Key Points
CAR transgene levels in blood may be associated with long-term responses, but may not predict the relapses in B-ALL and DLBCL.
In DLBCL, BCR was not associated with relapse, whereas in B-ALL, responses were maintained despite late BCR (>6 months).
Visual Abstract
Tisagenlecleucel is a CD19-directed autologous chimeric antigen receptor (CAR) T-cell therapy. Quantitative polymerase chain reaction assays are highly sensitive in defining in vivo kinetics by measuring CAR transgene in peripheral blood. This study aimed to identify clinically meaningful CAR T-cell blood levels that correlated with response/relapse. In pediatric/young adult patients with relapsed/refractory (R/R) B-cell acute lymphoblastic leukemia (B-ALL), maximum CAR T-cell blood levels were higher in patients with ongoing complete remission and in patients with CD19− relapse relative to those with CD19+ relapse. In adult patients with R/R diffuse large B-cell lymphoma (DLBCL), no apparent association between in vivo kinetics and response was noted, with a wide range of transgene levels at relapse. In B-ALL, patients with B-cell aplasia sustained >6 months had higher CAR T-cell expansion relative to those with early B-cell recovery (BCR) (<6 months after infusion); however, a definitive cutoff for BCR-associated expansion level could not be identified. In most patients with B-ALL, BCR >6 months maintained favorable responses. However, early BCR could not be confirmed as a potential indicator of relapse due to high censoring from transplant, following presumed risk of relapse. However, allografting in these patients may potentially mitigate the poor prognosis related to early BCR. In DLBCL, BCR was not associated with relapse. These findings suggest that blood CAR transgene levels may be associated with long-term responses; however, they lack robust predictive potential for relapses. As reported earlier, next-generation sequencing for minimal residual disease appears to be a reliable biomarker predictive of relapse for B-ALL.
Introduction
Tisagenlecleucel is an autologous cellular immunotherapy wherein a patient’s T cells are reprogrammed with a transgene encoding a CD19-specific chimeric antigen receptor (CAR). The in vivo kinetics of the CAR transgene in the blood of patients following tisagenlecleucel infusion can be reliably captured using quantitative polymerase chain reaction (qPCR) and flow cytometry.1-3 qPCR analysis of CAR transgene has generally been more robust and sensitive than flow cytometry assays.1,4 Indeed, cellular kinetics analysis of tisagenlecleucel in peripheral blood has shown shorter periods of persistence when detected using flow cytometry than using qPCR (554 vs 693 days, respectively).1-3
The expansion and persistence of tisagenlecleucel after infusion follows a characteristic biphasic pattern, with an initial peak within the first 28 days after infusion, followed by a decline over time, and persistently low levels of CAR T cells for >2.5 years in some patients. The relationship between transgene expansion, persistence, and clinical response is not completely understood. Additionally, B-cell aplasia, an “on-target” effect of tisagenlecleucel, has been considered a measure of its functional persistence,1-3 and loss of B-cell aplasia (B-cell recovery [BCR]) may indicate a relapse risk. In patients with relapsed or refractory (R/R) B-cell acute lymphoblastic leukemia (B-ALL), Pulsipher et al reported another test predicting relapse risk, which includes the assessment of minimal residual disease (MRD) positivity by next-generation sequencing (NGS).5 Recent studies of adult patients with R/R diffuse large B-cell lymphoma (DLBCL) receiving CAR T-cell therapy have demonstrated MRD monitoring using cell-free circulating tumor DNA as an informative tool in assessing disease relapse or response.6,7
This study determined whether CAR transgene detection by qPCR in the blood is associated with response or relapse, and how CAR levels relate to the duration of B-cell aplasia in pediatric and young adult patients with R/R B-ALL and adult patients with R/R DLBCL.
Methods
Patients
The analyses included pediatric and young adult patients from 2 phase 2, single-arm, multicenter studies on tisagenlecleucel for R/R B-ALL, and adult patients with R/R DLBCL.8-10 Pediatric and young adult patients were included from ELIANA (ClinicalTrials.gov identifier: NCT02435849; N = 79) and ENSIGN (ClinicalTrials.gov identifier: NCT02228096; N = 64).8,9 Patients were aged ≥3 years at screening and ≤21 years at diagnosis, had failed ≥2 prior treatment lines, and were ineligible for allogeneic stem cell transplant, or had relapsed after allogeneic allogenic stem cell transplant. The tisagenlecleucel dose administered was weight-based, with patients ≤50 kg receiving a planned dose of 0.2 × 106 to 5.0 × 106 cells per kg, and patients >50 kg receiving 0.1 × 108 to 2.5 × 108 CAR-positive viable T cells. Adult patients with R/R DLBCL (N = 115) were included from JULIET (ClinicalTrials.gov identifier: NCT02445248).10 Patients were aged ≥18 years, had failed ≥2 prior treatment lines, and had progressive disease after or were ineligible for autologous stem cell transplant. Patients received 0.1 × 108 to 6.0 × 108 CAR-positive viable T cells.
Among patients with B-ALL, the response was defined as complete remission (CR), or CR with incomplete blood count recovery. For patients with DLBCL, the response was defined as CR or partial response, and nonresponders were defined as progressive disease, stable disease, or unknown disease status. Details related to the detection of CAR transgene levels in peripheral blood samples, B-cell levels, MRD levels, and cellular kinetic parameter estimation were included in the supplemental Material. Additionally, details associated with receiver operating curve analyses to identify a cutoff for transgene levels to distinguish the expansion of CAR transgene in responders and nonresponders, along with the related results, have been included in the supplemental Material.
