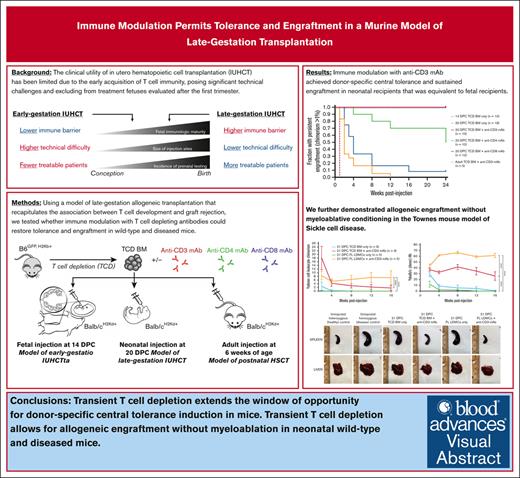
Key Points
Transient TCD extends the window of opportunity for donor–specific central tolerance induction in mice.
Transient TCD allows for allogeneic engraftment without myeloablation in neonatal wild-type and diseased mice.
Visual Abstract
In utero hematopoietic cell transplantation is an experimental nonmyeloablative therapy with potential applications in hematologic disorders, including sickle cell disease (SCD). Its clinical utility has been limited due to the early acquisition of T-cell immunity beginning at ∼14 weeks gestation, posing significant technical challenges and excluding treatment fetuses evaluated after the first trimester. Using murine neonatal transplantation at 20 days postcoitum (DPC) as a model for late-gestation transplantation (LGT) in humans, we investigated whether immune modulation with anti-CD3 monoclonal antibody (mAb) could achieve donor-specific tolerance and sustained allogeneic engraftment comparable with that of the early-gestation fetal recipient at 14 DPC. In allogeneic wild-type strain combinations, administration of anti-CD3 mAb with transplantation resulted in transient T-cell depletion followed by central tolerance induction confirmed by donor–specific clonal deletion and skin graft tolerance. Normal immune responses to third-party major histocompatibility complex and viral pathogens were preserved, and graft-versus-host disease did not occur. We further demonstrated the successful application of this approach in the Townes mouse model of SCD. These findings confirm the developing fetal T-cell response as a barrier to LGT and support transient T-cell depletion as a safe and effective immunomodulatory strategy to overcome it.
Introduction
In utero hematopoietic cell transplantation (IUHCT) is a nonmyeloablative transplantation approach that results in donor cell engraftment across immune barriers.1,2 It has the potential to treat a number of congenital immune, metabolic, and hematologic disorders, including sickle cell disease (SCD) and thalassemia.3-6 IUHCT has been successful in preclinical studies in the murine, canine, ovine, and porcine models.1,2,7,8 Clinical translation, however, has been disappointing. Despite early success in fetuses with severe combined immunodeficiency disease (SCID) and bare lymphocyte syndrome,9-12 achieving tolerance and meaningful engraftment in immunologically normal fetuses has proven elusive.13,14 One contributing factor is the acquisition of T-cell immune responses by the fetus at the time of transplantation15-20 and the significant technical and practical challenges of performing IUHCT earlier in gestation before that time.21-23 An additional factor is the competition to adult donor hematopoietic stem cells (HSCs) within the recipient hematopoietic niche.3,24-27 For IUHCT to fulfill its clinical potential, we must develop strategies to extend the window of opportunity for tolerance induction and engraftment to later in gestation and validate these strategies in relevant disease models.
We previously reported a neonatal mouse model of late-gestation transplantation (LGT) in humans that recapitulates the association between the emergence of mature T cells in the periphery and immunocompetence with respect to allograft rejection.19,28 We further demonstrated that enrichment of the donor allograft with regulatory T cells (Tregs) is 1 strategy by which to achieve tolerance and engraftment beyond this point.28 This work relied upon the supposition that donor-derived Tregs suppressed the nascent recipient alloreactive T-cell response, but this was never proven directly. Furthermore, Treg-based cell therapies are difficult to translate into preclinical large animal models and human trials.29,30 To build upon and improve upon our prior work, we investigated whether direct modulation of the recipient T-cell response with depleting antibodies can effectively and safely induce tolerance and engraftment in immunocompetent murine neonates. Rigorous validation of this approach in allogeneic wild-type and disease model murine strain combinations could serve as a foundation for subsequent studies of fetal immunomodulation in preclinical large animal models and potential clinical translation.
Methods
Mice
Balb/cJ (Balb/c, H2kd, Jackson strain no. 000651), C57BL/6J (B6, H2kb, Jackson strain no. 000664), C3H/HeJ (C3H, H2kk, Jackson strain no. 000659), and B6;129-Hbbtm2(HBG1,HBB∗)Tow/Hbbtm3(HBG1,HBB)Tow Hbatm1(HBA)Tow/J (Townes, H2kb, Jackson strain no. 013071) mice were purchased from Jackson Laboratories. C57BL/6TgN(act-EGFP)OsbY01 (B6GFP, H2kb) mice were provided by M. Okabe (Osaka University, Suita, Japan) and were maintained in our colony. The experimental protocols were approved by the Children’s Hospital of Philadelphia Institutional Animal Care and Use Committee and followed the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Murine models of early-gestation, late-gestation, and postnatal transplantation
Bone marrow (BM) was harvested from 6- to 8-week-old donors as previously described.31 Briefly, the tibias, femurs, and iliac bones were flushed with sterile phosphate-buffered saline (Corning ref no. 21-031-CM). The BM was then filtered and layered over sterile Ficoll-Paque PLUS (Cytiva catalog no. 17144003) to isolate the low-density mononuclear cell (LDMC) layer. T-cell depletion was performed by magnetic activated cell sorting using a CD3e MicroBead Kit, mouse (Miltenyi Biotec catalog no. 130-094-973) with CD3− purity ≥99.7%, as confirmed by flow cytometry. Fetuses of time-dated pregnant mice were injected at 14 days postcoitum (DPC) with 9.7 × 106 T-cell depleted (TCD) BM cells, as previously described, to model early-gestation IUHCT in humans.21 Briefly, midline laparotomy was performed under isoflurane anesthesia (3%) to expose the uterine horns. The vitelline vein was injected using a programmable microinjector (IM-300 Microinjector; MicroData Instrument Inc, S. Plainfield, NJ), a 100 μm beveled glass micropipette, and a dissecting microscope. All pups in the litter were injected. To account for the effect on engraftment of maternal antibodies transferred via breastmilk to newborn pups,32 all litters underwent transplantation before birth were fostered after birth with naïve Balb/c dams. Neonatal transplantation was performed 20 DPC, as previously described,28 using an equivalent dose of TCD BM to model late-gestation IUHCT in humans. The frequency of T-cell subtypes in the peripheral blood (PB) of uninjected littermates was assessed at the time of injection to confirm the immunologic maturity of the recipient litter, as previously reported. Neonatal transplantation does not require fostering because of the absence of maternal immune exposure to the donor major histocompatibility complex (MHC). Transplantation of 6-week-old adults via tail vein injection was used to model postnatal HSC transplantation. The TCD BM was transplanted at a dose equivalent by weight to that administered to neonates (5 × 108 cells per kg). No myeloablative conditioning was used for fetal, neonatal, or adult transplantation.
