intensity, had a significant effect on OS.
DRCI predicts 2-y OS
| Variable . | Total n . | Multivariable analysis . | ||
|---|---|---|---|---|
| Hazard ratio* . | 95% CI . | P . | ||
| All patients (N = 959) | ||||
| DRCI | <.01 | |||
| VLR | 117 | 1.0 | ||
| LR | 59 | 1.28 | 0.72-2.28 | |
| IR-1 | 378 | 1.69 | 1.14-2.51 | |
| IR-2 | 236 | 2.40 | 1.61-3.59 | |
| HR | 95 | 2.82 | 1.80-4.40 | |
| VHR | 74 | 3.85 | 2.44-6.07 | |
| Donor type | .06 | |||
| Adult donor | 444 | 1.0 | ||
| UCB | 515 | 1.20 | 0.99-1.46 | |
| Age, y | .36 | |||
| 18-49 | 485 | 1.0 | ||
| ≥50 | 474 | 1.09 | 0.90-1.32 | |
| Adult donor cohort (n = 444) | ||||
| DRCI | <.01 | |||
| VLR | 69 | 1.0 | ||
| LR | 27 | 1.71 | 0.74-3.96 | |
| IR-1 | 173 | 2.08 | 1.17-3.71 | |
| IR-2 | 95 | 3.20 | 1.76-5.81 | |
| HR | 43 | 3.33 | 1.71-6.48 | |
| VHR | 37 | 4.85 | 2.51-9.40 | |
| Age, y | .27 | |||
| 18-49 | 201 | 1.0 | ||
| ≥50 | 243 | 1.18 | 0.88-1.59 | |
| UCB cohort (n = 515) | ||||
| DRCI | <.01 | |||
| VLR | 48 | 1.0 | ||
| LR | 32 | 0.97 | 0.44-2.15 | |
| IR-1 | 205 | 1.37 | 0.80-2.34 | |
| IR-2 | 141 | 1.83 | 1.07-3.15 | |
| HR | 52 | 2.33 | 1.28-4.25 | |
| VHR | 37 | 3.05 | 1.63-5.73 | |
| Age, y | .81 | |||
| 18-49 | 284 | 1.0 | ||
| ≥50 | 231 | 1.03 | 0.80-1.33 | |
| Variable . | Total n . | Multivariable analysis . | ||
|---|---|---|---|---|
| Hazard ratio* . | 95% CI . | P . | ||
| All patients (N = 959) | ||||
| DRCI | <.01 | |||
| VLR | 117 | 1.0 | ||
| LR | 59 | 1.28 | 0.72-2.28 | |
| IR-1 | 378 | 1.69 | 1.14-2.51 | |
| IR-2 | 236 | 2.40 | 1.61-3.59 | |
| HR | 95 | 2.82 | 1.80-4.40 | |
| VHR | 74 | 3.85 | 2.44-6.07 | |
| Donor type | .06 | |||
| Adult donor | 444 | 1.0 | ||
| UCB | 515 | 1.20 | 0.99-1.46 | |
| Age, y | .36 | |||
| 18-49 | 485 | 1.0 | ||
| ≥50 | 474 | 1.09 | 0.90-1.32 | |
| Adult donor cohort (n = 444) | ||||
| DRCI | <.01 | |||
| VLR | 69 | 1.0 | ||
| LR | 27 | 1.71 | 0.74-3.96 | |
| IR-1 | 173 | 2.08 | 1.17-3.71 | |
| IR-2 | 95 | 3.20 | 1.76-5.81 | |
| HR | 43 | 3.33 | 1.71-6.48 | |
| VHR | 37 | 4.85 | 2.51-9.40 | |
| Age, y | .27 | |||
| 18-49 | 201 | 1.0 | ||
| ≥50 | 243 | 1.18 | 0.88-1.59 | |
| UCB cohort (n = 515) | ||||
| DRCI | <.01 | |||
| VLR | 48 | 1.0 | ||
| LR | 32 | 0.97 | 0.44-2.15 | |
| IR-1 | 205 | 1.37 | 0.80-2.34 | |
| IR-2 | 141 | 1.83 | 1.07-3.15 | |
| HR | 52 | 2.33 | 1.28-4.25 | |
| VHR | 37 | 3.05 | 1.63-5.73 | |
| Age, y | .81 | |||
| 18-49 | 284 | 1.0 | ||
| ≥50 | 231 | 1.03 | 0.80-1.33 | |
Hazard ratio denotes increased risk of mortality.