Statistical analysis
Cellular kinetics exposure parameters were estimated with noncompartmental methods using Phoenix WinNonlin, version 6.4 (Pharsight Corp, St. Louis, MO). The effects of cellular kinetics on response were evaluated using summary statistics. Transgene loss (Tloss) was determined as the first available time point after maximum concentration (Cmax) when transgene levels dropped below 50 copies per μg of DNA. Patients in whom the Tloss was not observed were considered to have persistence up to the time to last quantifiable transgene level (Tlast). The details related to statistical analyses performed have been included in the supplemental Material.
The ELIANA, ENSIGN, and JULIET studies were sponsored and designed by Novartis Pharmaceuticals Corporation, and were approved by the institutional review board at each participating institution.
Results
Individual patient cellular kinetic profiles
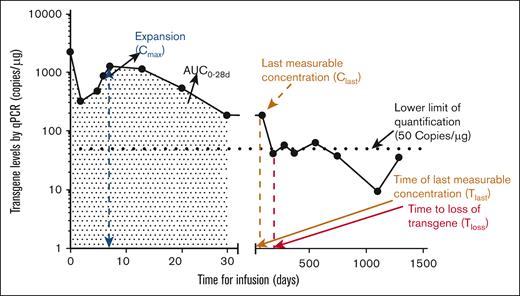
In patients with B-ALL and DLBCL, the expansion and persistence of tisagenlecleucel after infusion exhibit a characteristic biphasic decline pattern, as assessed by the time course of CAR transgene DNA levels in peripheral blood. Figure 1 shows the typical cellular kinetic profile of a patient with B-ALL. The cellular kinetics of transgene expansion typically include an initial peak within the first 28 days after infusion, followed by long-term persistence of CAR T cells, and eventual loss of persistence (transgene levels below the lower limit of quantification based on qPCR assay).
Example profile of CAR transgene expansion and persistence detected by qPCR in patients with B-ALL. Typical cellular kinetics of the tisagenlecleucel transgene expansion profile after infusion in a patient with B-ALL. The shaded area represents area under the curve (AUC) for up to 28 days. Tlast and Tloss denote the time corresponding to the last quantifiable transgene levels (persistence) and loss of transgene, respectively. Nonquantifiable transgene levels (on days 919 and 1451) are not displayed on this semilog plot. Clast, last measurable level of transgene present. Cmax refers to the maximal expansion of CAR transgene level in a patient, and time corresponding to Cmax is denoted as Tmax. The area of exposure up to a certain period can be quantified and denoted as AUC. Tloss was determined as the first available time point after Cmax when transgene levels dropped below 50 copies per μg of DNA. Patients in whom the Tloss was not observed were considered to have persistence up to and time to the last quantifiable transgene level (Tlast).
Example profile of CAR transgene expansion and persistence detected by qPCR in patients with B-ALL. Typical cellular kinetics of the tisagenlecleucel transgene expansion profile after infusion in a patient with B-ALL. The shaded area represents area under the curve (AUC) for up to 28 days. Tlast and Tloss denote the time corresponding to the last quantifiable transgene levels (persistence) and loss of transgene, respectively. Nonquantifiable transgene levels (on days 919 and 1451) are not displayed on this semilog plot. Clast, last measurable level of transgene present. Cmax refers to the maximal expansion of CAR transgene level in a patient, and time corresponding to Cmax is denoted as Tmax. The area of exposure up to a certain period can be quantified and denoted as AUC. Tloss was determined as the first available time point after Cmax when transgene levels dropped below 50 copies per μg of DNA. Patients in whom the Tloss was not observed were considered to have persistence up to and time to the last quantifiable transgene level (Tlast).
Supplemental Figure 1 shows typical scenarios regarding the time course of transgene levels and CD19+ B cells in peripheral blood in individual patients with B-ALL who achieved CR following tisagenlecleucel infusion, as well as an atypical profile for patients who remain in remission despite the loss of transgene and BCR. The horizontal dashed line corresponds to 50 copies per μg transgene levels and 1% CD19+ B cells. Data points below 50 copies per μg or above 1% CD19+ B cells indicate loss of transgene or recovery of B cells, respectively. Differences in the cellular kinetics between responders and nonresponders in patients with B-ALL and DLBCL are presented in the supplemental Material.