Transient TCD
Transient T-cell depletion was performed by intravascular injection of monoclonal antibodies (mAbs). CD3+ T-cell depletion was performed using anti-mouse CD3 Functional Grade Purified (eBioscience, Rat IgG2b kappa, clone 17A2, ref no. 16-0032-85) at a dose of 5 mg/kg. This dose was chosen based on the clinical dosing of antithymocyte globulin for graft-versus-host disease (GVHD) prophylaxis for unrelated allogeneic HSC transplantation.33 An alternative brand of anti-CD3 mAb, Ultra-LEAF Purified anti-mouse CD3 (BioLegend, Rat IgG2b kappa, clone 17A2, catalog no. 100238), was also tested at 5 mg/kg and found to be functionally equivalent. CD4+ T cell-depletion was performed using anti-mouse CD4 Functional Grade Purified (eBioscience, Rat IgG2b kappa, clone GK1.5, ref number16-0041-85) at a dose of 5 mg/kg. CD8+ T-cell depletion was performed using anti-mouse CD8a Functional Grade Purified (eBioscience, Rat IgG2a kappa, clone 53-6.7, ref no. 16-0081-85) at a dose of 5 mg/kg. PB was collected at intervals from neonates by decapitation, juveniles by facial vein phlebotomy, and weanlings/adults by retro-orbital venipuncture under isoflurane anesthesia (3%). Erythrolysis was performed using the BD Pharm Lyse lysing buffer (BD Biosciences catalog no. 555899). T-cell frequency analysis was performed by flow cytometry using BD FACSAria (BD Biosciences, San Jose, CA) using the antibody staining reported in supplemental Table 1.
Assessment of donor cell chimerism and multilineage engraftment
Antibody staining for chimerism analysis is reported in supplemental Table 1. Donor cell chimerism among CD45+ PB cells was calculated as percentage green fluorescent protein (GFP+) in the B6GFP→Balb/c strain combination, H2kd+H2kb−/(H2kd+GFP− + H2kd−GFP+) × 100 in the Balb/c→B6GFP strain combination, and H2kd+H2kb−/(H2kd+H2kb− + H2kd−H2kb+) in the Balb/c→Townes stain combination. Macroengraftment was defined as >1% donor cell chimerism. To assess donor engraftment at the level of the HSC, PB was analyzed at 6 months of age for donor cell frequency among various lineages, and BM LDMCs were analyzed for donor cell frequency among lineage−Sca-1+c-kit+ (LSK) cells. Antibody staining for multilineage and BM LSK analyses are reported in supplemental Tables 2 and 3, respectively.
Townes model of SCD
Townes mice are mixed B6;129-background (H2kb) mice in which endogenous murine β-globin genes have been replaced by human hemoglobin (Hb) α- and β-globin knock-in genes.34 So-called homozygous mice have 2 copies of the human sickle cell Hb β (βS) gene and recapitulate the phenotypic features of human SCD, including sickled erythrocytes, anemia, and multiorgan pathology secondary to chronic hypoxia and microvascular occlusion.34 Homozygous mice were used as therapeutic targets and, when uninjected, as disease controls. Heterozygous mice have 1 copy of the βS gene and 1 copy of the human wild-type Hb β (βA) gene, which is sufficient to rescue the phenotype. Uninjected heterozygous mice were therefore used as healthy controls. Homozygous Townes neonates were injected at 21 DPC via the facial vein with 19.4 × 106 TCD BM cells harvested from Balb/c (H2kd) adults as described above or 19.4 × 106 fetal liver (FL) LDMCs harvested from 14 DPC Balb/c (H2kd) fetuses. FL LDMCs were innately low in T cells (≥99% CD3− by flow cytometry) and thus were not depleted of T cells before injection. No myeloablative conditioning was used.
Statistics
The equality of the allograft survival curves was assessed using the log-rank (Mantel-Cox) test. Continuous, parametric outcomes including donor cell chimerism were compared between the 2 groups using Student's t test and among ≥3 groups using analysis of variance with multiple comparisons. Nonparametric continuous and ordinal outcomes were compared using the Kruskal-Wallis test. Statistical analysis and graphing were performed using Prism, version 9.4 (GraphPad, La Jolla, CA). All statistical tests were 2-sided with an α level set at .05 for statistical significance. Graph error bars are standard deviations unless otherwise specified.
Results
T cells can be safely and effectively depleted during early development
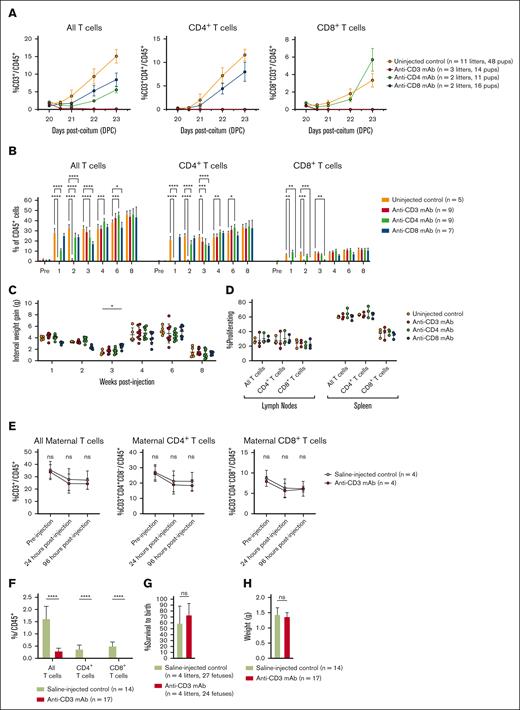
We have previously demonstrated that mature T cells emerge in the PB of developing mice shortly after birth, reaching sufficient prevalence to consistently reject allotransplants by 20 DPC.28 To determine whether antibodies could safely and effectively deplete PB T cells at this time, we administered 5 mg/kg anti-CD3, anti-CD4, or anti-CD8 mAb to Balb/c neonates at 20 DPC and then assessed T-cell prevalence in the short- and long-term. Antibody administration resulted in selective depletion of CD3+, CD4+, and CD8+ cells to 0% of all CD45+ cells within 12 hours of administration (Figure 1A), with recovery to normal prevalence within 3 to 4 weeks (Figure 1B). The survival was 100%, and weight gain was normal (Figure 1C). Quantitative T-cell recovery was accompanied by normal function, as demonstrated by the normal proliferation of both CD4+ and CD8+ T cells to nonself MHC by in vivo mixed lymphocyte reaction (MLR; Figure 1D).
Targeted antibody treatment rapidly, transiently, and safely depletes T cells with no long-term detriment to T-cell function. (A) Short-term depletion. Anti-CD3 mAb administered after birth at 20 DPC depletes PB T cells to 0% of all CD45+ cells within 12 hours of administration. (B) Quantitative T-cell recovery. All T-cell populations recover to baseline frequency within 4 weeks of antibody treatment. (C) Weight gain. The treated animals demonstrated normal weight gain, despite antibody depletion. (D) In vivo MLR. After quantitative recovery, T cells proliferate normally when presented with a nonself MHC. (E) Maternal TCD. Anti-CD3 mAb administered to the fetus before birth at 16 DPC did not deplete T cells in the maternal blood, which remained at frequencies equal to that observed after fetal saline injection. Fetal injection with anti-CD3 mAb resulted in neonatal T-cell depletion measured at birth (F) without detriment to fetal survival to birth (G) or birth weight (H). ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001. ns, not significant.