The effect of DRCI on OS was studied separately in adult donor and UCB cohorts. The probability of OS at 2 years for the adult donor cohort was 58% (95% CI, 53%-62%). OS at 2 years after adult donor transplantation for DRCI groups was 79% for VLR, 65% for LR, 62% for IR-1, 48% for IR-2, 45% for HR, and 35% for VHR (P < .01), similar to rates for the entire cohort. In multiple regression analysis, even after adjusting for patient age, DRCI remained strongly predictive of OS within the adult donor cohort.
The probability of OS at 2 years for the UCB cohort was 52% (95% CI, 47%-56%). OS at 2 years for DRCI groups within the UCB cohort was 66% for VLR, 69% for LR, 56% for IR-1, 46% for IR-2, 38% for HR, and 32% for VHR (P < .01). Again, after considering adjustment for patient age, in multiple regression analysis DRCI groups were associated with OS within the UCB cohort as well.
DFS
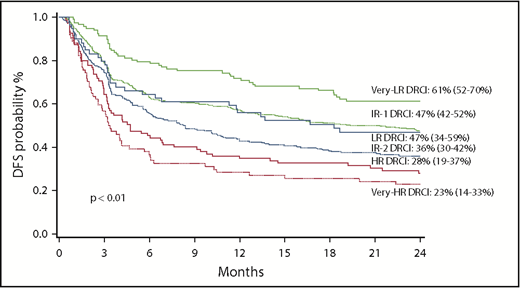
The probability of DFS at 2 years for all patients was 42% (95% CI, 39%-45%) for the entire cohort. For DRCI groups, DFS at 2 years was 61% for VLR, 47% for LR, 47% for IR-1, 36% for IR-2, 28% for HR, and 23% for VHR (P < .01; Figure 2). In multiple regression analysis, after considering adjustment for diagnosis and conditioning intensity, DRCI remained strongly predictive of DFS after alloHCT (Table 3). DFS was significantly worse for patients receiving RIC (hazard ratio, 1.30; 95% CI, 1.05-1.61; P = .01) versus myeloablative conditioning while patient age did not influence DFS.
Clinical outcomes of all patients by DRCI
| Variable . | Total n . | Multivariable analysis . | ||
|---|---|---|---|---|
| Hazard ratio* . | 95% CI . | P . | ||
| DFS at 2 y | ||||
| DRCI | <.01 | |||
| VLR | 117 | 1.0 | ||
| LR | 59 | 1.57 | 0.99-2.49 | |
| IR-1 | 378 | 1.64 | 1.18-2.27 | |
| IR-2 | 236 | 2.19 | 1.56-3.06 | |
| HR | 95 | 2.88 | 1.97-4.21 | |
| VHR | 74 | 3.42 | 2.31-5.08 | |
| Conditioning intensity | .01 | |||
| Myeloablative | 344 | 1.0 | ||
| RIC | 615 | 1.30 | 1.05-1.61 | |
| Age, y | .52 | |||
| 18-49 | 485 | 1.0 | ||
| ≥50 | 474 | 0.94 | 0.77-1.14 | |
| TRM at 2 y | ||||
| DRCI | ||||
| VLR | 117 | 1.0 | ||
| LR | 59 | 1.51 | 0.75-3.04 | .37 |
| IR-1 | 378 | 1.27 | 0.76-2.12 | .29 |
| IR-2 | 236 | 1.66 | 0.98-2.82 | .25 |
| HR | 95 | 1.38 | 0.76-2.52 | .06 |
| VHR | 74 | 2.70 | 1.47-4.94 | <.01 |
| Conditioning intensity | <.01 | |||
| Myeloablative | 344 | 1.0 | ||
| RIC | 615 | 0.65 | 0.49-0.86 | |
| CMV serostatus | .01 | |||
| Negative | 438 | 1.0 | ||
| Positive | 519 | 1.40 | 1.08-1.83 | |