Comparison of cellular kinetics in patients with B-ALL who had sustained response vs those who experienced disease relapse
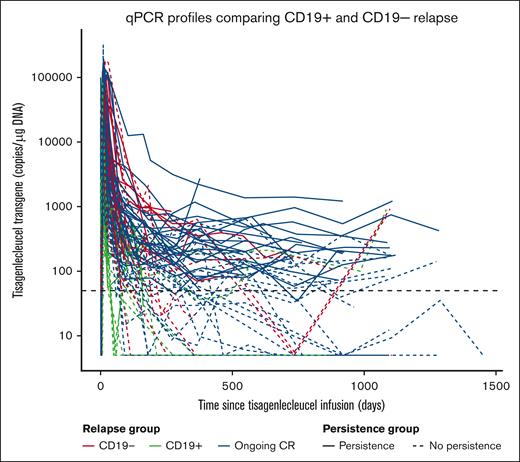
Cellular kinetics parameters were compared among patients with B-ALL who had ongoing CR and those with CD19+ and CD19− relapsed B-ALL, with and without CAR persistence. Notably, persistence was considered lost when transgene levels dropped below the lower limit of quantification of 50 copies per μg after maximum expansion. In patients with B-ALL, the geometric mean Cmax of CAR transgene levels was highest in patients with ongoing CR who demonstrated persistence, and in those with CD19− relapse (geometric mean Cmax: 49 800 copies per μg and 49 300 copies per μg, respectively; Figure 2), and lowest among those with CD19+ relapse (geometric mean Cmax = 12 000 copies per μg, n = 12). Only 2 patients with CD19+ relapse demonstrated persistence (geometric mean Cmax = 5250 copies per μg, n = 2); hence, a comparison of Cmax in this category could not be performed with that in other categories. Notably, in 1 of the 2 patients, relapse was associated with blood, bone marrow, and extramedullary involvement. The other patient’s blood, bone marrow, and central nervous system were not assessed, although extramedullary disease was present. The median Tlast was longest among patients with ongoing CR and continued transgene persistence, and shortest for those who experienced CD19+ relapse and no transgene persistence (733 and 70 days, respectively; Figure 2). Nearly half of the patients with ongoing CR (23/47, 49%) had long-term CAR persistence, whereas the other half showed loss of persistence (24/47, 51%) with a median of 355 days (range, 60-961 days).
Transgene expansion and persistence and response in patients with B-ALL. Tisagenlecleucel transgene levels over time among patients who achieved ongoing CR and among patients who experienced CD19− and CD19+ relapses. The solid transgene curves represent profiles with persistent transgene levels, whereas the profiles with loss of persistence, at some point during follow-up after CAR T-cell therapy, are represented by dashed lines. The thick horizontal black dashed line represents the lower limit of quantification. The geometric mean expansion (n, geometric mean % coefficient of variation) in ongoing CR with persistence, ongoing CR without persistence, CD19− relapse, patients with CD19+ transgene persistence, and patients with CD19+ relapse with no persistence were observed to be 49 800 (n = 23, 95%), 43 600 (n = 24, 204%), 49 300 (n = 25, 110%), 5250 (n = 2, 212%), and 12 000 copies per μg (n = 12, 190%), respectively.
Transgene expansion and persistence and response in patients with B-ALL. Tisagenlecleucel transgene levels over time among patients who achieved ongoing CR and among patients who experienced CD19− and CD19+ relapses. The solid transgene curves represent profiles with persistent transgene levels, whereas the profiles with loss of persistence, at some point during follow-up after CAR T-cell therapy, are represented by dashed lines. The thick horizontal black dashed line represents the lower limit of quantification. The geometric mean expansion (n, geometric mean % coefficient of variation) in ongoing CR with persistence, ongoing CR without persistence, CD19− relapse, patients with CD19+ transgene persistence, and patients with CD19+ relapse with no persistence were observed to be 49 800 (n = 23, 95%), 43 600 (n = 24, 204%), 49 300 (n = 25, 110%), 5250 (n = 2, 212%), and 12 000 copies per μg (n = 12, 190%), respectively.
Transgene levels and B-cell aplasia in patients with B-ALL
B-cell counts in the blood serve as a pharmacodynamic marker for the functional persistence of tisagenlecleucel. Therefore, BCR may indicate a loss of CAR T-cell persistence and an increased risk of relapse. Patients with BCR occurring <3 months, between 3 and 6 months, >6 months, and sustained B-cell aplasia >6 months demonstrated median Tloss (n/N = patients that demonstrated Tloss/total patients in the respective category) of 49 days (7/9 patients), 143 days (14/15 patients), 230 days (19/20 patients), and 361 days (21/57 patients), respectively. Notably, most patients (63%; 36/57) with sustained B-cell aplasia did not demonstrate Tloss.
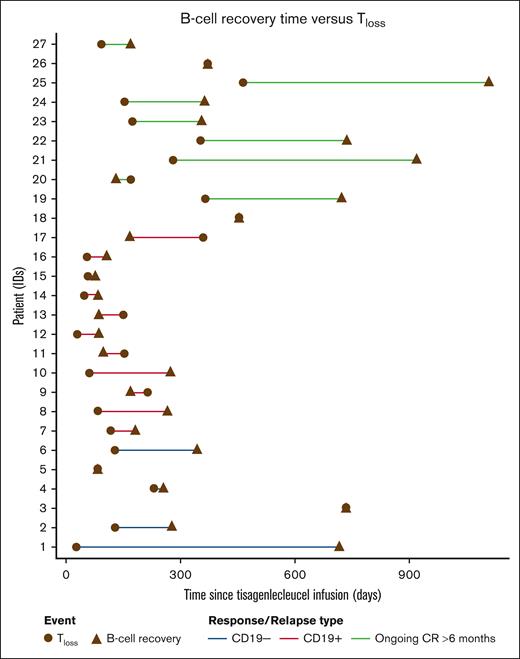
An analysis of the time interval between CAR Tloss and BCR demonstrated that all patients with ongoing CR (10/10 patients) showed loss of transgene at or before BCR (Figure 3). In patients who experienced CD19+ relapse, Tloss and BCR usually occur close in time. However, in patients with ongoing CR and in those with CD19− relapse, some patients demonstrated early loss of transgene and delayed BCR (Figure 3; n = 27, includes only ongoing CR and patients who experienced CD19+/CD19− relapse who demonstrated both loss of transgene and BCR). Among patients in CR with BCR, BCR tended to occur >6 months relative to Tloss (supplemental Figure 2A). In patients who experienced CD19− relapse with B-ALL, Tloss and/or recovery of B cells tended to appear later than observed relapses (supplemental Figure 2B), while in patients with CD19+, both the events were close to each other and usually prior to the relapse (supplemental Figure 2C). These plots indicated that Tloss and BCR are associated with CD19+ relapses, but not with CD19− relapses or with durable responses.