Targeted antibody treatment rapidly, transiently, and safely depletes T cells with no long-term detriment to T-cell function. (A) Short-term depletion. Anti-CD3 mAb administered after birth at 20 DPC depletes PB T cells to 0% of all CD45+ cells within 12 hours of administration. (B) Quantitative T-cell recovery. All T-cell populations recover to baseline frequency within 4 weeks of antibody treatment. (C) Weight gain. The treated animals demonstrated normal weight gain, despite antibody depletion. (D) In vivo MLR. After quantitative recovery, T cells proliferate normally when presented with a nonself MHC. (E) Maternal TCD. Anti-CD3 mAb administered to the fetus before birth at 16 DPC did not deplete T cells in the maternal blood, which remained at frequencies equal to that observed after fetal saline injection. Fetal injection with anti-CD3 mAb resulted in neonatal T-cell depletion measured at birth (F) without detriment to fetal survival to birth (G) or birth weight (H). ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001. ns, not significant.
Although the emergence of mature T cells after birth in mice necessitates neonatal transplantation studies to evaluate the effect of immune modulation on alloengraftment and tolerance induction, we also performed anti-CD3 mAb injection in fetal recipients at 16 DPC to determine whether it had any deleterious effects on the mother or fetus. Fetal injection of 5 mg/kg anti-CD3 mAb (based on fetal weight) did not result in depletion of T cells in the maternal blood (Figure 1E), despite effectively reducing T cells in the fetal blood measured after birth on 20 DPC (Figure 1F). No increase in fetal mortality or pregnancy loss was observed compared with fetal saline injection (Figure 1G), and birth weight was comparable (Figure 1F). These results suggest that T cell-depleting antibodies can be administered before birth without detrimental effects on the mother or fetus, providing a foundation for future studies in large animal models.
Transient TCD restores long-term allogeneic engraftment after neonatal transplantation
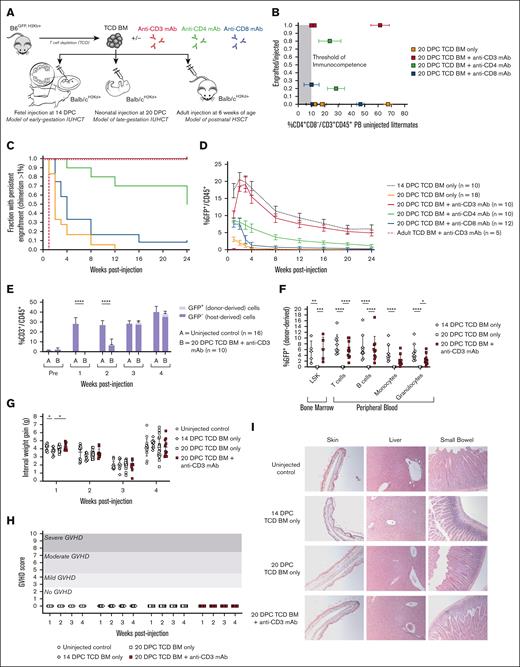
We next tested whether combining antibody administration with allogeneic neonatal transplantation could achieve long-term engraftment. Neonatal transplantation was performed after birth at 20 DPC with allogeneic TCD BM alone or in combination with anti-CD3, anti-CD4, or anti-CD8 mAbs. Engraftment was compared with fetal transplantation at 14 DPC of allogeneic TCD BM alone and with adult transplantation at 6 weeks of age of allogeneic TCD BM combined with anti-CD3 mAb (Figure 2A). All neonatal litters included in the study were confirmed to have reached our previously reported metric of sufficient immune maturity for allograft rejection (ie, circulating CD4+ T cells ≥10% of all PB CD45+ cells in uninjected littermates)28 (Figure 2B). In contrast to neonatal recipients of allogeneic transplantation alone, in which there was loss of the graft in 100% of recipients, coadministration of anti-CD3 mAb with allogeneic transplantation restored long-term engraftment in all injected neonates. Isolated depletion of CD4+ or CD8+ T-cell subsets alone was insufficient to achieve uniform long-term engraftment. The efficacy of anti-CD3 mAb was also unique to neonatal recipients, as all allografts were quickly rejected by adult recipients of TCD BM plus anti-CD3 mAb despite the successful depletion of PB T cells (Figure 2C; supplemental Figure 1). PB donor leukocyte chimerism was comparable between neonates receiving allogeneic transplantation plus anti-CD3 mAb and fetuses receiving allogeneic transplantation alone (Figure 2D). Quantitative recovery of both host- and donor-derived T cells was observed 3 weeks after transplantation (Figure 2E). All other hematopoietic lineages were similarly reconstituted by the donor-derived BM and persisted at 24 weeks after transplantation, consistent with the level of donor engraftment observed among BM LSK cells (Figure 2F) and confirming long-term engraftment at the level of the hematopoietic progenitor cell. This engraftment was achieved in the absence of GVHD, as demonstrated by normal weight gain (Figure 2G) and no phenotypic (Figure 2H) or histologic features of GVHD (Figure 2I).
Transient T-cell depletion restores long-term multilineage engraftment after neonatal transplantation. (A) Experimental design. Mice were injected at 3 developmental stages: before birth at 14 DPC to model early-gestation IUHCT, after birth at 20 DPC to model late-gestation IUHCT, and in adulthood at 6 weeks of age to model postnatal HSC transplantation (HSCT). TCD BM harvested from B6GFP donors was injected alone or in combination with T cell–depleting antibodies (either anti-CD3, anti-CD4, or anti-CD8 mAbs). (B) Comparison of engraftment and prevalence of CD4+ T cells in uninjected littermates. “Engrafted” was defined as chimerism >1% at 24 weeks after injection. All the litters included in this study had sufficient circulating T cells to reject the allograft (ie, exceeded the threshold of immunocompetence). (C) Kaplan-Meier curve of persistent alloengraftment. The fraction of injected animals with chimerism >1% at each time point is shown. Injection of TCD BM + anti-CD3 mAb at 20 DPC results in sustained alloengraftment at 6 months in all animals, comparable with that observed after injection of TCD BM only at 14 DPC. (D) PB donor cell chimerism over time. The trajectory and magnitude of chimerism were equal between mice injected at 20 DPC with TCD BM + anti-CD3 mAb and 14 DPC with TCD BM only. (E) Quantitative T-cell recovery. Mice injected at 20 DPC with TCD BM + anti-CD3 mAb demonstrated T-cell recovery to levels comparable with those in uninjected controls at 3 weeks of age. Dual-lineage (host- and donor-derived) T-cell recovery was observed. (F) Multilineage analysis. Donor cell engraftment was observed among BM LSK cells (a population enriched for HSCs), with reconstitution of all leukocyte lineages, including T cells, B cells, monocytes, and macrophages. Comparable engraftment and multilineage reconstitution were observed between mice injected at 20 DPC with TCD BM + anti-CD3 mAb and mice injected at 14 DPC with TCD BM only. Mice injected at 20 DPC with TCD BM + anti-CD3 mAb demonstrated no signs of GVHD, as demonstrated by normal weight gain (G), phenotype scores (H), and hematoxylin and eosin (H&E) histology of GVHD-prone organs captured at 10× original magnification (I). ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001.
