Key Points
Lung function parameters as CO-diffusion abnormalities influences outcome in patients undergoing auto-HSCT.
Several clinical factors, including smoking, are associated with decreased CO diffusion prior to auto-HSCT.
Abstract
High-dose chemotherapy, followed by autologous hematopoietic stem cell transplantation (auto-HSCT), is an established therapy for patients with hematological malignancies. The age of patients undergoing auto-HSCT and, therefore, the comorbidities, has increased over the last decades. However, the assessment of organ dysfunction prior to auto-HSCT has not been well studied. Therefore, we retrospectively analyzed the association of clinical factors and lung and cardiac function with outcome and complications after conditioning with BEAM (BCNU/carmustine, etoposide, cytarabine, melphalan) or high-dose melphalan in patients undergoing auto-HSCT. This study included 629 patients treated at our institution between 2007 and 2017; 334 and 295 were conditioned with BEAM or high-dose melphalan, respectively. The median follow-up was 52 months (range, 0.2-152) and 50 months (range, 0.5-149), respectively. In the multivariate analysis, we identified that progressive disease, CO-diffusion capacity corrected for hemoglobin (DLCOcSB) ≤ 60% of predicted, Karnofsky Performance Status (KPS) ≤ 80%, Hematopoietic Cell Transplantation Comorbidity Index (HCT-CI) score ≥ 4, and age > 70 years were associated with decreased overall survival (OS) in patients treated with BEAM. Similarly, DLCOcSB ≤ 60% of predicted, HCT-CI score ≥ 4, and age > 60 years were identified in patients treated with high-dose melphalan. Abnormalities in DLCOcSB ≤ 60% of predicted were associated with chemotherapy with lung-toxic substances, mediastinal radiotherapy, KPS ≤ 80%, current/previous smoking, and treatment in the intensive care unit. More often, patients with DLCOcSB ≤ 60% of predicted experienced nonrelapse mortality, including pulmonary causes of death. In summary, we identified DLCOcSB ≤ 60% of predicted as an independent risk factor for decreased OS in patients conditioned with BEAM or high-dose melphalan prior to auto-HSCT.
Introduction
High-dose chemotherapy, followed by autologous hematopoietic stem cell transplantation (auto-HSCT), is an established therapy for patients with hematological malignancies.1 The age and, therefore, the comorbidities of patients undergoing auto-HSCT have increased during the last decades.2 Comorbidities and age were reported to impact the prognosis of patients with lymphoma undergoing auto-HSCT.3,4 However, the assessment of organ dysfunction prior to auto-HSCT has not been well studied in an effort to predict complications and outcome after auto-HSCT.
The BEAM protocol (BCNU/carmustine, etoposide, cytarabine, and melphalan) is the most common conditioning regimen that is used to treat patients with Hodgkin or non-Hodgkin lymphoma prior to auto-HSCT.5,6 However, it may be associated with major pulmonary toxicity, as a result of the high dose of BCNU, and induce early treatment-related complications.7,8 High-dose melphalan is used as conditioning prior to auto-HSCT in patients with multiple myeloma.9-11 It is well tolerated by patients; mucositis, infections, and diarrhea are the most common side effects.12,13 Higher comorbidity indexes and lower performance scores were reported to be associated with worse outcome in patients with multiple myeloma undergoing auto-HSCT.14,15 Very few studies have systematically addressed how lung function affects outcomes and complications after auto-HSCT in patients conditioned with BEAM or high-dose melphalan. Nevertheless, lung function was identified as a highly relevant factor for patients with myeloma in the Revised Myeloma Comorbidity Index (R-MCI).16-18
Therefore, we analyzed the outcome, clinical factors, and early organ toxicity associated with lung and cardiac function parameters of patients undergoing auto-HSCT after conditioning with BEAM or high-dose melphalan at our institution between 2007 and 2017. We analyzed both cohorts separately and identified specific risk factors, including lung function abnormalities, that were associated with unfavorable outcome in patients undergoing auto-HSCT with each conditioning protocol, which might lead to preventive or early therapeutic interventions.
Material and methods
Patient selection and characteristics
We included 629 patients treated with auto-HSCT (334 patients conditioned with BEAM and 295 patients conditioned with high-dose melphalan) at our institution between 1 January 2007 and 31 December 2017. We included all patients undergoing auto-HSCT. Clinical data were prospectively collected and retrospectively analyzed each cohort separately. Patient characteristics, as well as lung and cardiac function parameters, are described in Table 1. Comorbidities, including the Hematopoietic Cell Transplantation Comorbidity Index (HCT-CI),19,20 and early complications after auto-HSCT, defined as complications after auto-HSCT and before discharge, are described in supplemental Table 1. Conditioning regimen–related early toxicity by organ system, after auto-HSCT and before discharge, was assessed as previously described.21 Causes of death were adapted from previously described criteria.22 This study was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from the subjects for the use of their data in clinical research, and the Institutional Review Board of the University of Freiburg Medical Center approved the study protocol. All data were evaluated as of 31st May 2020, and all authors had access to primary clinical trial data.