Tisagenlecleucel loss of transgene relative to the time of B-cell recovery in patients with B-ALL. Tloss was determined as the first available time point after Cmax when transgene levels dropped <50 copies per μg of DNA.
Tisagenlecleucel loss of transgene relative to the time of B-cell recovery in patients with B-ALL. Tloss was determined as the first available time point after Cmax when transgene levels dropped <50 copies per μg of DNA.
To understand the relationship between the time of BCR (<3 months, 3-6 months, and ≥6 months) or sustained aplasia and relapse or continued response status, the number of patients in each of the related categories was summarized (supplemental Table 1).
In B-ALL, out of 13 patients who demonstrated BCR >6 months, 9 patients remained in remission for 94 to 926 days after BCR; 6 of those remained in remission it for approximately ≥1 year based on data cutoff date, while 1 patient maintained remission for 94 days after BCR. One patient received new therapy on day 91 after BCR, while 1 patient underwent hematopoietic stem cell transplant (HSCT) before BCR. The remaining 4 patients were censored before BCR for HSCT or new therapy.
In contrast, there were 11 patients with early BCR (<6 months, n = 16), 9 of whom underwent transplant while in CR and censored for HSCT. Additionally, 2 patients were censored for inadequate assessment while in CR (patients missed at least 2 scheduled assessments before the data cutoff date with no other event or censoring reason recorded). One patient was still in CR for up to 559 days (as of the data cutoff date). Finally, 2 of the 16 patients experienced early (<6 months) disease relapse, and 2 patients relapsed after >6 months. These data cannot support or refute early loss of B-cell aplasia as a marker due to the high rates of censoring, as clinicians responding to earlier reports moved patients with early loss of BCR to transplant.
Transgene levels and B-cell aplasia in patients with DLBCL
In patients with DLBCL, the observed recovery of B cells in a limited number of patients was not associated with relapse.9 There was a clear trend toward increasing levels of blood B cells with declining tisagenlecleucel transgene levels.1 In this setting, most patients had ongoing responses despite having BCR within the normal range.
Relationship between persistence of CAR T cells and clinical response (ongoing CR, relapsed, and nonresponding patients)
Cox regression analysis for time to loss of transgene by response categories showed a longer time to loss of transgene in responding patients relative to nonresponding patients in both B-ALL and DLBCL indications. However, there was a difference in follow-up between responding and nonresponding patients, and the nonresponding population among patients with B-ALL was too small to draw strong conclusions. Patients who did not demonstrate loss of transgene (<50 copies per μg) were censored at the time of the last observed sample in this analysis (Figure 4), irrespective of any clinical observation or new therapy, including HSCT. Patients with ongoing CR were less likely to experience CAR Tloss than those without response. Patients with B-ALL who had CD19+ relapse tended to lose the CAR transgene at a similar rate to nonresponders, while those with CD19− relapse tended to lose the CAR transgene at a similar rate to patients with ongoing CR. Among patients with B-ALL (Figure 4A), the hazard ratio of 1.79 (95% confidence interval, 0.64-4.99) suggested an increased risk of CAR Tloss in patients with no response on day 28 relative to responders. Among patients with DLBCL (Figure 4B), an increased incidence of CAR Tloss was observed in patients with no response at month 3 (hazard ratio, 4.19; 95% confidence interval, 2.19-8.02).
Kaplan-Meier analysis of the observed time to Tloss by day 28 response category in patients with B-ALL and month 3 response category in patients with DLBCL. (A) Among patients with B-ALL, the HR for response vs no response (NR) was 1.79 (95% confidence interval [CI], 0.64-4.99). (B) Among patients with DLBCL, the HR for response vs NR was 4.19 (95% CI, 2.19-8.02). For these analyses, the first observed Tloss for a patient was considered as an event, while patients without an observed Tloss were censored at Tlast. Tloss was determined as the first available time point after Cmax when transgene levels dropped below 50 copies per μg of DNA and was considered as an event, while patients with no observed Tloss were censored at the last observation (Tlast). PD, progressive disease; PR, partial response; SD, stable disease; UNK, unknown.
Kaplan-Meier analysis of the observed time to Tloss by day 28 response category in patients with B-ALL and month 3 response category in patients with DLBCL. (A) Among patients with B-ALL, the HR for response vs no response (NR) was 1.79 (95% confidence interval [CI], 0.64-4.99). (B) Among patients with DLBCL, the HR for response vs NR was 4.19 (95% CI, 2.19-8.02). For these analyses, the first observed Tloss for a patient was considered as an event, while patients without an observed Tloss were censored at Tlast. Tloss was determined as the first available time point after Cmax when transgene levels dropped below 50 copies per μg of DNA and was considered as an event, while patients with no observed Tloss were censored at the last observation (Tlast). PD, progressive disease; PR, partial response; SD, stable disease; UNK, unknown.