Transient T-cell depletion restores long-term multilineage engraftment after neonatal transplantation. (A) Experimental design. Mice were injected at 3 developmental stages: before birth at 14 DPC to model early-gestation IUHCT, after birth at 20 DPC to model late-gestation IUHCT, and in adulthood at 6 weeks of age to model postnatal HSC transplantation (HSCT). TCD BM harvested from B6GFP donors was injected alone or in combination with T cell–depleting antibodies (either anti-CD3, anti-CD4, or anti-CD8 mAbs). (B) Comparison of engraftment and prevalence of CD4+ T cells in uninjected littermates. “Engrafted” was defined as chimerism >1% at 24 weeks after injection. All the litters included in this study had sufficient circulating T cells to reject the allograft (ie, exceeded the threshold of immunocompetence). (C) Kaplan-Meier curve of persistent alloengraftment. The fraction of injected animals with chimerism >1% at each time point is shown. Injection of TCD BM + anti-CD3 mAb at 20 DPC results in sustained alloengraftment at 6 months in all animals, comparable with that observed after injection of TCD BM only at 14 DPC. (D) PB donor cell chimerism over time. The trajectory and magnitude of chimerism were equal between mice injected at 20 DPC with TCD BM + anti-CD3 mAb and 14 DPC with TCD BM only. (E) Quantitative T-cell recovery. Mice injected at 20 DPC with TCD BM + anti-CD3 mAb demonstrated T-cell recovery to levels comparable with those in uninjected controls at 3 weeks of age. Dual-lineage (host- and donor-derived) T-cell recovery was observed. (F) Multilineage analysis. Donor cell engraftment was observed among BM LSK cells (a population enriched for HSCs), with reconstitution of all leukocyte lineages, including T cells, B cells, monocytes, and macrophages. Comparable engraftment and multilineage reconstitution were observed between mice injected at 20 DPC with TCD BM + anti-CD3 mAb and mice injected at 14 DPC with TCD BM only. Mice injected at 20 DPC with TCD BM + anti-CD3 mAb demonstrated no signs of GVHD, as demonstrated by normal weight gain (G), phenotype scores (H), and hematoxylin and eosin (H&E) histology of GVHD-prone organs captured at 10× original magnification (I). ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001.
Engrafted recipients demonstrate central tolerance induction with preservation of immune responses to third-party MHC and viral pathogens
We next sought to evaluate whether allogeneic neonatal transplantation with transient TCD achieved reciprocal immune tolerance between host- and donor-derived cells. To do so, we first used the mammary tumor virus (MTV) system to examine the prevalence of T-cell receptor clones specific for nonself antigen. As described in detail in the supplemental Methods, the MTV system allowed us to evaluate the deletion of donor-derived T-cell populations in the B6GFP→Balb/c strain combination. To evaluate the deletion of host-derived T-cell populations, we needed to first repeat our transplantation studies in the reverse (Balb/c→B6GFP) strain combination. We found that neonatal transplantation with transient T-cell depletion in the Balb/c→B6GFP strain combination similarly achieved long-term, multilineage engraftment comparable with fetal transplantation (supplemental Figure 2A-D). However, we noted 2 differences compared with the B6GFP→Balb/c strain combination: (1) an equivalent dose of donor TCD BM achieved half the BM LSK chimerism (supplemental Figure 3A), and (2) B6GFP neonatal recipients developed T cell–dependent allograft rejection responses slightly later than Balb/c recipients (21 DPC vs 20 DPC; supplemental Figure 3B).
After completing transplantation studies in both necessary strain combinations, we then performed MTV clonal deletion analysis. Deletion of T-cell receptor clones specific for nonself antigen was observed in both the donor- and host-derived T-cell populations to a degree comparable with that of fetal transplantation, consistent with the induction of central tolerance (Figure 3A). To determine if this clonal deletion was accompanied by functional tolerance to the donor, skin grafting was performed 24 weeks after injection. Neonates transplanted with coadministration of anti-CD3 mAb showed uniform survival of donor-strain skin grafts (Figure 3B), the histology of which demonstrated normal skin architecture without lymphocyte infiltration (Figure 3C). Notably, comparable skin graft tolerance was observed in the reciprocal strain combination (supplemental Figure 2E-F). To confirm that this donor-specific tolerance was not associated with immunocompromise, we performed in vivo MLR for both the donor and third-party strains, as well as viral inoculation studies. In vivo MLR demonstrated decreased T-cell proliferation in the presence of donor MHC with preservation of normal T-cell proliferation in the presence of third-party MHC, consistent with the induction of donor-specific tolerance (Figure 3D). After inoculation with adeno-associated virus (AAV), a normal anti-AAV humoral response, including immunoglobulin M (IgM) to IgG class switching, was observed among neonates transplanted with anti-CD3 mAb coadministration (Figure 3E). Furthermore, normal anti-AAV T-cell immunity was observed, as measured by interferon gamma enzyme-linked immunospot (Figure 3F). In aggregate, these results demonstrated donor-specific central tolerance induction with the preservation of normal immune development to nonself and viral antigens.
Transient T-cell depletion induces donor-specific central tolerance with preserved T-cell responses to third-party MHC and viral pathogens. (A) T-cell receptor (TCR) deletion analysis. To assess clonal deletion consistent with thymic deletion (central tolerance induction), expression of TCR clones was measured at 24 weeks after injection among donor-derived cells in the B6GFP→Balb/c strain combination and among host-derived cells in the Balb/c→B6GFP strain combination. In both cases, a partial deletion significantly below the baseline frequency in uninjected B6GFP controls was observed in mice receiving TCD BM + anti-CD3mAb. The degree of deletion was comparable with that observed among mice injected at 14 DPC with TCD BM only. (B) Kaplan-Meier curve of skin graft survival. Mice injected with TCD BM + anti-CD3 mAb at 20 DPC demonstrated uniform, long-term survival of donor-derived skin grafts at 28 days postoperatively, comparable with mice injected with TCD BM only at 14 DPC. Mice injected at 20 DPC with TCD BM only uniformly rejected the skin grafts by 12 days postoperatively, comparable with uninjected controls. (C) Skin graft stereomicroscopy and histology. Skin grafts were harvested for stereomicroscopy and H&E histology 28 days postoperatively or on the day of rejection. Donor-strain (B6GFP) skin grafted onto Balb/c mice injected with TCD BM + anti-CD3 mAb at 20 DPC and with TCD BM only at 14 DPC showed de novo hair growth with bright green skin under ultraviolet light. Histologic examination demonstrated normal skin architecture, including hair follicles (green arrows), in the absence of lymphocytic infiltration (red arrows) at the interface between the donor and recipient skin. In contrast, skin grafted onto Balb/c uninjected controls and Balb/c mice injected with TCD BM only at 20 DPC demonstrated erythema, desiccation, and dehiscence of skin grafts with loss of green fluorescence at 11 to 13 days postoperatively, consistent with allograft rejection. Histologic examination demonstrated significant lymphocytic infiltration (red arrows) at the interface between the donor and recipient skin, with neoepithelialization by the recipient underneath the graft (blue arrows). (D) In vivo MLR. T cells harvested from mice injected with TCD BM + anti-CD3 mAb at 20 DPC demonstrated decreased proliferation in the presence of donor strain MHC class I comparable to T cells harvested from mice injected with TCD BM only at 14 DPC. However, they proliferated normally in the presence of third-party MHC class I. This demonstrated that our transient T cell deletion strategy resulted in donor-specific tolerance rather than pervasive immunocompromise. (E) Pathogen humoral immune response. Anti-adeno–associated virus serotype 9 (AAV9) antibodies were measured using enzyme-linked immunosorbent assay 28 days after primary inoculation. Comparable amounts of both IgM and IgG were measured in uninjected controls and mice injected at 21 DPC with TCD BM + anti-CD3 mAb, demonstrating normal humoral immune responses to viral pathogens, including unimpaired B cell class switching from IgM to IgG, which is T-cell-dependent. (F) Pathogen T-cell immune response. Anti-AAV9 T-cell responses were measured using interferon gamma (IFN-γ) enzyme-linked immunospot assay 28 days after primary inoculation. Spleen-derived T cells harvested from mice injected at 21 DPC with TCD BM + anti-CD3 mAb secreted IFN-γ in the presence of the AAV9 peptide library in equal amounts as uninjected controls, demonstrating no long-term detriment in T-cell response to viral pathogens. ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001, not significant (ns). SFU, spot-forming units.