Clinical characteristics and lung and cardiac function parameters of patients conditioned with BEAM or high-dose melphalan prior to auto-HSCT
| Variables . | BEAM (n = 334) . | MEL (n = 295) . |
|---|---|---|
| Patient characteristics | ||
| Age at diagnosis, median (range), y | 54.3 (17-78) | 57.2 (29-74) |
| Age at auto-HSCT, median (range), y | 56.3 (19-78) | 58.6 (29-74) |
| Females | 128 (38) | 120 (41) |
| KPS ≤ 80% | 89 (28) | 109 (39) |
| Pulmonary function tests prior to auto-HSCT | 320 (96) | 281 (95) |
| FEV1, % of predicted, median (range) | 91 (34-145) | 89 (35, 136) |
| FEV1/FVC ratio, median (range) | 0.80 (0.45-0.97) | 0.79 (0.42, 1.15) |
| MEF50, % of predicted, median (range) | 67 (11-173) | 62 (10, 165) |
| MEF25, % of predicted, median (range) | 38 (4-193) | 34 (5, 165) |
| DLCOcSB, % of predicted, median (range) | 77 (29-126) | 79 (35, 116) |
| RV, % of predicted, median (range) | 105 (36-249) | 104 (31-273) |
| RV/TLC ratio, median (range) | 0.35 (0.15-1.16) | 0.36 (0.15-0.69) |
| TLC, % of predicted, median (range) | 93 (54-134) | 93 (41-132) |
| Arterial CO2 mm Hg absolute, median (range) | 36 (25-52) | 36 (25-46) |
| Arterial O2 mm Hg absolute, median (range) | 81 (55-102) | 78 (56-97) |
| Transthoracic heart echography | 316 (95) | 281 (95) |
| EF 60-65% | 76 (23) | 62 (21) |
| EF 55-59% | 194 (58) | 184 (62) |
| EF 50-54% | 28 (8) | 21 (7) |
| EF ≤ 49% | 18 (5) | 14 (5) |
| Disease characteristics | ||
| Disease | ||
| B-cell NHL | 222 (68) | |
| T-cell NHL | 46 (13) | |
| Hodgkin lymphoma | 34 (9) | |
| MM | 8 (3) | 295 (100) |
| ALL | 4 (1) | |
| Burkitt lymphoma | 16 (5) | |
| B symptoms at diagnosis | 118 (34.8) | |
| B-stage at diagnosis as Durie-Salmon criteria | — | 37 (14) |
| Progressive disease at auto-HSCT | 28 (8) | 18 (6) |
| Auto-HSCT characteristics, median (range) | ||
| Days on the ward after auto-HSCT | 14 (9-110) | 15 (10-67) |
| CD34+ cells transplanted (×106 per kg bw) | 6.0 (0.15-55.6) | 5.2 (2.14-27.2) |
| Day of engraftment | ||
| Neutrophils | 10 (0-29) | 11 (2-53) |
| Thrombocytes | 11 (0-52) | 11 (1-80) |
| Treatments post auto-HSCT | ||
| Chemotherapy post auto-HSCT | 77 (23) | 170 (58) |
| Radiotherapy post auto-HSCT | 43 (13) | 90 (30) |
| Second auto-HSCT | 12 (4) | 102 (35) |
| Allo-HSCT after auto-HSCT | 41 (12) | 58 (20) |
| Follow-up, median (range), mo | 52 (0.2-153) | 50 (0.6-149) |
| Variables . | BEAM (n = 334) . | MEL (n = 295) . |
|---|---|---|
| Patient characteristics | ||
| Age at diagnosis, median (range), y | 54.3 (17-78) | 57.2 (29-74) |
| Age at auto-HSCT, median (range), y | 56.3 (19-78) | 58.6 (29-74) |
| Females | 128 (38) | 120 (41) |
| KPS ≤ 80% | 89 (28) | 109 (39) |
| Pulmonary function tests prior to auto-HSCT | 320 (96) | 281 (95) |
| FEV1, % of predicted, median (range) | 91 (34-145) | 89 (35, 136) |
| FEV1/FVC ratio, median (range) | 0.80 (0.45-0.97) | 0.79 (0.42, 1.15) |
| MEF50, % of predicted, median (range) | 67 (11-173) | 62 (10, 165) |
| MEF25, % of predicted, median (range) | 38 (4-193) | 34 (5, 165) |
| DLCOcSB, % of predicted, median (range) | 77 (29-126) | 79 (35, 116) |
| RV, % of predicted, median (range) | 105 (36-249) | 104 (31-273) |
| RV/TLC ratio, median (range) | 0.35 (0.15-1.16) | 0.36 (0.15-0.69) |
| TLC, % of predicted, median (range) | 93 (54-134) | 93 (41-132) |
| Arterial CO2 mm Hg absolute, median (range) | 36 (25-52) | 36 (25-46) |
| Arterial O2 mm Hg absolute, median (range) | 81 (55-102) | 78 (56-97) |
| Transthoracic heart echography | 316 (95) | 281 (95) |
| EF 60-65% | 76 (23) | 62 (21) |
| EF 55-59% | 194 (58) | 184 (62) |
| EF 50-54% | 28 (8) | 21 (7) |
| EF ≤ 49% | 18 (5) | 14 (5) |
| Disease characteristics | ||
| Disease | ||
| B-cell NHL | 222 (68) | |
| T-cell NHL | 46 (13) | |
| Hodgkin lymphoma | 34 (9) | |
| MM | 8 (3) | 295 (100) |
| ALL | 4 (1) | |
| Burkitt lymphoma | 16 (5) | |
| B symptoms at diagnosis | 118 (34.8) | |
| B-stage at diagnosis as Durie-Salmon criteria | — | 37 (14) |
| Progressive disease at auto-HSCT | 28 (8) | 18 (6) |
| Auto-HSCT characteristics, median (range) | ||
| Days on the ward after auto-HSCT | 14 (9-110) | 15 (10-67) |
| CD34+ cells transplanted (×106 per kg bw) | 6.0 (0.15-55.6) | 5.2 (2.14-27.2) |
| Day of engraftment | ||
| Neutrophils | 10 (0-29) | 11 (2-53) |
| Thrombocytes | 11 (0-52) | 11 (1-80) |
| Treatments post auto-HSCT | ||
| Chemotherapy post auto-HSCT | 77 (23) | 170 (58) |
| Radiotherapy post auto-HSCT | 43 (13) | 90 (30) |
| Second auto-HSCT | 12 (4) | 102 (35) |
| Allo-HSCT after auto-HSCT | 41 (12) | 58 (20) |
| Follow-up, median (range), mo | 52 (0.2-153) | 50 (0.6-149) |
Unless other noted, data are n (%).
ALL, acute lymphoblastic leukemia; allo-HSCT, allogeneic hematopoietic stem cell transplantation; bw, body weight; EF, ejection fraction; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; MEF25, mid-expiratory flow 25% of vital capacity; MEF50, mid-expiratory flow 50% of vital capacity; MEL, high-dose melphalan; MM, multiple myeloma; NHL, non-Hodgkin lymphoma; RV, residual volume; TLC, total lung capacity.