Patients with B-ALL who had MRD positive by NGS assay had an earlier loss of CAR transgene than those with continued responses >6 months (Figure 5). However, early loss of the CAR transgene was observed in some patients with a continued response. Notably, 51% of patients with ongoing CR maintained their remission despite prior loss of the CAR transgene. The median time to CAR Tloss was longest among patients with ongoing CR, and shortest among patients with no response to tisagenlecleucel (Figure 5). Interestingly, the median time to loss of transgene in patients who experienced CD19− relapse was similar to the median observed in ongoing patients with CR, while patients with CD19+ relapse had a comparatively shorter time to the loss of the CAR transgene.
CD19+ MRD and transgene persistence in patients with B-ALL. Time to loss of transgene for an ongoing response >6 months or CD19 status color coded by day 28 NGS MRD status. Horizontal bars represent median values, and circles represent values outside of 1.5 × interquartile range. Lower and upper whiskers extend to the most extreme points within 1.5 × interquartile range of quartile 1 and quartile 3, respectively. Tloss was determined as the first available time point after Cmax when transgene levels dropped <50 copies per μg of DNA.
CD19+ MRD and transgene persistence in patients with B-ALL. Time to loss of transgene for an ongoing response >6 months or CD19 status color coded by day 28 NGS MRD status. Horizontal bars represent median values, and circles represent values outside of 1.5 × interquartile range. Lower and upper whiskers extend to the most extreme points within 1.5 × interquartile range of quartile 1 and quartile 3, respectively. Tloss was determined as the first available time point after Cmax when transgene levels dropped <50 copies per μg of DNA.
Transgene levels and disease relapse in patients with DLBCL
There was a high variability in transgene levels in patients with DLBCL at the time of relapse, suggesting that transgene levels are not indicative of relapse in these patients. The transgene levels at the time of relapse ranged from 0 to 1330 copies per μg, with a geometric mean value of 190 copies per μg (n = 17, geometric coefficient of variation = 250%).
Duration of B-cell aplasia and relationship with expansion in patients with B-ALL
The expansion levels on day 11, day 28, month 3, and at Cmax by BCR or aplasia were plotted to determine whether a certain threshold of tisagenlecleucel transgene expansion was related to early recovery of B cells. All patients in the sustained B-cell aplasia group had ongoing CR or relapsed after >6 months. A higher proportion (30% vs 11%) of patients who had BCR showed a Cmax of <10 000 copies per μg genomic DNA relative to patients with sustained B-cell aplasia (supplemental Figure 3). However, 6 patients maintained B-cell aplasia despite having Cmax <10 000 copies per μg genomic DNA.
A higher proportion of patients with sustained B-cell aplasia >6 months showed higher transgene levels ie, >10 000, >1000, and >100 copies per μg genomic DNA on day 11, day 28, and month 3, respectively, than patients who had BCR, although no definitive cutoff for the transgene level could be determined (supplemental Figure 3).
Discussion
The qPCR analysis is a highly specific, sensitive, and quantitative analytical assay used to evaluate the in vivo cellular kinetics of CAR T cells and monitor their persistence. In this study, the qPCR assay measured tisagenlecleucel transgene levels in the blood of pediatric and young adult patients with B-ALL and adult patients with DLBCL, aiming to identify its utility in predicting continued responses or relapse. Consistent with previous analyses,1-3 patients with B-ALL who responded following tisagenlecleucel infusion exhibited more rapid and greater in vivo expansion of tisagenlecleucel, and longer persistence than nonresponders. In patients with DLBCL, the maximum CAR transgene expansion and time to maximum expansion were similar between responders and nonresponders; however, tisagenlecleucel persistence was longer in responders. The half-life of tisagenlecleucel was longer in responders than in nonresponders in both B-ALL and DLBCL indications. This suggests that peak in vivo tisagenlecleucel expansion is not necessarily associated with response in patients with DLBCL. By contrast, tisagenlecleucel persistence appears to be an important factor contributing to the durability of response.
As observed in our analysis, patients with sustained response to tisagenlecleucel tend to have persistent CAR transgene levels in the blood. However, in some cases, favorable clinical responses were maintained despite the loss of CAR transgene levels in the blood. Indeed, transgene partitioning from blood to bone marrow has been demonstrated previously, particularly at later time points. Transgene distribution in bone marrow was 44% of that in blood on day 28, and 67% at month 3),1-3 suggesting that CAR T cells may localize to bone marrow (or other extramedullary disease sites) over time and are undetected in the blood.