Transient T-cell depletion induces donor-specific central tolerance with preserved T-cell responses to third-party MHC and viral pathogens. (A) T-cell receptor (TCR) deletion analysis. To assess clonal deletion consistent with thymic deletion (central tolerance induction), expression of TCR clones was measured at 24 weeks after injection among donor-derived cells in the B6GFP→Balb/c strain combination and among host-derived cells in the Balb/c→B6GFP strain combination. In both cases, a partial deletion significantly below the baseline frequency in uninjected B6GFP controls was observed in mice receiving TCD BM + anti-CD3mAb. The degree of deletion was comparable with that observed among mice injected at 14 DPC with TCD BM only. (B) Kaplan-Meier curve of skin graft survival. Mice injected with TCD BM + anti-CD3 mAb at 20 DPC demonstrated uniform, long-term survival of donor-derived skin grafts at 28 days postoperatively, comparable with mice injected with TCD BM only at 14 DPC. Mice injected at 20 DPC with TCD BM only uniformly rejected the skin grafts by 12 days postoperatively, comparable with uninjected controls. (C) Skin graft stereomicroscopy and histology. Skin grafts were harvested for stereomicroscopy and H&E histology 28 days postoperatively or on the day of rejection. Donor-strain (B6GFP) skin grafted onto Balb/c mice injected with TCD BM + anti-CD3 mAb at 20 DPC and with TCD BM only at 14 DPC showed de novo hair growth with bright green skin under ultraviolet light. Histologic examination demonstrated normal skin architecture, including hair follicles (green arrows), in the absence of lymphocytic infiltration (red arrows) at the interface between the donor and recipient skin. In contrast, skin grafted onto Balb/c uninjected controls and Balb/c mice injected with TCD BM only at 20 DPC demonstrated erythema, desiccation, and dehiscence of skin grafts with loss of green fluorescence at 11 to 13 days postoperatively, consistent with allograft rejection. Histologic examination demonstrated significant lymphocytic infiltration (red arrows) at the interface between the donor and recipient skin, with neoepithelialization by the recipient underneath the graft (blue arrows). (D) In vivo MLR. T cells harvested from mice injected with TCD BM + anti-CD3 mAb at 20 DPC demonstrated decreased proliferation in the presence of donor strain MHC class I comparable to T cells harvested from mice injected with TCD BM only at 14 DPC. However, they proliferated normally in the presence of third-party MHC class I. This demonstrated that our transient T cell deletion strategy resulted in donor-specific tolerance rather than pervasive immunocompromise. (E) Pathogen humoral immune response. Anti-adeno–associated virus serotype 9 (AAV9) antibodies were measured using enzyme-linked immunosorbent assay 28 days after primary inoculation. Comparable amounts of both IgM and IgG were measured in uninjected controls and mice injected at 21 DPC with TCD BM + anti-CD3 mAb, demonstrating normal humoral immune responses to viral pathogens, including unimpaired B cell class switching from IgM to IgG, which is T-cell-dependent. (F) Pathogen T-cell immune response. Anti-AAV9 T-cell responses were measured using interferon gamma (IFN-γ) enzyme-linked immunospot assay 28 days after primary inoculation. Spleen-derived T cells harvested from mice injected at 21 DPC with TCD BM + anti-CD3 mAb secreted IFN-γ in the presence of the AAV9 peptide library in equal amounts as uninjected controls, demonstrating no long-term detriment in T-cell response to viral pathogens. ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001, not significant (ns). SFU, spot-forming units.
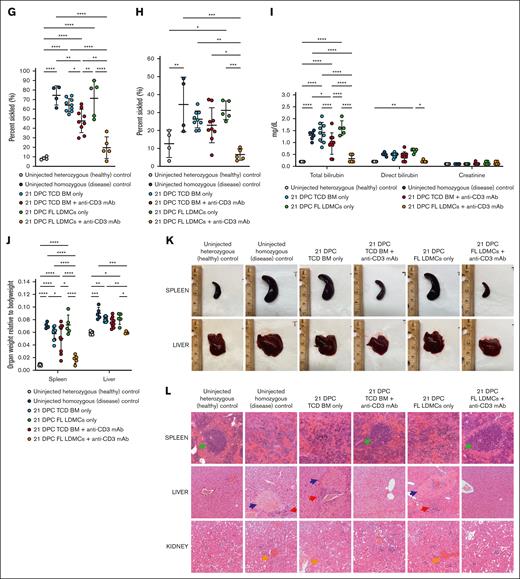
Neonatal transplantation achieves engraftment in a murine model of SCD without myeloablation or boosting
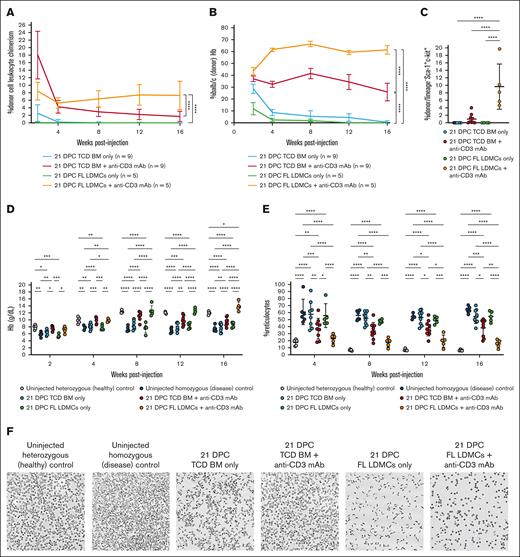
Having demonstrated tolerance and engraftment in allogeneic wild-type strain combinations, we next tested our strategy using the Townes mouse model of SCD. Transplantation of allogeneic Balb/c TCD BM plus anti-CD3 mAb into homozygous Townes neonates at 21 DPC resulted in down-trending, low-level leukocyte chimerism (mean, 1.7%; Figure 4A), and erythrocyte engraftment (mean, 25.9%) at 16 weeks after injection (Figure 4B). This was consistent with the observed low-level donor chimerism among BM LSK (mean, 1.0%; Figure 4C). Partial correction of anemia (Figure 4D) and reticulocyte percentage (Figure 4E) was observed. Ex vivo sickling assays demonstrated a moderate reduction in sickled cells (Figure 4F-G). Sickling after in vivo hypoxia-reoxygenation was similarly reduced (Figure 4H), with an associated reduction in hyperbilirubinemia resulting from erythrolysis (Figure 4I). Consistent with the observed reduction in circulating sickled cells and alleviation of chronic anemia, hepatosplenomegaly was also reduced, with improvement in both organ size (Figure 4J) and gross histologic disease (Figure 4K). Microscopic examination showed reduced severity of the signature features of disease pathology otherwise present in the uninjected homozygous Townes controls, including massive expansion of the red pulp with complete loss of lymphoid follicular structure in the spleen and multifocal necrosis, hemosiderin deposition, and extramedullary hematopoiesis in the liver (Figure 4L). In aggregate, these results showed some therapeutic benefit for most neonatal transplant recipients but not the high-level, durable engraftment required for a consistent clinical cure.