Conditioning protocol
Patients received conditioning parenterally via a central venous catheter according to the BEAM protocol (BCNU/carmustine, 300 mg/m2 at day −7; cytarabine, 2 × 200 mg/m2 per day from day −6 to day −3; etoposide, 2 × 100 mg/m2 per day from day −6 to day −3; and melphalan, 140 mg/m2 at day −2) or the high-dose melphalan protocol (melphalan, 100 mg/m2 at days −3 and −2). Chemotherapy- and granulocyte colony-stimulating factor–mobilized peripheral blood stem cell grafts were used in all cases.
Pulmonary function tests
Pulmonary function tests (PFTs), including diffusion capacity for CO, were performed according to international standards23,24 a week before auto-HSCT. PFT parameters were evaluated and expressed as a percentage of predicted normal values, calculated using published equations25 that were based on a population (white, central Europe) very similar to the patients included in this study.
Study end points, definitions, and statistical analysis
Statistical analyses of patient characteristics, PFTs, cumulative incidences, and hazard ratios (HRs) were calculated using STATA v11.0 (College Station, TX). Overall survival (OS) was defined as time from auto-HSCT until death from any cause, and progression-free survival (PFS) was calculated as time from auto-HSCT until death from any cause or relapse, whichever occurred first. For those patients not experiencing the event in question for OS or PFS during follow-up, time to last contact was used as a censored observation. Relapse was defined as detection of disease via cytological, histological, or radiological assessment after auto-HSCT; death without prior relapse was considered a competing risk and denoted as nonrelapse mortality (NRM).
The Cox proportional-hazards regression model was used to estimate HRs with corresponding confidence intervals (CIs) for OS and PFS. We applied the Fine and Gray model to compare cumulative incidence rates in the presence of competing risks26,27 and present subdistribution hazard ratios (SHRs) for relapse incidence and NRM.
Multivariate analyses for OS were conducted using a Cox proportional-hazards regression model and a backward-selection strategy. Covariates, including clinical characteristics, pulmonary and cardiac function tests values with a univariate P value < 0.1 were initially included. HR and 2-sided 95% CI of prognostic factors for OS were estimated.
Pearson’s χ2 test and Fisher’s exact test were used to compare categorical variables as appropriate,28 and the Student t test was used to compare continuous variables, assuming a normal distribution.
Results
Clinical features of patients treated with BEAM conditioning protocols prior to auto-HSCT
Between 1st January 2007 and 31st December 2017, 629 patients (334 patients after conditioning with BEAM and 295 patients after conditioning with high-dose melphalan) underwent auto-HSCT at the University of Freiburg Medical Center. The median age was 56 years (range, 19-78) and 59 years (range, 29-74) in the BEAM and high-dose melphalan groups, respectively. One hundred and twenty-eight (38%) patients treated with BEAM and 120 (41%) patients treated with high-dose melphalan were female. The Karnofsky Performance Score (KPS) was ≤ 80% in 89 (28%) patients treated with BEAM and in 109 (39%) patients treated with high-dose melphalan. Before auto-HSCT, patients had several comorbidities; arterial hypertension and gastroenterologic, nephrologic, and cardiac diseases were among the most frequent. Notably, 33 patients (10.5%) in the BEAM group and 95 (32%) patients in the high-dose melphalan group had a history of pulmonary diseases, including asthma, chronic obstructive pulmonary disease, pneumothorax, obstructive sleep apnea syndrome, pneumonia, pleural effusions, lung surgery, or pulmonary congestion. Patients had a median HCT-CI score19 of 3 (range, 0-11) in the BEAM group and a score of 2 (range, 0-13) in the high-dose melphalan group (supplemental Table 1A). The majority of patients had an available lung function test prior to auto-HSCT (n = 601; 95.5%), CO diffusion capacity corrected for hemoglobin (DLCOcSB; n = 592; 94.1%), and transthoracic heart echography (n = 597; 94.9%) (Table 1).
In patients conditioned with BEAM, the main indication for auto-HSCT was B-cell non-Hodgkin lymphoma (NHL) (68%), followed by T-cell NHL (13%) and Hodgkin lymphoma (9%). All patients conditioned with high-dose melphalan had multiple myeloma. Two patients with secondary amyloidosis were included in the analysis and had normal cardiac function prior to auto-HCT. The patients were on the ward for a median of 14 days (range, 9-110) or 15 days (range, 10-67) after auto-HSCT and conditioning with BEAM or high-dose melphalan, respectively (Table 1).
Few patients in the BEAM cohort (28; 8%) and in the high-dose melphalan cohort (18; 6%) had progressive disease at auto-HSCT. Seventy-seven patients (23%) received chemotherapy and 43 (13%) received radiotherapy after treatment with BEAM and auto-HSCT. In contrast, 170 (58%) were treated with chemotherapy and 90 (30%) were treated with radiotherapy after high-dose melphalan and auto-HSCT. Of patients treated with BEAM, 12 patients (4%) underwent a second auto-HSCT and 41 patients (12%) underwent an allogeneic hematopoietic stem cell transplantation (allo-HSCT) after auto-HSCT, whereas of patients treated with high-dose melphalan, 102 (35%) and 58 (20%) patients underwent a subsequent auto-HCT or allo-HSCT, respectively. The median follow-up was 52 months (range, 0.2-153) and 50 months (range, 0.6-149) for patients treated with BEAM or high-dose melphalan, respectively (Table 1).
Early complications after auto-HSCT
Patients undergoing auto-HSCT showed early complications that precluded the success of therapy for some. We analyzed which early complications were detected in our patients before discharge. Almost all patients had signs of infections, including elevated C-reactive protein as an indicator of inflammation after BEAM treatment (n = 323; 96%) and after high-dose melphalan (n = 284, 96%) and auto-HSCT. Fifty-three patients (16%) and 37 patients (13%) had a bacteremia, as defined by the detection of bacteria or fungi in blood cultures, and 38 (11%) and 11 (4%) suffered from pneumonia after BEAM treatment or high-dose melphalan, respectively. Again, few patients (21 [6%] after BEAM and 3 [1%] after high-dose melphalan) had to be admitted to the intensive care unit for further treatment (supplemental Table 1B).
Impact of patient characteristics, including comorbidities and organ function prior to auto-HSCT, on outcome
We focused on studying the association between clinical patient characteristics, as well as lung and cardiac function, and OS using univariate und multivariate analyses (Cox proportional-hazards regression). In the univariate analyses, several patient clinical characteristics, comorbidities, and several lung and cardiac function parameters were shown to be associated with increased risk of death after BEAM (supplemental Table 2) and high-dose melphalan conditioning prior to auto-HSCT (supplemental Table 3).