| Relapse at 2 y | ||||
| DRCI | ||||
| VLR | 117 | 1.0 | ||
| LR | 59 | 1.42 | 0.73-2.77 | .31 |
| IR-1 | 378 | 1.72 | 1.03-2.87 | .04 |
| IR-2 | 236 | 2.17 | 1.29-3.67 | <.01 |
| HR | 95 | 3.92 | 2.23-6.91 | <.01 |
| VHR | 74 | 2.53 | 1.35-4.75 | <.01 |
| Conditioning intensity | <.01 | |||
| Myeloablative | 344 | 1.0 | ||
| RIC | 615 | 1.96 | 1.48-2.60 | |
| Variable . | Total n . | Multivariable analysis . | ||
|---|---|---|---|---|
| Hazard ratio* . | 95% CI . | P . | ||
| DFS at 2 y | ||||
| DRCI | <.01 | |||
| VLR | 117 | 1.0 | ||
| LR | 59 | 1.57 | 0.99-2.49 | |
| IR-1 | 378 | 1.64 | 1.18-2.27 | |
| IR-2 | 236 | 2.19 | 1.56-3.06 | |
| HR | 95 | 2.88 | 1.97-4.21 | |
| VHR | 74 | 3.42 | 2.31-5.08 | |
| Conditioning intensity | .01 | |||
| Myeloablative | 344 | 1.0 | ||
| RIC | 615 | 1.30 | 1.05-1.61 | |
| Age, y | .52 | |||
| 18-49 | 485 | 1.0 | ||
| ≥50 | 474 | 0.94 | 0.77-1.14 | |
| TRM at 2 y | ||||
| DRCI | ||||
| VLR | 117 | 1.0 | ||
| LR | 59 | 1.51 | 0.75-3.04 | .37 |
| IR-1 | 378 | 1.27 | 0.76-2.12 | .29 |
| IR-2 | 236 | 1.66 | 0.98-2.82 | .25 |
| HR | 95 | 1.38 | 0.76-2.52 | .06 |
| VHR | 74 | 2.70 | 1.47-4.94 | <.01 |
| Conditioning intensity | <.01 | |||
| Myeloablative | 344 | 1.0 | ||
| RIC | 615 | 0.65 | 0.49-0.86 | |
| CMV serostatus | .01 | |||
| Negative | 438 | 1.0 | ||
| Positive | 519 | 1.40 | 1.08-1.83 | |
| Relapse at 2 y | ||||
| DRCI | ||||
| VLR | 117 | 1.0 | ||
| LR | 59 | 1.42 | 0.73-2.77 | .31 |
| IR-1 | 378 | 1.72 | 1.03-2.87 | .04 |
| IR-2 | 236 | 2.17 | 1.29-3.67 | <.01 |
| HR | 95 | 3.92 | 2.23-6.91 | <.01 |
| VHR | 74 | 2.53 | 1.35-4.75 | <.01 |
| Conditioning intensity | <.01 | |||
| Myeloablative | 344 | 1.0 | ||
| RIC | 615 | 1.96 | 1.48-2.60 | |
Hazard ratio denotes increased risk of mortality.
TRM and relapse
The cumulative incidence of TRM at 2 years was 26% (95% CI, 23%-29%) for the entire cohort. For DRCI groups, TRM was 18% for VLR, 23% for LR, 24% for IR-1, 29% for IR-2, 24% for HR, and 40% for VHR (P < .01). In multiple regression analysis, after adjusting for conditioning intensity and recipient CMV serostatus, VHR DRCI resulted in a 2.7-fold greater TRM as compared with VLR DRCI (hazard ratio, 2.70; 95% CI, 1.47-4.94; P < .01). Although higher, the risks of TRM were not individually significant for each DRCI group. TRM was significantly higher for patients receiving myeloablative conditioning (hazard ratio, 0.65; 95% CI, 0.49-0.86 vs RIC; P < .01) and for CMV seropositive patients (hazard ratio, 1.40; 95% CI, 1.08-1.83 vs CMV seronegative; P = .01).