Transient T-cell depletion performed in the Townes model of SCD. (A) Donor leukocyte chimerism. Transient TCD achieved donor cell chimerism that was higher after transplantation with FL LDMCs than with TCD BM. (B) High-performance liquid chromatography. Shown is the percentage of hemoglobin (Hb) that is derived from Balb/c donor erythrocytes as opposed to the homozygous Townes recipient. A high level %Balb/c (donor) Hb is indicative of a high percentage of donor-derived erythrocytes in the PB. This was observed in some animals injected with TCD BM + anti-CD3 mAb and in all animals injected with FL LDMCs + anti-CD3 mAb. (C) Donor engraftment among BM LSK cells. BM LSK is a population enriched for HSCs. (D) Hb concentration. (E) Reticulocyte percentage. Anemia with an associated elevation in percentage reticulocyte is characteristic of untreated homozygous Townes mice. Both were improved after the injection of TCD BM + anti-CD3 mAb and fully corrected after the injection of FL LDMCs + anti-CD3 mAb. (F) Images of reticulocytes after ex vivo hypoxia. (G) Quantification of sickling after ex vivo hypoxia. Sickling was significantly reduced in neonates injected with TCD BM + anti-CD3 mAb and FL LDMCs + anti-CD3 mAb. (H) Quantification of sickling after in vivo hypoxia-reoxygenation. In vivo sickling was similarly reduced in neonates undergoing immune modulation at the time of transplantation. (I) Serum chemistry after in vivo hypoxia-reoxygenation. Uninjected homozygous mice demonstrate profound hyperbilirubinemia after in vivo hypoxia-reoxygenation due to erythrolysis and impaired liver function. This was significantly improved in the successfully treated mice. (J) Quantification of hepatosplenomegaly (HSM). Injection of FL LDMCs + anti-CD3 mAb prevented HSM in all recipients, whereas injection of TCD BM + anti-CD3 mAb prevented HSM in some but not all recipients. (K) Gross histology of the spleen and liver at terminal analysis. Healthy/corrected mice showed normal spleen and liver sizes with a smooth capsule. (L) Microscopic histology at terminal analysis. Shown are H&E preparations imaged at 10× original magnification. Diseased spleens showed significant expansion of the red pulp with pooling of sinusoidal erythrocytes and total loss of lymphoid follicular structure (green arrows). Healthy/corrected spleens showed normal splenic red and white pulps without pooling of sickle erythrocytes or infarcts. Diseased livers show focal areas of necrosis (blue arrows) and congestion of the intrahepatic vasculature with aggregates of sickled red blood cells (RBCs). Erythroid progenitors are evident in the sinusoid, consistent with extramedullary hematopoiesis (red arrows). There was also hemosiderin deposition resulting from Kupffer cell phagocytosis. In contrast, focal areas of necrosis and aggregation of sickled erythrocytes were not observed in healthy/corrected livers, and extramedullary hematopoiesis and hemosiderin deposition were absent. Diseased kidneys show engorgement and occlusion of blood vessels with sickled erythrocytes (orange arrows), resulting in vascular, tubular, and glomerular changes. Healthy/corrected kidneys appeared normal and free of disruptive vascular RBC pooling and hemosiderin deposition. ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001.
Transient T-cell depletion performed in the Townes model of SCD. (A) Donor leukocyte chimerism. Transient TCD achieved donor cell chimerism that was higher after transplantation with FL LDMCs than with TCD BM. (B) High-performance liquid chromatography. Shown is the percentage of hemoglobin (Hb) that is derived from Balb/c donor erythrocytes as opposed to the homozygous Townes recipient. A high level %Balb/c (donor) Hb is indicative of a high percentage of donor-derived erythrocytes in the PB. This was observed in some animals injected with TCD BM + anti-CD3 mAb and in all animals injected with FL LDMCs + anti-CD3 mAb. (C) Donor engraftment among BM LSK cells. BM LSK is a population enriched for HSCs. (D) Hb concentration. (E) Reticulocyte percentage. Anemia with an associated elevation in percentage reticulocyte is characteristic of untreated homozygous Townes mice. Both were improved after the injection of TCD BM + anti-CD3 mAb and fully corrected after the injection of FL LDMCs + anti-CD3 mAb. (F) Images of reticulocytes after ex vivo hypoxia. (G) Quantification of sickling after ex vivo hypoxia. Sickling was significantly reduced in neonates injected with TCD BM + anti-CD3 mAb and FL LDMCs + anti-CD3 mAb. (H) Quantification of sickling after in vivo hypoxia-reoxygenation. In vivo sickling was similarly reduced in neonates undergoing immune modulation at the time of transplantation. (I) Serum chemistry after in vivo hypoxia-reoxygenation. Uninjected homozygous mice demonstrate profound hyperbilirubinemia after in vivo hypoxia-reoxygenation due to erythrolysis and impaired liver function. This was significantly improved in the successfully treated mice. (J) Quantification of hepatosplenomegaly (HSM). Injection of FL LDMCs + anti-CD3 mAb prevented HSM in all recipients, whereas injection of TCD BM + anti-CD3 mAb prevented HSM in some but not all recipients. (K) Gross histology of the spleen and liver at terminal analysis. Healthy/corrected mice showed normal spleen and liver sizes with a smooth capsule. (L) Microscopic histology at terminal analysis. Shown are H&E preparations imaged at 10× original magnification. Diseased spleens showed significant expansion of the red pulp with pooling of sinusoidal erythrocytes and total loss of lymphoid follicular structure (green arrows). Healthy/corrected spleens showed normal splenic red and white pulps without pooling of sickle erythrocytes or infarcts. Diseased livers show focal areas of necrosis (blue arrows) and congestion of the intrahepatic vasculature with aggregates of sickled red blood cells (RBCs). Erythroid progenitors are evident in the sinusoid, consistent with extramedullary hematopoiesis (red arrows). There was also hemosiderin deposition resulting from Kupffer cell phagocytosis. In contrast, focal areas of necrosis and aggregation of sickled erythrocytes were not observed in healthy/corrected livers, and extramedullary hematopoiesis and hemosiderin deposition were absent. Diseased kidneys show engorgement and occlusion of blood vessels with sickled erythrocytes (orange arrows), resulting in vascular, tubular, and glomerular changes. Healthy/corrected kidneys appeared normal and free of disruptive vascular RBC pooling and hemosiderin deposition. ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001.
The observed competitive advantage of healthy donor erythrocytes and variable phenotypic improvement in the SCD model after neonatal transplantation of adult TCD BM with transient TCD was similar to our previously published results after fetal transplantation in an alternative strain combination.1 Considering the lower engraftment observed in the Balb/c→B6GFP and Balb/c→Townes strain combinations compared with B6GFP→Balb/c (supplemental Figure 3A) and previous studies demonstrating low-level engraftment of adult HSCs transplanted into the fetal/neonatal hematopoietic niche,35 we next performed neonatal transplantation with transient TCD using a donor stem cell source that was developmentally similar to the recipient, specifically FL LDMCs. Transplantation of allogeneic Balb/c FL LDMCs plus anti-CD3 mAb resulted in stable, higher-level chimerism among PB leukocytes (mean, 7.3%; Figure 4A), erythrocytes (mean, 61.3%; Figure 4B), and BM LSK (mean, 9.6%; Figure 4C) at 16 weeks after injection, confirming improved engraftment at the level of the HSC. Uniform correction of anemia (Figure 4D) and reticulocyte percentage (Figure 4E) was observed. Ex vivo sickling assays demonstrated a significant reduction in sickled cells after synthetic exposure to hypoxia (Figure 4F-G). After in vivo hypoxia-reoxygenation, in vivo sickling was similarly corrected (Figure 4H), with associated elimination of hyperbilirubinemia resulting from erythrolysis (Figure 4I). Hepatosplenomegaly was also corrected, with normalization both of organ size (Figure 4J) and gross histologic disease (Figure 4K). Microscopic examination showed correction of the multiorgan disease pathology (Figure 4L). Therefore, by all metrics, neonatal transplantation of FL LDMCs plus anti-CD3 mAb achieved successful engraftment 16 weeks after injection in the Townes model without myeloablative conditioning or boosting.