In the multivariate analysis, age ≥ 70 years, KPS ≤ 80%, HCT-CI score ≥ 4, DLCOcSB ≤ 60% of predicted, and progressive disease status prior to auto-HSCT were shown to be independent risk factors for mortality in patients conditioned with BEAM prior to auto-HSCT (Table 2). Similarly , age > 60 years, HCT-CI score ≥ 4, and DLCOcSB ≤ 60% of predicted were associated with higher mortality in patients treated with high-dose melphalan prior to auto-HSCT (Table 2). These data identified clinical characteristics, such as age, HCT-CI score ≥ 4, and DLCOcSB ≤ 60% of predicted (P < 0.1), as common independent risk factors for patients conditioned with BEAM or high-dose melphalan.
Clinical and lung function parameters associated with OS in multivariate Cox regression analysis after conditioning using BEAM or high-dose melphalan
| Variables . | BEAM . | High-dose melphalan . | ||||
|---|---|---|---|---|---|---|
| Patients, n . | HR (95% CI) . | P . | Patients, n . | HR (95% CI) . | P . | |
| DLCOcSB ≤ 60 % of predicted | 37 | 2.47 (1.47-4.13) | .001 | 31 | 1.74 (0.92-3.28) | .08 |
| Age > 70 y | 23 | 2.13 (1.07-4.22) | .03 | 126* | 2.05 (1.28-3.26)* | .003* |
| KPS ≤ 80% | 89 | 1.89 (1.20-2.97) | .005 | 109 | 1.28 (0.81-2.03) | .28 |
| HCT-CI score ≥ 4 | 137 | 1.46 (0.94-2.28) | .09 | 56 | 2.11 (1.26-3.53) | .004 |
| Progressive disease status prior to auto-HSCT | 28 | 7.18 (4.14-12.4) | .001 | 18 | 1.07 (0.46-2.50) | .88 |
| Variables . | BEAM . | High-dose melphalan . | ||||
|---|---|---|---|---|---|---|
| Patients, n . | HR (95% CI) . | P . | Patients, n . | HR (95% CI) . | P . | |
| DLCOcSB ≤ 60 % of predicted | 37 | 2.47 (1.47-4.13) | .001 | 31 | 1.74 (0.92-3.28) | .08 |
| Age > 70 y | 23 | 2.13 (1.07-4.22) | .03 | 126* | 2.05 (1.28-3.26)* | .003* |
| KPS ≤ 80% | 89 | 1.89 (1.20-2.97) | .005 | 109 | 1.28 (0.81-2.03) | .28 |
| HCT-CI score ≥ 4 | 137 | 1.46 (0.94-2.28) | .09 | 56 | 2.11 (1.26-3.53) | .004 |
| Progressive disease status prior to auto-HSCT | 28 | 7.18 (4.14-12.4) | .001 | 18 | 1.07 (0.46-2.50) | .88 |
Multivariate analysis was performed with the Cox proportional-hazards regression model using parameters influencing outcome and DLCOcSB ≤ 60% of predicted as hypothesis covariate.
Age > 60 years.
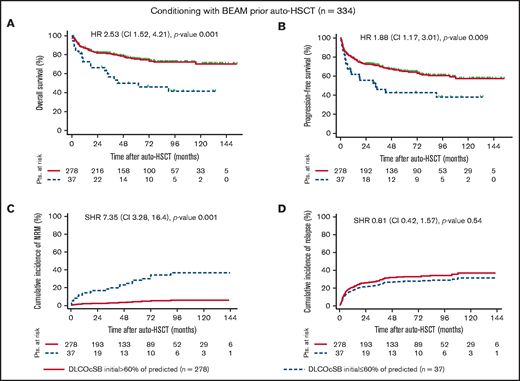
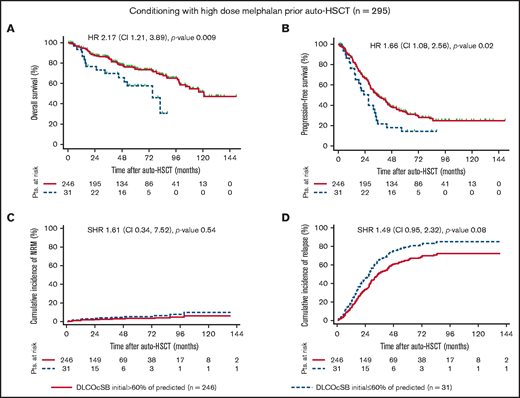
Notably, DLCOcSB ≤ 60% of predicted was associated with decreased PFS and increased NRM in patients treated with BEAM in univariate analyses (Figure 1). The impact of DLCOcSB ≤ 60% of predicted on OS was higher in patients treated with BEAM (HR, 2.53; P = .001) vs patients treated with high-dose melphalan (HR, 2.17; P = .01) (Figures 1 and 2). Hence, DLCOcSB ≤ 60% of predicted is associated with increased NRM in the multivariate analysis in patients treated with BEAM (Table 3), suggesting that this parameter is a useful factor to predict the outcomes of patients undergoing auto-HSCT after high-dose BEAM chemotherapy. In contrast, age > 60 years and HCT-CI score ≥ 4 (but not DLCOcSB < 60% of predicted) were the only factors associated with increased NRM in the multivariate analysis (P < 0.1) in patients undergoing auto-HSCT after chemotherapy with high-dose melphalan (Table 3). The above-mentioned clinical characteristics and lung function parameters previously identified to affect OS in patients treated with BEAM in the multivariate analysis did not always influence OS in patients treated with high-dose melphalan, suggesting differences across the treatment cohorts (supplemental Figures 1 through 4).