The cumulative incidence of relapse at 2 years was 30% (95% CI, 27%-33%) for all study patients. For DRCI groups, the incidence of relapse was 17% for VLR, 26% for LR, 27% for IR-1, 33% for IR-2, 45% for HR, and 36% for VHR (P < .01). In multiple regression analysis, after adjusting for conditioning intensity, the risk of relapse increased with DRCI score compared with the VLR group. The risk of relapse was also significantly higher for patients undergoing RIC alloHCT (hazard ratio, 1.96; 95% CI, 1.48-2.60; P < .01) vs myeloablative conditioning.
GVHD and GRFS
For the entire cohort, the cumulative incidence of grade 2 to 4 acute GVHD at day 180 was 50% (95% CI, 47%-54%); the cumulative incidence of chronic GVHD at 2 years was 34% (95% CI, 30%-37%), but DRCI was not associated with risk of GVHD. For DRCI groups, grade 2 to 4 acute GVHD incidence was 49% for VLR, 44% for LR, 53% for IR-1, 49% for IR-2, 47% for HR, and 50% for VHR (P = .78). The incidence for chronic GVHD was 39% for VLR, 35% for LR, 36% for IR-1, 37% for IR-2, 27% for HR, and 29% for VHR (P = .32). In multiple regression analysis, DRCI had no significant impact on risk of grade 2 to 4 acute GVHD or chronic GVHD. After UCB donor alloHCT, grade 2 to 4 acute GVHD was more frequent (hazard ratio, 1.33; 95% CI, 1.10-1.60; P < .01), although grade 3 to 4 acute GVHD was not (hazard ratio, 0.93, 95% CI, 0.70-1.24; P = .64). However, UCB alloHCT was associated with a significantly lower risk of chronic GVHD (hazard ratio, 0.47; 95% CI, 0.37-0.59; P < .01) vs adult donor HCT. AlloHCT performed after 2006 was associated with lower risk of both grade 2 to 4 acute GVHD (hazard ratio, 0.82; 95% CI, 0.68-0.99; P = .04) and chronic GVHD (hazard ratio, 0.57; 95% CI, 0.45-0.72; P < .01). Women had lower risk of chronic GVHD (hazard ratio, 0.78; 95% CI, 0.62-0.99; P = .04), whereas the risk of acute or chronic GVHD was not significantly influenced by patient age or conditioning intensity.
The 2-year probability of the composite end point GRFS was 20% (95% CI, 18%-23%) for the entire cohort. For DRCI groups, the incidence of GRFS was 29% for VLR, 22% for LR, 24% for IR-1, 15% for IR-2, 16% for HR, and 11% for VHR (P < .01). In multiple regression analysis, after adjusting for patient age, donor type, and year of HCT, DRCI significantly influenced GRFS. In addition, GRFS was significantly better for those receiving a UCB graft (hazard ratio, 0.81; 95% CI, 0.70-0.94; P < .01) or undergoing HCT after 2006 (hazard ratio, 0.84; 95% CI, 0.72-0.98; P = .03), whereas patient age had no significant effect on GRFS.
Discussion
In this study, we introduced the novel DRCI pretransplantation prognostic scoring system, which integrates 2 independent and previously validated pretransplantation prognostic tools, the refined DRI and HCT-CI, and stratifies patients into 6 significantly different risk groups. We found that DRCI was the strongest prognostic factor for OS among adult patients undergoing alloHCT for hematological malignancies, independent of age, donor type, and conditioning intensity. DRCI also retained prognostic significance for OS in both adult donor and UCB cohorts when analyzed separately.