Discussion
IUHCT is an experimental treatment for congenital immune, metabolic, and hematologic disorders with established efficacy in several animal models but with heretofore disappointing clinical results in immunologically normal fetuses. The early acquisition of T-cell immunity in the human fetus and the practical challenges of performing IUHCT before that time have contributed to the lack of clinical success, and it is, therefore, necessary to develop strategies to extend the window of opportunity for IUHCT to later in gestation. In this study, we demonstrated that immune modulation by means of transient antibody-mediated T-cell depletion restores allograft receptivity in a neonatal murine model of LGT, achieving donor-specific central tolerance and long-term HSC engraftment comparable to fetal transplantation without collateral immunodeficiency or GVHD. Furthermore, we demonstrated that this 1-shot approach can also be successful in the Townes mouse model of SCD, achieving engraftment without myeloablative conditioning, secondary transplantation, or boosting.1
T-cell depletion in neonatal mice has been shown to be tolerogenic in other transplant models, including pancreatic islet cell transplantation when paired with intrathymic BM injection.36-38 Relatedly, antibody-mediated T-cell depletion with concomitant BM transplantation (BMT) in adult mice has been shown to result in skin graft tolerance in minor MHC-mismatch models.39 Our work innovates beyond these classic immune studies in important ways. First, we used a validated metric of recipient immunocompetence to ensure that all mice included in our study would have otherwise rejected the allograft if immunomodulation was not performed. This is supported in the current study by the absence of long-term tolerance and engraftment in neonatal mice with TCD BM or FL LDMCs alone and is concordant with data from our previous study.28 Second, we demonstrated that long-term tolerance and engraftment can be achieved through exclusively intravascular injection rather than through a combination of peripheral T-cell depletion and direct intrathymic antigen injection, validating an approach that is amenable to clinical translation given that IUHCT is performed by intravascular injection into the umbilical cord, similar to in utero transfusion.40,41 Third and most importantly, our study demonstrated tolerance and engraftment after neonatal transplantation in major MHC-mismatch strain combinations without any myeloablative conditioning or boosting. We observed comparable engraftment after fetal vs neonatal transplantation despite injecting donor cells after the migration of the hematopoietic niche from the FL to the BM, which occurs at 17 DPC in mice.42 When the critical immune barrier posed by mature T cells was eliminated, the early BM hematopoietic niche at 20 DPC proved to be as receptive to donor HSC engraftment as the FL at 14 DPC. This window of opportunity for sustained engraftment without myeloablation was unique to the neonatal period, however. A weight-equivalent dose of TCD BM plus anti-CD3 mAb administered at 6 weeks of age resulted in rapid allograft rejection without tolerance induction despite successful depletion of peripheral T cells. In this way, the neonatal period in mice appears to be a unique window of opportunity for both nonmyeloablative engraftment and targeted immune reprogramming.
Elegant studies by Shaaban at al43-46 have demonstrated the importance of natural killer (NK) cells for sustained tolerance and engraftment after IUHCT and delineated the minimum circulating hematopoietic chimerism that must be achieved before NK cell maturation (≥1.8%). NK cells do not express cell-surface CD347; therefore, we did not evaluate their depletion after anti-CD3 mAb conditioning. However, chimerism at terminal analysis at 24 weeks after injection was ≥1.8% in 9 of 10 mice (90%) injected with TCD BM plus anti-CD3 at 20 DPC and in 10 of 10 mice (100%) injected with TCD BM only at 14 DPC (P = 1.0). This suggests that achieving NK cell tolerance in our late-gestation model does not require the direct modulation of NK cells. Prevention of early graft rejection by T cells resulting in sustained antigen presentation is sufficient.
Although the ontogeny of allograft rejection responses in humans has not been studied systematically, we can reasonably approximate a timeline by combining seminal studies of developmental immunology with historical experience with clinical IUHCT. Mature T cells that proliferate in response to nonself antigens emerge in the PB of the human fetus as early as 14 weeks gestation.17,18,48 Early attempts at clinical IUHCT in immunologically normal fetuses typically failed after this gestational age,13,15,20 although more recent efforts suggest that the opportunity for tolerance induction fades gradually beyond this point.14 In sheep, this apparent relationship between T-cell development and transplant outcome has been demonstrated more directly; allograft rejection corresponds closely with the emergence of CD4+ T cells in the PB.19 Having shown this to also be the case with mice,28 we believe the neonatal mouse studies presented here are a reasonable model48 for clinical IUHCT when intravascular injection becomes technically feasible at ∼20 weeks gestation.40,41 However, because of differences between humans and mice in the timing of T-cell emergence (including Tregs) relative to birth, our mouse model of LGT requires postnatal injection. This is problematic because it fails to capture the important interactions between the maternal and fetal immune systems during normal pregnancy and after fetal intervention.32,49-53
Suppressive T-cell populations, including traditional CD4+CD25+FoxP3+ Tregs, interleukin-10-secreting type 1 regulatory T cells, and NK T cells (NKT cells), have been implicated in tolerance at the maternal-fetal placental interface in normal and pathologic pregnancy49,50,54 and reciprocal tolerance induction after fetal and adult hematopoietic cell transplantation.55-57 We have demonstrated the contribution of peripheral Treg populations to the induction of donor-specific tolerance and prevention of GVHD after fetal and neonatal transplantation.28,58 Others have elucidated the importance of fetal Tregs for tolerance to noninherited maternal antigens that cross the placenta50 and the role of NKT cells in conserving pregnancy by regulating immune responses.59,60 As CD3-expressing populations, both fetal Tregs and NKT cells were vulnerable to collateral depletion by our antibody conditioning, and their depletion could have detrimental effects on human pregnancy. Although our analysis of tolerance induction demonstrated that reciprocal tolerance between host and donor was successfully induced despite collateral transient depletion of suppressive T-cell subsets, our study does not answer the major question of whether antibody condition would harm reciprocal tolerance between mother and fetus in human pregnancy. It will be critical, therefore, to test our approach in a large animal model in which the fetus produces mature T-cell populations, including Tregs and NKT cells, while still in utero2,19,61 to assess the breakdown of reciprocal maternal-fetal tolerance and associated adverse pregnancy outcomes.
Some indications of the potential safety of transiently T cell depleting the human fetus can be seen in clinical experience with SCID, an inborn absence of fetal T cells. Infants with SCID are generally healthy at birth, with protection from external pathogens conferred by maternal IgG transmitted during gestation.62 They also generally do well with BMT performed either in the early postnatal period63,64 or in utero.9 Beyond the risk of a transient T cell-deficient state, however, consideration must also be given to the potential toxicity of the conditioning agent itself. T cell-depleting antibodies, such as polyclonal antithymocyte globulin (ATG) and monoclonal Muromonab-CD3 (OKT3), are used as induction or acute rejection therapy for organ transplantation to induce lymphocyte depletion by complement-dependent cytolysis or the prevention of proliferation. Both can provoke the release of inflammatory cytokines with influenza-like symptoms and in rare cases, distributive shock.65,66 Studies demonstrating the absence of adverse fetal outcomes observed after maternal antibody-mediated cytolysis of allotransplants53 or exogenous antibody-mediated depletion of fetal HSCs67,68 are reassuring in this regard, whereas previous studies of alternative fetal immune modulation strategies for IUHCT, including maternal busulfan administration69 and primed donor lymphocyte infusion,70 demonstrate the potential for decreased fetal survival with aggressive conditioning regimens. As such, it will be necessary to closely evaluate cytokine release affecting the placenta or fetal hemodynamics in large animal studies of transient T-cell depletion.