Outcome variables by DLCOcSB > 60% or ≤60% of predicted in patients conditioned with BEAM prior to auto-HSCT. Kaplan-Meier curves represent OS (A) and PFS (B), and the cumulative incidence curves represent NRM (C) and relapse (D) incidence in patients with impaired lung function, as defined by DLCOcSB > 60% or ≤60% of predicted. Statistical analysis of OS and PFS was performed using the log-rank test, whereas that for the cumulative incidence of NRM and relapse was performed using Fine and Gray regression models in the presence of competing risks. HRs and SHRs represent the risk of DLCOcSB ≤ 60% of predicted compared with DLCOcSB > 60% of predicted. DLCOcSB values before auto-HSCT and prior to conditioning with BEAM were not available for 19 patients (5.4%). Pts., patients.
Outcome variables by DLCOcSB > 60% or ≤60% of predicted in patients conditioned with BEAM prior to auto-HSCT. Kaplan-Meier curves represent OS (A) and PFS (B), and the cumulative incidence curves represent NRM (C) and relapse (D) incidence in patients with impaired lung function, as defined by DLCOcSB > 60% or ≤60% of predicted. Statistical analysis of OS and PFS was performed using the log-rank test, whereas that for the cumulative incidence of NRM and relapse was performed using Fine and Gray regression models in the presence of competing risks. HRs and SHRs represent the risk of DLCOcSB ≤ 60% of predicted compared with DLCOcSB > 60% of predicted. DLCOcSB values before auto-HSCT and prior to conditioning with BEAM were not available for 19 patients (5.4%). Pts., patients.
Outcome variables by DLCOcSB > or ≤60% of predicted in patients conditioned with high-dose melphalan prior to auto-HSCT. Kaplan-Meier curves represent OS (A) and PFS (B), and the cumulative incidence curve represent NRM (C) and relapse (D) incidence in patients with impaired lung function, as defined by DLCOcSB > 60% or ≤60% of predicted. Statistical analysis of OS and PFS was performed using the log-rank test, whereas that for the cumulative incidence of NRM and relapse was performed using Fine and Gray regression models in the presence of competing risks. HRs and SHRs represent the risk of DLCOcSB ≤ 60% of predicted compared with DLCOcSB > 60% of predicted. DLCOcSB values before auto-HSCT and prior to conditioning with high-dose melphalan were not available for 18 patients (6.1%). Pts., patients.
Outcome variables by DLCOcSB > or ≤60% of predicted in patients conditioned with high-dose melphalan prior to auto-HSCT. Kaplan-Meier curves represent OS (A) and PFS (B), and the cumulative incidence curve represent NRM (C) and relapse (D) incidence in patients with impaired lung function, as defined by DLCOcSB > 60% or ≤60% of predicted. Statistical analysis of OS and PFS was performed using the log-rank test, whereas that for the cumulative incidence of NRM and relapse was performed using Fine and Gray regression models in the presence of competing risks. HRs and SHRs represent the risk of DLCOcSB ≤ 60% of predicted compared with DLCOcSB > 60% of predicted. DLCOcSB values before auto-HSCT and prior to conditioning with high-dose melphalan were not available for 18 patients (6.1%). Pts., patients.
Multivariate analysis for NRM of clinical characteristics and lung and cardiac function parameters in patients conditioned with BEAM or high-dose melphalan prior to auto-HSCT
| Variables . | BEAM . | High-dose melphalan . | ||||
|---|---|---|---|---|---|---|
| n . | SHR (95% CI) . | P . | n . | SHR (95% CI) . | P . | |
| DLCOcSB ≤ 60% of predicted | 37 | 6.50 (2.35-17.9) | .001 | 31 | 0.71 (0.11- 4.4) | .71 |
| Solid tumor in previous diagnosis | 32 | 4.83 (1.73-13.4) | .003 | |||
| Hematological disease prior to current diagnosis | 32 | 3.33 (1.03-10.8) | .04 | |||
| Lung disease prior to auto-HSCT | 33 | 3.88 (1.12-7.37) | .03 | |||
| FEV1 initial ≤ 70% of predicted | 41 | 2.15 (0.94-4.92) | .07 | |||
| Age > 70 y | 23 | 1.12 (0.23-5.40) | .89 | 126* | 3.1 (0.76- 12.6)* | .10* |
| KPS ≤ 80% | 89 | 1.53 (0.66-3.54) | .32 | 109 | 1.59 (0.44- 5.68) | .47 |
| HCT-CI score ≥ 4 | 137 | 1.10 (0.66-2.28) | .85 | 56 | 4.4 (0.98-19.6) | .05 |
| Variables . | BEAM . | High-dose melphalan . | ||||
|---|---|---|---|---|---|---|
| n . | SHR (95% CI) . | P . | n . | SHR (95% CI) . | P . | |
| DLCOcSB ≤ 60% of predicted | 37 | 6.50 (2.35-17.9) | .001 | 31 | 0.71 (0.11- 4.4) | .71 |
| Solid tumor in previous diagnosis | 32 | 4.83 (1.73-13.4) | .003 | |||
| Hematological disease prior to current diagnosis | 32 | 3.33 (1.03-10.8) | .04 | |||
| Lung disease prior to auto-HSCT | 33 | 3.88 (1.12-7.37) | .03 | |||
| FEV1 initial ≤ 70% of predicted | 41 | 2.15 (0.94-4.92) | .07 | |||
| Age > 70 y | 23 | 1.12 (0.23-5.40) | .89 | 126* | 3.1 (0.76- 12.6)* | .10* |
| KPS ≤ 80% | 89 | 1.53 (0.66-3.54) | .32 | 109 | 1.59 (0.44- 5.68) | .47 |
| HCT-CI score ≥ 4 | 137 | 1.10 (0.66-2.28) | .85 | 56 | 4.4 (0.98-19.6) | .05 |
Multivariate analysis following backward selection was performed using the Fine and Gray regression model in the presence of competing risks and using age, HCT-CI score, and KPS as hypothesis covariates.
FEV1, forced expiratory volume in 1 second.
Age > 60 years.
