Key Points
Our multicenter study correlated sleep study outcomes with health care utilization (emergency visits and admissions) in children with SCD.
We showed that nocturnal hypoxemia and periodic leg movements are associated with increased health care utilization.
Visual Abstract
Sickle cell disease (SCD) is associated with increased health care utilization. Sleep disorders have been linked to increased SCD complications. We aimed to study the association between sleep study outcomes and health care utilization in children with SCD. This is a multicenter retrospective cohort study of children with SCD who underwent polysomnography (PSG) in 3 centers in the United States. We reviewed health care utilization (emergency department [ED] visits, hospital admissions, and total encounters) related to SCD 2 years before and after the PSG. A total of 210 children with SCD from the 3 centers with PSG were included in the analysis. Univariate analysis showed that nocturnal hypoxemia (lower mean oxygen saturation percent [SpO2] and a higher percentage of total sleep time spent with SpO2 <90% [TST90]) and increased periodic leg movement index correlated with increased health care utilization; however, obstructive sleep apnea and arousal index did not. Older age, sickle cell anemia genotypes, hemoglobin level, and hydroxyurea use also correlated with increased health care utilization. After including age, genotype, and hemoglobin in a multivariate linear regression analysis, only percent TST90 maintained a significant association with health care utilization. In conclusion, our study showed that nocturnal hypoxemia (indicated by higher percent TST90) is associated with increased health care utilization (more than doubled) in children with SCD. Our multicenter study determined targetable interventions that could reduce ED visits and hospital admissions in children with SCD. Further studies are needed to confirm these results and study the effect of interventions to treat sleep disorders on health care utilization in SCD.
Introduction
Sickle cell disease (SCD) is the most prevalent hereditary hematologic disorder among individuals of African descent. According to the World Health Organization, its prevalence varies by geographic region and is estimated to affect ∼20 million people worldwide, representing a significant global health burden.1 In the United States, SCD affects ∼100 000 individuals, with >90% being non-Hispanic Black or African American and an estimated 3% to 9% being Hispanic or Latino.2 This group of disorders arises from the inheritance of 2 copies of a mutant β-globin gene, 1 from each parent, forming hemoglobin S (HbS), which is the underlying cause of SCD. The mutation, GAG→GTG, substitutes valine for glutamic acid at position 6 in the β-globin chain of HbA, resulting in HbS formation.3-6 Under hypoxia conditions, the abnormal Hb tetramer can polymerize, altering red blood cell rheology and activating local vascular injury and inflammation, ultimately leading to vaso-occlusion and hemolysis.7-9 The extent of HbS polymerization is a primary determinant of the severity of SCD.10 Tissue hypoxia in individuals with HbS is the primary cause of organ damage, mainly due to vaso-occlusion and the altered affinity of HbS for oxygen, causing the oxyhemoglobin dissociation curve to shift to the right. Therefore, HbS requires higher partial pressures of oxygen to achieve the same Hb saturation level as normal HbA.11
SCD is associated with increased health care utilization in acute care settings, contributing to the high burden of the disease.12-14 Obstructive sleep apnea (OSA) and nocturnal hypoxemia measured via polysomnography (PSG) are more prevalent in children with SCD and have been linked to increased SCD complications.15,16 OSA affects ∼40% of children of African ancestry with SCD when using an obstructive apnea-hypopnea index (oAHI) cutoff of ≥1 in PSG or 10% with a cutoff of ≥5.17 Furthermore, more than one-third of children with SCD experience lower than normal daytime oxygenation, indicated by oxygen saturation percent (SpO2) levels <96%. This condition may arise from complex alterations in lung parenchyma, airways, and vascular systems, leading to impaired gas exchange and ventilation-perfusion mismatch.3,15
Ongoing sickling in children with SCD leads to progressive multiorgan involvement, often resulting in increased morbidity and mortality rates.18,19 Highly vascularized organs, such as the lungs, are particularly affected. Chronic respiratory complications of SCD include alterations in pulmonary vasculature, parenchyma, and airways, with a progressive decline in lung function often beginning in childhood.18 Pulmonary hypertension is a severe complication that occurs more frequently in adults than in children with SCD.20 These pulmonary complications of SCD will ultimately lead to further hypoxemia with increased sickling.21
Although there is growing attention to the detrimental effects of OSA, nighttime hypoxemia, and abnormal sleep quality on patients with SCD, evidence regarding the association between sleep disorders and health care utilization in children with SCD remains limited. In this multicenter retrospective study, we evaluated the association between sleep study outcomes and health care utilization in children with SCD in 3 pediatric pulmonary centers in the United States.