We observed that among patients with B-ALL, Cmax was higher in patients with ongoing CR with persistent transgene levels and in those with CD19− relapse relative to those with CD19+ relapse. Additionally, transgene levels persisted longer in patients with ongoing CR or CD19− relapse. In patients with DLBCL, a wide range of transgene levels were observed at the time of relapse, suggesting that transgene levels may not be predictive of relapse. This is consistent with the findings from the phase 3 BELINDA study in patients with non-Hodgkin lymphoma treated with tisagenlecleucel.11 The potential reasons for relapses in DLBCL despite quantifiable transgene levels in some patients may be, but not limited to, antigen escape, loss of CAR T-cell function, or tissue/target-site compartmentalization. Previous data in the JULIET study of pre-infusion tumor biopsy samples demonstrated similar response rates between patients with unequivocal CD19 expression and patients with low or negative expression; however, data related to antigen expression at the time of relapse were not available. Additionally, previous analyses have shown that exhaustion of CAR T cells could potentially lead to relapses or lack of responses.10 Schuster et al showed that even though the association between best overall response and average or median programmed cell death protein 1–programmed cell death ligand interaction score or percentage of cells expressing immune checkpoint-related protein from pre-infusion tumor biopsy samples was not apparent, some patients with the highest interaction score or highest percentage of LAG3+ T cells (among total T cells) either did not respond or experienced early relapses within 3 to 6 months.10 To note, the data were limited to draw strong conclusions and might warrant further investigations. Finally, the transgene levels measured routinely in blood in patients with lymphoma may not reflect the transgene levels at the site of action (ie, lymph node or extramedullary tissues); hence, it may be challenging to associate relapses with the persistence of CAR transgene in blood.1
With regard to the maximal expansion differences between B-ALL and DLBCL, the observed transgene Cmax was lower in patients with DLBCL than in those with B-ALL, possibly due to mechanistic differences based on cancer site and/or T-cell intrinsic factors.1 Tumor burden, prior therapy, and T-cell exhaustion may influence CAR T-cell expansion and/or persistence.2,3,12 Furthermore, CAR T cells undergo trafficking to different target sites in patients with DLBCL compared with the trafficking in patients with B-ALL, where the CD19+ target is usually present in the blood. Therefore, qPCR measurements in the blood may not reflect the trafficking of CAR+ T cells to tumor sites outside the blood compartment in patients with DLBCL. These findings underscore the importance of characterizing and understanding the trafficking of the CAR transgene to the tumor site by collecting progression biopsies and biopsies after treatment in patients with DLBCL.
The receiver operating curve on days 7, 11, 14, and 28 yielded an area under the curve (AUC) of nearly 0.5 in patients with DLBCL, indicating that expansion levels during the first 28 days do not distinguish responders from nonresponders (by month 3 response). On the contrary, the area under the curve ranging from 0.37 to 0.87 for expansion levels in patients with B-ALL during the first 28 days indicates the clinical utility of the qPCR analysis as a measure of CAR T-cell expansion levels that distinguishes responders from nonresponders (additional details available in the supplemental Material).
Similar to the analysis of Cmax, higher expansion and persistence of CAR T cells were observed in patients with B-ALL who had ongoing CR and CD19− relapse relative to patients with CD19+ relapse. This observation corroborates with the known mechanism of CD19+ relapse related to the rapid disappearance of CAR T cells.13 Interestingly, nearly half of the patients with ongoing CR had persistent CAR T cells, whereas the other half of the patients remained in remission despite the loss of transgene levels at an earlier time point. On the contrary, 86% of the patients with CD19+ relapse had lost the measurable transgene at the time of relapse. These data suggest that some patients with ongoing CR may maintain durable responses following the loss of transgene, possibly due to the initial expansion and short-term persistence that may have eradicated the tumor.
Currently, BCR is generally used as a clinical indicator for patients with B-ALL who are at relapse risk. A trend for higher expansion and longer persistence was observed among patients with sustained B-cell aplasia relative to patient groups with BCR in <3 months and low CAR transgene expansion.2,3
Some patients with either ongoing CR or CD19− relapses had lost CAR transgenes early after infusion, with a subsequent delay in BCR (>6 months for most patients). For patients with CD19+ relapse, Tloss events tended to occur slightly earlier than BCR, mostly within the first 6 months. Tloss and BCR tended to show a substantial lead time before the relapse; however, the advantage of the slightly earlier lead time offered by Tloss is negated by its lack of specificity, as shown by events (loss of transgene and BCR) occurring in some of the patients with ongoing CR. The data indicate that more than half of the patients with ongoing CR demonstrated durability of response despite the early loss of persistence, while early BCR was observed in almost all of the patients with CD19+ relapse. The mechanism of CD19− relapse is not related to loss of the transgene, and therefore, no consistent pattern was observed regarding loss of transgene or BCR and the time of relapse. The current practice of monitoring B-cell levels within the first 6 months after infusion as a marker for potential CD19+ relapse may be informative, as some patients with early BCR showed CD19+ relapses, and as reported in the literature,14,15 while BCR was not associated with the risk of CD19− relapse.15 However, most patients in our analysis set were censored for additional therapy, including HSCT after BCR. By contrast, no CD19+ relapses were observed in patients with BCR >6 months.
The Kaplan-Meier analysis showed an increased risk of Tloss in nonresponders with B-ALL and patients who relapsed with CD19+ B-ALL relative to patients with ongoing CR and those who relapsed with CD19− disease. In accordance with these results, nonresponders with DLBCL had more frequent Tloss than responders (by response at month 3). Interestingly, geometric mean Cmax and time to reach maximum concentration (Tmax) estimates were similar between responders and nonresponders in patients with DLBCL; however, Tloss vs response analysis showed an association between early loss of the CAR transgene and lack of response. There are a few caveats, such as limited or no cellular kinetics samples being available after relapse, and post hoc categorization of patients based on response following infusion; however, there could be additional underlying factors that may be further explored in the future. Notably, some nonresponders with DLBCL showed Tloss beyond month 3, while some of the responders with DLBCL showed Tloss before month 3 response assessment. Therefore, Tloss may not be utilized as a predictive marker for disease relapse. To note, Tloss was considered an event for these analyses. and the clinical events were not accounted for censoring. Therefore, new anticancer therapy, including HSCT, did not directly impact the analysis, although it can have an indirect impact as it can impact the duration of follow-up for a certain patient.