CO-diffusion abnormalities are associated with specific clinical characteristics prior to auto-HSCT and complications after auto-HSCT
Because of the association between DLCOcSB ≤ 60% of predicted and outcome in patients treated with BEAM and high-dose melphalan, we assessed the association between this lung parameter and clinical features, as well as the impact of this factor on the frequency of complications after auto-HSCT and cause of death. DLCOcSB ≤ 60% of predicted was associated with smokers or previous smokers, treatment with lung-toxic substances (ie, bleomycin and brentuximab), and mediastinal radiotherapy prior to BEAM treatment (supplemental Table 4A). These patients suffered from more complications, such as respiratory insufficiency, acute renal failure, cardiac and hemodynamic instability, and bleeding/thrombosis, after auto-HSCT; as a consequence, they were more treated more frequently in the intensive care unit and remained on the ward after auto-HSCT (supplemental Table 4A). Patients with DLCOcSB ≤ 60% and who were treated with high-dose melphalan prior to auto-HSCT exhibited an association with clinical characteristics, such as KPS ≤ 80% and current/previous smoking (supplemental Table 4B).
Several lung function parameters, including higher residual volume/total lung capacity ratio, reduced total lung capacity, and lower mid-expiratory flow at 25% of vital capacity, were associated with patients with DLCOcSB ≤ 60%. A total of 35 of 37 (95%) and 6 of 31 (19%) computed tomography scans were performed before auto-HSCT in patients with an initial DLCOcSB ≤ 60% and conditioned with BEAM or high-dose melphalan, respectively. Several structural changes were detected as lung infiltrates, lung involvement with lymphoma, and fibrotic and emphysema-associated changes prior to auto-HSCT (supplemental Table 5).
Overall, 96 patients (28.7%) died within the median observation period ∼ 50 months in the BEAM-treated cohort: 67 patients (24.1%) in the group with DLCOcSB > 60% of predicted and 19 patients (51.3%) in the group with DLCOcSB ≤ 60% of predicted. DLCOcSB values were not available prior to auto-HSCT for 10 patients in the BEAM group who died. Overall, patients with DLCOcSB ≤ 60% of predicted had a higher NRM [n = 7 (19%)] compared with patients with DLCOcSB > 60% of predicted [n = 8 (3%)]. Causes of NRM were aspergillosis, Pseudomonas aeruginosa–related sepsis, ST-elevated myocardial infarction, central nervous system (CNS) organic-psycho syndrome, multiorgan dysfunction, CNS hemorrhage, and secondary malignancies, such as acute myeloid leukemia (Table 4). Among patients treated with high-dose melphalan, 106 patients (36%) died: 74 (30%) in the group with DLCOcSB > 60% of predicted and 14 (45%) in the group with DLCOcSB ≤ 60% of predicted. DLCOcSB values were not available prior to auto-HSCT for 18 patients in the high-dose melphalan group who died. Only 4 (1%) patients with DLCOcSB > 60% of predicted and only 1 patient (3%) with DLCOcSB ≤ 60% of predicted died from NRM. Causes included pancreatic cancer, bronchial carcinoma, heart arrhythmia, and acute graft-versus-host disease after allo-HSCT in the former group and hypoxia related to pneumonia in the latter group (Table 5).
Cause of mortality by DLCOcSB > 60% or ≤60% of predicted in patients undergoing auto-HSCT conditioned with BEAM
| . | DLCOcSB > 60% of predicted . | DLCOcSB ≤ 60% of predicted . |
|---|---|---|
| Patients, n | 278 | 37 |
| Alive | 211 (76) | 18 (49) |
| Relapse | 57 (20) | 10 (27) |
| NRM | 8 (3) | 7 (19) |
| Cause of death (months after auto-HSCT) | Pneumocystis jiroveci pneumonia (5.6) | Aspergillosis (0.6) |
| Pneumonia, hypoxia by aspiration (0.7) | Sepsis by Pseudomonas aeruginosa (10.2) | |
| Pneumonic sepsis (4.0) | ST-elevated myocardial infarction (17.8) | |
| ARDS (0.5) | CNS-organic psycho-syndrome (2.1) | |
| Guillain-Barré syndrome (32) | Multiorgan dysfunction (1.3) | |
| Acute abdomen (3.7) | CNS hemorrhage (0.4) | |
| Hemophagocytic syndrome (43.8) | Second neoplasia (AML) (41.7) | |
| Second neoplasia (tongue carcinoma) (55) | ||
| Unknown cause of death, n (%) | 2 (1) | 2 (5) |
| . | DLCOcSB > 60% of predicted . | DLCOcSB ≤ 60% of predicted . |
|---|---|---|
| Patients, n | 278 | 37 |
| Alive | 211 (76) | 18 (49) |
| Relapse | 57 (20) | 10 (27) |
| NRM | 8 (3) | 7 (19) |
| Cause of death (months after auto-HSCT) | Pneumocystis jiroveci pneumonia (5.6) | Aspergillosis (0.6) |
| Pneumonia, hypoxia by aspiration (0.7) | Sepsis by Pseudomonas aeruginosa (10.2) | |
| Pneumonic sepsis (4.0) | ST-elevated myocardial infarction (17.8) | |
| ARDS (0.5) | CNS-organic psycho-syndrome (2.1) | |
| Guillain-Barré syndrome (32) | Multiorgan dysfunction (1.3) | |
| Acute abdomen (3.7) | CNS hemorrhage (0.4) | |
| Hemophagocytic syndrome (43.8) | Second neoplasia (AML) (41.7) | |
| Second neoplasia (tongue carcinoma) (55) | ||
| Unknown cause of death, n (%) | 2 (1) | 2 (5) |
Unless otherwise noted, data are n (%). DLCOcSB values were not available for 19 patients (5.4%) before auto-HSCT and prior to conditioning with BEAM.
AML, acute myeloid leukemia; ARDS, acute respiratory distress syndrome.
Cause of mortality by DLCOcSB > 60% or ≤60% of predicted in patients undergoing auto-HSCT conditioned with high-dose melphalan
| . | DLCOcSB > 60% of predicted . | DLCOcSB ≤ 60% of predicted . |
|---|---|---|
| Patients, n | 246 | 31 |
| Alive | 172 (70) | 17 (55) |
| Relapse | 66 (27) | 12 (39) |
| NRM | 4 (1) | 1 (3) |
| Cause of death (months after auto-HSCT) | Pancreatic cancer (99.5) | Pneumonia, hypoxia (12) |
| Arrhythmia (4.5) | ||
| Bronchial carcinoma (12.4) | ||
| Acute GvHD (9.8) | ||
| Unknown cause of death | 5 (2) | 1 (3) |
| . | DLCOcSB > 60% of predicted . | DLCOcSB ≤ 60% of predicted . |
|---|---|---|
| Patients, n | 246 | 31 |
| Alive | 172 (70) | 17 (55) |
| Relapse | 66 (27) | 12 (39) |
| NRM | 4 (1) | 1 (3) |
| Cause of death (months after auto-HSCT) | Pancreatic cancer (99.5) | Pneumonia, hypoxia (12) |
| Arrhythmia (4.5) | ||
| Bronchial carcinoma (12.4) | ||
| Acute GvHD (9.8) | ||
| Unknown cause of death | 5 (2) | 1 (3) |
Unless otherwise noted, data are n (%). DLCOcSB values were not available for 18 patients (6.1%) before auto-HSCT and prior to conditioning with high-dose melphalan.