In addition to OS, DRCI predicted DFS, relapse, and GRFS after alloHCT. DRCI provides a comprehensive pretransplantation prognostic scoring system, accounting for all of the components of the refined DRI (including disease type, disease status, and cytogenetic risk of acute myeloid leukemia [AML]/myelodysplastic syndrome [MDS]) and all 17 components of patient comorbidities incorporated into HCT-CI. Our clinically important observation indicates that DRCI can serve as an easily available prognostic tool to evaluate patients eligible for alloHCT and stratify patients participating in alloHCT clinical trials.
Other previously studied pretransplantation prognostic scoring systems include the European Group for Blood and Marrow Transplantation (EBMT) risk score34 and the pretransplantation assessment of mortality (PAM) score.35 The EBMT risk score was originally developed to predict the post-HCT outcomes of chronic myeloid leukemia patients, but it was later shown to be prognostic for survival of those undergoing alloHCT with various hematological diagnoses.34 In contrast to DRCI, the EBMT score was developed largely from earlier myeloablative conditioning (85%) alloHCT experiences (1980 to 2005), and it considered only 5 factors (patient age, disease stage, time from diagnosis to transplantation, donor type, and donor-recipient sex combination).34 Subsequent studies expanded on the initial EBMT model by incorporating additional risk factors to achieve better prediction of survival outcome after alloHCT, particularly for the assessment of specific disease groups (eg, acute leukemia EBMT score).36,37 However, genetic data such as cytogenetics in AML, which are included in DRCI and have been consistently demonstrated to be a powerful predictor of post-HCT survival, have not yet been considered in existing predictive models.2,8,15,37
PAM was originally developed in 2006 by the Seattle group for prediction of 2-year OS after alloHCT in patients with hematological diseases.35 Similar to the EBMT risk score, PAM was also developed mainly based on myeloablative conditioning alloHCT experiences (6% patients received RIC), and it incorporated only 8 factors (patient age, disease risk, donor type, conditioning regimen, forced expiratory volume, carbon monoxide diffusing capacity, serum alanine aminotransferase value, and serum creatinine concentration).35 In contrast to DRCI, the PAM scoring system did not account for other comorbid conditions included in HCT-CI or the cytogenetic risk in AML and MDS in disease risk assessment. In addition, the Seattle group recently reported that the performance of the original PAM score was diminishing over time and was less reliable after RIC.19 Additionally, the liver and kidney organ assessment parameters of the original PAM were no longer predictive for OS.19 The value of the original HCT-CI components has been recently validated in a multicenter prospective study,5,17 and their inclusion in DRCI reflects its coordinate value in integrating survival predictions with disease status. DRCI was valuable in both UCB and adult donor cohorts, and donor type had no independently significant effect on OS.
Our institutional study cohort included only a modest number of patients with VHR DRI, and therefore, they were analyzed along with the HR DRI group. A larger patient cohort, such as a large registry-based study, will be valuable for validation of the DRCI pretransplantation prognostic scoring system. A similar model has recently been applied in AML induction therapy.38
In conclusion, our data demonstrate that DRCI is a comprehensive and easily applied pretransplantation prognostic scoring system that can serve as a reliable predictor of OS after alloHCT. It can provide transplantation physicians and patients the tools to better predict who will best benefit from alloHCT.
Acknowledgments
The authors acknowledge Michael Franklin for assistance in editing this manuscript.
This work was supported in part by National Institutes of Health, National Cancer Institute grant P01 CA65493 (C.G.B. and J.E.W.) and Leukemia and Lymphoma Society Scholar in Clinical Research Award R6029-07 (C.G.B.).
Authorship
Contribution: N.B., C.G.B., and D.J.W. conceived the study; Q.C. analyzed and interpreted data; N.B. wrote the manuscript; and all authors interpreted and edited the manuscript and approved the final manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Nelli Bejanyan, Department of Blood and Marrow Transplant and Cellular Immunotherapy, Moffitt Cancer Center, 12902 Magnolia Dr, Tampa, FL 33612; e-mail: nelli.bejanyan@moffitt.org.
References
Author notes
N.B. and C.G.B. contributed equally to this work.