The level of long-term HSC chimerism achieved by the transplantation of adult TCD BM in the various strain combinations was below the level expected to achieve a cure in α and β hemoglobinopathies (10%-20%).71,72 Although tolerance was the primary focus of this study, competition for donor HSCs within the recipient niche is an equally important consideration for the successful clinical translation of IUCHT. Several approaches to increase donor engraftment have recently been reported, including mobilization or elimination of recipient HSCs,31,67,68,73 optimization or enrichment of the donor inoculum,31,74 low-dose myeloablative or immune-mediated boosting in adulthood,70,75 and the use of a donor stem cell source (FL or cord blood) that is developmentally similar to the fetal recipient and may engraft better than adult BM cells.11,35,76-78 Subsequent studies combining our immune-modulating strategy with these methods of engraftment boosting are necessary before large animal and clinical applications. As a start, we here combined transient T-cell depletion with transplantation of a donor stem cell source that was developmentally similar to the host (ie, FL LDMCs). This resulted in sufficient HSC and erythrocyte chimerism to prevent both anemia and multiorgan microvascular injury in the Townes SCD model at a relatively short time point of 16 weeks of age. The clinical relevance of murine FL LDMC transplantation as a corollary of clinical IUHCT using human FL or cord blood cells is controversial, and the use of banked cells poses potential challenges (low availability, ethical considerations, high contamination rates during tissue retrieval, and nonrenewable sources for potential booster therapy). However, the clinical use of cord blood and FL cells as donor sources has been suggested in the past,77,78 and indeed the first reported case of successful IUHCT used FL cells.12 Regardless, the immune-modulating strategy reported here is compatible with any of the myriad engraftment-boosting strategies reported in recent literature and can be combined in future studies to ensure adequate resolution of both the tolerance and engraftment barriers to the successful clinical translation of IUHCT.
The slowly down-trending chimerism observed among Balb/c recipients of adult B6GFP TCD BM and Townes recipients of adult Balb/c TCD BM were equally present in neonatal and fetal transplant recipients. This has been noted in previous studies of allogeneic IUCHT,73 although never definitively explained. It is unique to allogeneic IUHCT and is not observed in congenic models,21 and it is only present in certain strain combinations (eg, we observed stable chimerism in B6GFP recipients of adult Balb/c TCD BM; supplemental Figure 2C). One explanation for this observation is that it represents chronic, low-level immunologic rejection (ie, that there is partial but not complete deletion of alloreactive host T-cell clones that, over time, destroy the graft despite anergy and Treg-mediated suppression). An alternative explanation is that allogeneic HSCs do not fare as well in the BM microenvironment of another strain as they do in their own strain. A third explanation is that the adult donor-derived HSCs do not fare as well in the fetal microenvironment as the endogenous fetal HSCs, and so over time they are generating less and less of the circulating leukocytes and erythrocytes. It is interesting to note that in our Townes model, transplantation of FL LDMCs did not result in the same slowly down-trending chimerism observed after transplantation of Balb/c adult TCD BM. This suggests that it is more related to the BM niche than an immune phenomenon and that FL HSCs may be better able to overcome the disadvantages of an allogeneic hematopoietic microenvironment than adult-derived cells. A dedicated study of strain-specific differences in the hematopoietic niche, as it applies to murine IUHCT models, is necessary to definitely explain this phenomenon and should be performed in the future.
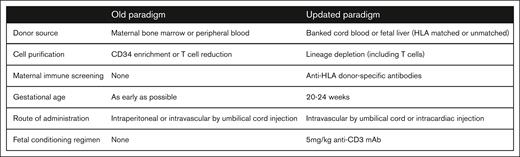
Renewed interest in IUHCT for the treatment of hemoglobinopathies must be considered in the context of recent advances in gene editing. Successful treatment of SCD has recently been reported by ex vivo gene editing of the Bcl11a locus with subsequent autologous transplantation.79,80 Although eliminating the risk of GVHD inherent to postnatal BMT (previously the only curative treatment for SCD), autologous transplantation of ex vivo modified HSCs still requires myeloablative conditioning (typically busulfan), with its associated risks including pancytopenia/infection, hepatic veno-occlusive disease, interstitial pulmonary fibrosis, seizure, and malignancy.81-83 Future work optimizing in vivo therapeutic gene editing will mitigate these risks, but additional safety parameters, including the delivery vehicle and unintended DNA on- and off-target effects, must be considered. In contrast, LGT may not require myeloablative conditioning to achieve therapeutic levels of engraftment when using an HSC source developmentally similar to the recipient.35,84,85 Furthermore, LGT can treat any disease for which postnatal BMT is curative (and perhaps others where postnatal BMT or gene editing is too late to prevent irreversible pathology) without the need for an HLA-matched donor. This includes not only hematologic diseases but also metabolic and potentially neurologic diseases (eg, Krabbe disease).86 For these reasons, we argue that the IUHCT warrants renewed interest, specifically in the context of an updated paradigm of LGT informed by our recent investigations (Figure 5).2,21,53,74
An updated paradigm for IUHCT. After the early successes of IUHCT in severe combined immunodeficiency and bare lymphocyte syndrome, subsequent efforts in immunologically normal fetuses failed to achieve sufficient tolerance and engraftment for therapeutic cure. These setbacks prompted a return to large and small animal studies over the past 2 decades to better understand and circumvent the barriers to successful clinical translation. This has yielded an updated paradigm for the IUHCT to guide renewed efforts.
An updated paradigm for IUHCT. After the early successes of IUHCT in severe combined immunodeficiency and bare lymphocyte syndrome, subsequent efforts in immunologically normal fetuses failed to achieve sufficient tolerance and engraftment for therapeutic cure. These setbacks prompted a return to large and small animal studies over the past 2 decades to better understand and circumvent the barriers to successful clinical translation. This has yielded an updated paradigm for the IUHCT to guide renewed efforts.
In summary, this study demonstrates that immune modulation by transient TCD permits safe and effective allogeneic transplantation in a neonatal mouse model of LGT. This work will serve as a useful foundation for subsequent preclinical and clinical studies aimed to overcome the persistent barrier to the successful clinical translation of IUHCT.
Acknowledgments
The authors thank Aaron Weilerstein for his assistance with animal care and Antonetta Radu for her assistance with histology.
Research reported in this publication was supported by the National Institutes of Health (NIH) Director New Innovator Award (DP2HL152427 [W.H.P.]), generous family gifts to the Center for Fetal Research at the Children's Hospital of Philadelphia (W.H.P.), a Ruth L. Kirschstein National Research Service Award Individual Postdoctoral Fellowship (F32HD106688) from the Eunice Kennedy Shriver National Institute of Child Health and Human Development of the NIH (J.S.R.), and a research scholarship from the American College of Surgeons (J.S.R.).
The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Authorship
Contribution: J.S.R. designed the research study, conducted the experiments, acquired and analyzed the data, and wrote the manuscript; C.L.B., V.L.L., A.D., and M.C.C.-O. conducted the experiments; J.P. and A.W.F. analyzed the data; O.A. conceptualized and designed assays, conducted the experiments, acquired and analyzed the data, and edited the manuscript; and W.H.P. designed the study, analyzed the data, and edited the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: William H. Peranteau, Center for Fetal Research, Children's Hospital of Philadelphia, 3615 Civic Center Blvd, ARC 1114, Philadelphia, PA 19104; email: peranteauw@chop.edu.
References
Author notes
Data are available on request from the corresponding author, William H. Peranteau (peranteauw@chop.edu).
The full-text version of this article contains a data supplement.