GvHD, graft-versus-host disease.
We evaluated the influence of auto-HSCT on cardiac and pulmonary function (supplemental Table 6). We included ∼30% of patients conditioned with BEAM and ∼60% of patients conditioned with high-dose melphalan with available echocardiography and PFT prior to and after auto-HSCT. Interestingly, the percentage of DLCOcSB predicted showed the most significant decrease in patients conditioned with BEAM (median, 77% vs 68% of predicted; P = .01) or high-dose melphalan (median, 78% vs 74% of predicted; P = .003), suggesting that the auto-HSCT procedure including conditioning chemotherapy affects CO-diffusion capacity. Significant changes were also observed in the cardiac ejection fraction and RV/TLC ratio (supplemental Table 6).
Discussion
The impact of organ dysfunction, including lung and cardiac function abnormalities, has been studied extensively in patients undergoing allo-HSCT. However, less is known about the influence of lung and cardiac dysfunction in patients undergoing auto-HSCT. Lung function parameters are included in comorbidity scores used in allo-HSCT (eg, HCT-CI19,20 ), as well as in disease-specific scores (eg, R-MCI).16-18 The R-MCI score was developed to assess the frailty of patients with multiple myeloma, giving a high weight to lung function, including forced expiratory volume in 1 second (FEV1) and DLCOcSB. However, optimal cutoff values have not been established for auto-HSCT for patients with aggressive lymphoma or multiple myeloma. Therefore, we analyzed lung and cardiac function parameters and their impact on outcome, complications, and causes of death in patients undergoing auto-HSCT after conditioning with BEAM and high-dose melphalan at our institution between 1st January 2007 and 31st December 2017.
Impaired initial CO-diffusion capacity, as defined by DLCOcSB ≤ 60%, was associated with decreased OS in patients after auto-HSCT with BEAM or high-dose melphalan conditioning (Figures 1 and 2; Table 2). Similar results were observed in patients undergoing allo -HSCT using a BCNU-containing protocol (FBM; fludarabine, BCNU/carmustine, melphalan) at our institution.29,30 BCNU may cause lung damage at high doses as part of chemotherapy for solid tumors, such as in auto-HSCT and allo-HSCT conditioning protocols.31,32 This might explain the higher toxicity of BEAM in patients with previously damaged lungs. Similarly, another study found that impaired lung function, including a lower lung diffusion index, in patients with lymphoma or multiple myeloma prior to auto-HSCT was associated with the use of pneumotoxic substances and worse outcomes.33 In our cohort, patients with CO-diffusion abnormalities also exhibited higher toxicity and incidence of pulmonary complications without receiving lung-toxic substances in the group receiving high-dose melphalan, suggesting frailty as another important factor in this patient population.
Lung disease prior to auto-HSCT, including pneumonia, pulmonary embolism, chronic obstructive pulmonary disease, and sleep apnea syndrome, as well as obstructive lung dysfunction (defined as FEV1 ≤ 70% of predicted), were also identified in multivariate analysis as being related to increased NRM in patients treated with BEAM (Table 3). Interestingly, DLCOcSB and FEV1 are included in the HCT-CI and are known to impact the outcome of patients undergoing allo-HSCT and auto-HSCT. We hypothesize that patients with lung damage prior to auto-HSCT are less suitable for BEAM conditioning, most likely because of the high-dose BCNU, which causes further lung toxicity.
Moreover, clinical characteristics, early complications, and cause of death were analyzed in patients with CO abnormalities who were treated with BEAM or high-dose melphalan. Similar to previous studies, decreased DLCOcSB was associated with previous or active smoking in patients treated with BEAM or high-dose melphalan, as well as with lung-toxic substances, such as bleomycin and brentuximab and mediastinal radiotherapy, prior to auto-HSCT in patients treated with BEAM.33 As expected, patients with DLCOcSB abnormalities suffer more frequently from severe early auto-HSCT complications; as a consequence, they were treated more frequently in the intensive care unit and stayed longer on the ward (supplemental Table 4A). Of note, patients with DLCOcSB ≤ 60% and subsequently poor prognosis showed the respiratory physiological pattern of emphysema; presumably, cigarette smoking contributes to the poor prognosis (supplemental Table 5). Hence, although these data should also be interpreted with caution because cigarette smoking was not assessed systematically, the auto-HSCT procedure including conditioning chemotherapy probably affects the CO diffusion capacity of transplanted patients (supplemental Table 6).
Patients with lung function parameters (DLCOcSB ≤ 60% of predicted, FEV1 ≤ 70% of predicted, or MEF25 ≤ 20% of predicted) or clinical parameters (lung disease prior to auto-HSCT, age > 70 years, HCT-CI score ≥ 4, or decreased performance status [KPS ≤ 80%]) that are associated with decreased OS and increased NRM might also profit from less-intensive conditioning chemotherapy prior to auto-HSCT. Efforts have been made to replace BCNU with thiotepa in conditioning protocols prior to auto-HSCT. Thiotepa-based protocols, such as thiotepa, etoposide, cytarabine, and melphalan or thiotepa, etoposide, cyclophosphamide, cytarabine, and melphalan, were shown to have similar efficacy, as well as a similar frequency of infectious and noninfectious complications, as BEAM in auto-HSCT.34,35 In the allo-HSCT setting at our institution, we first reduced the dose of BCNU and later replaced it with thiotepa in reduced-intensity conditioning to decrease or eliminate the risk of lung toxicity.36 De-escalation of the melphalan dose while maintaining the antitumor activity is a good alternative, especially for older and fragile patients with multiple myeloma.37 The most suitable protocols for specific patients with advanced age, comorbidities, and/or impaired lung and cardiac function are important research questions that should be addressed in the future in the context of controlled randomized clinical trials. However, randomized clinical trials in the context of conditioning prior to auto-HSCT are difficult to conduct, and retrospective data analysis may provide valuable information.