Measuring MRD using NGS of B-cell and T-cell repertoire allows better detection of leukemia cells than qPCR or multiparametric flow cytometry methods.16 NGS-MRD also enables clonal tracking to identify dominant clonal sequences for MRD tracking, and allows for earlier intervention to prevent relapse. Pulsipher et al reported that the predictive power of NGS-MRD was demonstrated from month 3 after infusion and onward in B-ALL (ELIANA and ENSIGN studies). In this study, all but 1 patient who tested positive using NGS-MRD either relapsed or underwent other anticancer therapies, including HSCT. On the contrary, BCR, which indicates the absence of persisting functional CAR T cells, does not have a high predictive ability with regard to relapse as a CD19– relapse can occur despite B-cell aplasia or presence of CAR transgene.5 Most patients with B-ALL who had MRD positivity by NGS assay showed loss of the CAR transgene at an earlier time than those with continued clinical response. Some patients with continued remission also had an early loss of the CAR transgene. Therefore, a clear cutoff for the time to loss of transgene that distinguished ongoing CR with MRD positive by NGS vs MRD negative CR could not be determined. Therefore, NGS assay is the best biomarker available thus far for assessing relapse risk during the first year following the infusion of CAR T-cell therapy in patients with B-ALL.5
In our study, favorable clinical responses were not observed in 10 patients with B-ALL who experienced early BCR (<6 months), except for 3 patients who had ongoing CR despite the BCR occurring between 3 and 6 months. Conversely, patients with BCR >6 months (n = 13) experienced sustained remission. However, 4 of 13 patients were censored for HSCT or new anticancer therapy. A higher proportion (30% vs 11%) of patients who had BCR showed a Cmax of <10 000 copies per μg genomic DNA relative to patients with sustained B-cell aplasia, indicating the possible utility of measuring CAR transgene levels during the CAR T-cell expansion phase. However, no threshold could be determined, as 6 patients maintained B-cell aplasia despite Cmax values being <10 000 copies per μg. BCR in patients with DLBCL was detected in fewer patients and was not associated with relapse. However, a clear trend toward increasing B-cell levels with declining tisagenlecleucel transgene levels was observed in recent reports.1-3
Most clinicians rely on B-cell aplasia as a marker for functional persistence of CAR T cells. In patients with early BCR (<6 months), a high censoring rate was observed; therefore, our study could not confirm its association with relapse. Continued clinical responses were observed in patients with BCR >6 months; however, an overlapping range of loss of persistence was noted between patients with ongoing CR and CD19+ relapse. Recent research has shown that NGS-MRD positivity (or flow or PCR-based MRD) can be used to predict the probability of relapse after CAR T cells in B-ALL, and perhaps guide the decision whether to consolidate the patient with HSCT. B-cell aplasia is not a robust marker for sustained response in DLBCL, as data from clinical trials have shown that patients may recover B cells in peripheral blood despite maintaining clinical response.1,10,17 These findings improve our understanding of the relationship between tisagenlecleucel transgene levels and B-cell aplasia, and suggest that the in vivo kinetics of the tisagenlecleucel transgene may reflect therapeutic responses in patients with B-ALL and DLBCL, although they may lack the predictive potential to relate transgene levels with clinical responses. Further characterization of tisagenlecleucel cellular kinetics and identification of influencing factors will help improve CAR T-cell therapy outcomes.
Acknowledgments
The authors sincerely thank the patients and their families, as well as the investigators and supporting staff. The authors also thank Eshvendar Reddy Kasala for providing editorial support and assistance with the submission of this manuscript. The authors express their gratitude for the coordination and continued support offered by Avishek Pal in finalizing this manuscript.
This study was sponsored by Novartis Pharmaceuticals Corporation.