Patients with comorbidities, especially cardiac and lung diseases, prior to auto-HSCT may have a more dismal outcome. Although the HCT-CI score was not developed to address the risk for NRM in patients undergoing auto-HSCT, it was also shown to predict the outcome of patients undergoing auto-HSCT.15,38,39 We applied the HCT-CI score19 to our cohort of patients and used age as a covariate similar to the modified HCT-CI score.20 We found an association with decreased OS in both cohorts in the multivariate analysis. Nevertheless, an HCT-CI score ≥ 4 was identified as an independent factor in the multivariate analysis for NRM in the high-dose melphalan cohort but not in the BEAM cohort. We hypothesize that identified lung function parameters, together with the HCT-CI score, may better predict NRM and OS in patients undergoing auto-HSCT with different conditioning protocols.
Clinical characteristics, as well as the lung and cardiac function, of patients with multiple myeloma or lymphoma are very different. As we show throughout this article, we identified different patient characteristics and lung and cardiac function parameters associated with decreased OS and worse outcome in each independent cohort. Hence, variables identified in both cohorts in the multivariate analysis (eg, age) also differed with regard to the identified cutoff values. Depending on the underlying disease, we should take into account different parameters to assess the risk of decreased OS and NRM after auto-HSCT.
Remarkably, 15 (4.5%) patients treated with BEAM and 5 (1.3%) patients treated with high-dose melphalan experienced NRM. Only 6 patients (1.7%) treated with BEAM and no patient treated with high-dose melphalan died within the first 100 days after auto-HSCT, which might be considered transplantation-related mortality (Table 4 and Table 5). In contrast, 237 (71%) patients treated with BEAM and 189 (64%) patients treated with high-dose melphalan prior to auto-HSCT were alive at last follow-up, suggesting auto-HSCT as a potential curative treatment with low morbidity and mortality.
This study has several limitations. First, we analyzed the data for patients undergoing auto-HSCT retrospectively after high-dose chemotherapy conditioning at a single institution. Future prospective interventional studies should elucidate which conditioning protocols are the most suitable for patients of older age with comorbidities and/or with lung and cardiac dysfunction. Second, to have a homogenous treatment, we only included patients conditioned with BEAM and high-dose melphalan (200 mg/m2 in total). Different lung and cardiac function parameters might be associated with outcome in patients undergoing auto-HSCT with different conditioning protocols. Our data should be evaluated in patients with different conditioning protocols, such as TEAM (thiotepa, etoposide, cytarabine, and melphalan) or TECAM (thiotepa, etoposide, cyclophosphamide, cytarabine, and melphalan), or a reduced dose of melphalan. Multicenter international cooperative studies with larger numbers of patients should correct for the bias of single-center studies. Therefore, our data should be interpreted with caution and validated in different patient cohorts.
In summary, we identified several pulmonary function abnormalities as risk factors associated with decreased OS and increased NRM in patients conditioned with BEAM and high-dose melphalan prior to auto-HSCT. These findings may lead to important preventive and early therapeutic interventions in patients undergoing auto-HSCT that will decrease morbidity and mortality further. This study should help to identify a population at risk that might profit from less intense conditioning protocols to reduce mortality and improve outcome after auto-HSCT.
Acknowledgments
The authors thank J. Urban for managing the data bank of transplanted patients, the medical technicians in the Stem Cell Transplantation Unit and Pulmonary Function Test Unit of the University Medical Center Freiburg, and all of the patients and staff on the Löhr, Holthusen, and Thannhauser wards. They also thank C. Duque-Afonso for graphic design.
This work was supported by a grant from the German Federal Ministry of Education and Research (BMBF 01 EO 0803). J.D.-A., M.W., and G.I. received support from Else Kröner-Fresenius-Stiftung (2018-A56). J.D.-A. received research support from the German Research Foundation (Deutsche Forschungsgemeinschaft, ref. DU 1287/3-1 and 1287/5-1), the Forschungskommission of the University of Freiburg Medical School (DUQ1106/16), and the Berta Ottenstein-Program for Advanced Clinician Scientists, Faculty of Medicine, University of Freiburg.
Authorship
Contribution: J.D.-A. and M.E. conceived and designed the study; J.D.-A., S.E., R.Z., T.S., R.W., H.B., J.D., J.F., J.M.-Q., and R.M. acquired data; J.D.-A., S.E., G.I., M.W., and M.E. analyzed and interpreted data; J.D.-A. and G.I. performed statistical analyses; and J.D.-A., S.E., G.I., M.W., T.S., R.Z., R.W., J.M.-Q., R.M., J.D., J.F., and M.E. critically revised the manuscript.
Conflict-of-interest disclosure: J.F. has received research support and speaker’s honoraria from Medac, Neovii, and Riemser. J.D.-A. has received speaker’s honoraria from Roche, Amgen, AstraZeneca, Riemser, and Sobi and travel support from Gilead and Sobi. R.Z. has received speaker’s honoraria from Incyte, Novartis, Roche, and Mallinckroth. The remaining authors declare no competing financial interests.
Correspondence: Jesús Duque-Afonso, Department of Hematology/Oncology/Stem Cell Transplantation, Faculty of Medicine and University of Freiburg Medical Center, Hugstetterstr. 55, D-79106 Freiburg, Germany; e-mail: jesus.duque.afonso@uniklinik-freiburg.de.
References
Author notes
J.D.-A. and S.E. contributed equally to this study as joint first authors.
R.M. and M.E. contributed equally to this study as joint senior authors.
Presented in abstract form at the 47th Annual Meeting of the European Society for Blood and Marrow Transplantation (virtual), 14th March 2021.
Data sharing requests should be sent to Jesús Duque-Afonso (jesus.duque.afonso@uniklinik-freiburg.de).
The full-text version of this article contains a data supplement.