Authorship
Contribution: All authors substantially contributed to the conception or design of the work, the acquisition, analysis, or interpretation of data for the work, the drafting of the work or reviewing it critically for important intellectual content, and provided final approval of the version to be published and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Conflict-of-interest disclosure: R.A., A.C., and A.M. are currently employed with Novartis, with stocks/stock options. S.A.G. reports consultancy with Novartis, Vertex, Jazz, Cellectis, Juno, Cabaletta, Adaptimmune, Allogene, and Cellular Biomedicine Group; research funding from Novartis, Vertex, Jazz, Kite, and Servier; and membership on an entity's board of directors or advisory committees for Jazz, Cellectis, Juno, Cabaletta, Adaptimmune, Allogene, and CBMG. E.R.W. reports current employment with Daiichi Sankyo (work was completed while employed by Novartis). C.S.T. reports honoraria from Janssen, AbbVie, BeiGene, Loxo Oncology, and Novartis; and research funding from Janssen, AbbVie, and BeiGene. S.R. reports personal fees from Servier, Celgene/Bristol Myers Squibb (BMS), Kite/Gilead, Amgen, and Novartis; honoraria from Servier, Celgene/BMS, Kite/Gilead, Amgen, Novartis, and OneChain Immunotherapeutics; and consultancy for Novartis, Autolus, and OneChain Immunotherapeutics. J.P.M. reports research funding from Pluristem Therapeutics, Allovir, Kite, Novartis, Fresenius Biotech, Astellas Pharma, Bellicum Pharmaceuticals, and Gamida Cell; and consultancy with Allovir, Kite, Magenta Therapeutics, Juno Therapeutics, and EcoR1 Capital. M.A.P. reports membership on an entity's board of directors or advisory committees for Cargo Therapeutics, Equillium, Medexus, Vertex, bluebird bio, Gentibio, and Novartis. U.J. reports research funding from Innovative Medicines Initiative 2 Joint Undertaking; and honoraria from BMS, Novartis, Gilead, Miltenyi, Janssen, and Roche. G.D.M. reports participation in advisory board, speakers' bureau, and consultancy with Novartis; and research funding from AlloVir. P.B. reports honoraria from BMS and MSD Oncology; and received research funding from MSD Oncology and Takeda. S.J.S. reports consultancy with Novartis, Mustang Bio, Incyte, Caribou Biosciences, Genmab, Kite Pharma, Nordic Nanovector, MorphoSys, Legend Biotech, and Genentech/Roche; honoraria from Novartis and Takeda; patents and royalties with Novartis; and research funding from Merck and Genentech/Roche. M.R.B. reports honoraria from Agios, Incyte, Servier, Sanofi, ADC Therapeutics, Kite/Gilead, BMS, and Novartis; speakers' bureau fees from Agios, Incyte, Servier, Sanofi, and ADC Therapeutics; participation as an advisory board member for Optum Health, Autolus, In8bio, Sana Biotechnology, Chimeric Therapeutics, Arcellx, Achieve Clinics, CRISPR Therapeutics, Novartis, Kite/Gilead, and BMS; and consultancy with Optum Health, Autolus, In8bio, Sana Biotechnology, Chimeric Therapeutics, Arcellx, Achieve Clinics, CRISPR Therapeutics, Novartis, Kite/Gilead, and BMS. A.C. reports current employment with Novartis Institutes for BioMedical Research. A.M. reports current employment with Novartis Pharmaceuticals Corporation. A.B. reports honoraria from AstraZeneca, Clinigen, Jazz, Novartis, and Servier. E.K.W. reports research funding from Secura, NCI R01, Sanofi, BMS, ORCA, Partners Therapeutics, CSL Behring, Verastem, and Novartis; consultancy with CSL Behring, Verastem, Novartis, CRISPR, and Allovir; and current equity holding in private company for Cambium Oncology and Cambium Medical Technologies. The remaining authors declare no competing financial interests.
The current affiliation for E.R.W. is Daiichi Sankyo, Tokyo, Japan.
Correspondence: Rakesh Awasthi, BioMedical Research, Novartis Pharmaceuticals Corporation, A23/05 Building 337, One Health Plaza, East Hanover, NJ 07936; email: rakesh.awasthi@novartis.com.
References
Author notes
A.B. and E.K.W. are joint last authors.
Presented in oral form at the 60th annual meeting of the American Society of Hematology, San Diego, CA, 1 to 4 December 2018, and at the annual meeting of the American Association for Cancer Research, Atlanta, GA, 29 March to 3 April 2019.
Novartis is committed to sharing with qualified external researchers access to patient-level data and supporting clinical documents from eligible studies. These requests are reviewed and approved by an independent review panel on the basis of scientific merit. All data provided are anonymized to respect the privacy of patients who have participated in the trial in line with the applicable laws and regulations. The availability of trial data is according to the criteria and process described at www.clinicalstudydatarequest.com.
The full-text version of this article contains a data supplement.


![Kaplan-Meier analysis of the observed time to Tloss by day 28 response category in patients with B-ALL and month 3 response category in patients with DLBCL. (A) Among patients with B-ALL, the HR for response vs no response (NR) was 1.79 (95% confidence interval [CI], 0.64-4.99). (B) Among patients with DLBCL, the HR for response vs NR was 4.19 (95% CI, 2.19-8.02). For these analyses, the first observed Tloss for a patient was considered as an event, while patients without an observed Tloss were censored at Tlast. Tloss was determined as the first available time point after Cmax when transgene levels dropped below 50 copies per μg of DNA and was considered as an event, while patients with no observed Tloss were censored at the last observation (Tlast). PD, progressive disease; PR, partial response; SD, stable disease; UNK, unknown.](https://ash.silverchair-cdn.com/ash/content_public/journal/bloodadvances/9/17/10.1182_bloodadvances.2024014995/2/m_blooda_adv-2024-014995-gr4.jpeg?Expires=1767143641&Signature=MxujjkC-3Jqqc2uy9OGiNwf5nQYta0Y0JGtsgmtrmOjJSd-sqr61zhlvgt2T3~RRncmcyI2hXocp8RPsmm8ffUZ-NkLQ3gdKbXx8AXHePY2FbER-8-1Jfl96EKQAqaDKqvquYpBftBLaVMRj527yBwePQZU1lEMvRc4EeOeMGdHiLK6nyryoGjJsaLixaVr5c-A-~sYQJ9PyW1ca-w4XEc4Ijve23UHKPo8mTubf00TFFiF5otH9uHAmxau87Ws78ozw7m2vE8vYTKXFeJFAakO8TDPiceCsdRFzAB9JWso55omxXeQnhMY50sBJrSzQpLX6Y~K4YUD0~2fiaVx9LA__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