Methods
In this multicenter retrospective cohort study, we evaluated the association between the outcomes of overnight PSG and health care utilization (including emergency department [ED] visits and admissions) in children with SCD. The 3 centers involved in this study are located in the Southeast United States: The University of Alabama at Birmingham (UAB), the University of Florida (UF), and Duke University Hospital (DUH). All centers were accredited by the American Academy of Sleep Medicine (AASM) to perform PSG in children continuously during the study period. The institutional review board approved all studies in each of the 3 institutions and waived the consent forms due to minimal risk to the participants (UAB, IRB-170530002; UF, IRB202103030; DUH, Pro00110256). Based on our previous publication, the collection of standardized PSG and clinical data for children with SCD were already established for the multicenter collaboration.1
We included all sleep studies performed in children and young adults aged 0 to 21 years over the 10-year period from 2012 to 2022. Sleep studies followed the American Academy of Sleep Medicine guidelines and were conducted overnight in a sleep laboratory.22 The 12-channel PSG studies included monitoring of the electroencephalogram, eye movements, limb electromyogram, nasal airflow, oral airflow, chest and abdominal effort, chin electromyogram, electrocardiogram, continuous CO2 monitoring, continuous pulse oximetry, and snoring sensor.
The PSG scoring was based on the newest American Academy of Sleep Medicine guidelines at the time of the study. We extracted the following measurements from the PSGs: mean SpO2, percent of total sleep time spent with SpO2 <90% (TST90), percent of total sleep time spent with CO2 >50 mm Hg, oAHI, periodic leg movement index (PLMI), and arousal index. We defined OSA as oAHI >1 per hour. We considered nocturnal mean SpO2 <90%, percent TST90 >5, and PLMI >5 as abnormal. In patients with multiple sleep studies, we selected the initial sleep study only to avoid the effect of interventions implemented after the PSG that may alter the severity of sleep study abnormalities.
We used the PSG database in each institution to search for PSGs done on children with SCD during the study period. To maximize the capture of pediatric patients with SCD who underwent PSG, we crossmatched the PSG and the SCD databases and searched International Classification of Diseases, 10th Revision (ICD-10) codes related to SCD in the PSG databases. We then reviewed each patient's medical records to identify all ED visits and admissions during the 2 years before and after the PSG.
We documented the date of encounter, discharge, and discharge diagnosis. We collected every patient's baseline and SCD-related characteristics, including age, sex, race, SCD genotype (SS and Sβ0 are considered severe genotypes vs Sβ+ and SC), hydroxyurea use, and the hematologic parameters closest to the sleep study date (Hb and mean corpuscular volume), which were included only if they were within 3 months of the sleep study (before or after).
The pooled data from the 3 centers were tested for normality of distribution and skewness to determine the statistical analysis. We used the mean, standard error of the mean, median, and frequency distribution percentiles as appropriate. We then performed a Pearson r correlation analysis to determine the association of each PSG outcome with the number of ED visits, hospital admissions, and total encounters. We performed a univariate analysis to assess the relationship between each PSG variable and the health care utilization indicators. Subsequently, we conducted a multivariate linear regression analysis to adjust for potential confounders recognized in the univariate analysis and determine the independent effects of covariates after controlling for the confounders, ensuring a more robust and comprehensive evaluation. We developed 4 models for the multiple linear regression analysis, with the first 3 models including each covariate significantly associated with health care utilization, whereas model 4 included all significant covariates. We represented the regression analysis data with the estimate, 95% confidence interval (CI), and P value to show the direction, magnitude, and significance of the association between the dependent and independent variables. We used the Student t test to compare continuous variables between 2 groups. We presented the data as mean ± standard error of the mean as appropriate. We assessed the hypotheses using 2-tailed analyses, with a P value <.05 denoting statistical significance, using GraphPad Prism version 10.4.1 (San Diego, CA).
Results
Baseline characteristics
Of the 210 children with SCD included in the study, 110 were from UAB, 72 were from UF, and 28 were from DUH. The mean age at the time of the sleep study was 9.7 years, with an equal sex distribution, and 80% of the participants had severe HbSS or HbSβ0 genotypes (Table 1).
Correlation of baseline characteristics and health care utilization in the multicenter cohort of children with SCD who underwent PSG
| . | All (N = 210) . | ED visits, r (P value) . | Hospital admissions, r (P value) . | All encounters, r (P value) . |
|---|---|---|---|---|
| Age, mean (SEM), y | 9.7 (0.3) | 0.28 (<.0001) | 0.38 (<.0001) | 0.35 (<.0001) |
| Sex, males (%) | 102 (49) | 0.03 (.66) | 0.04 (.49) | 0.04 (.57) |
| Genotype | ||||
| SS + Sβ0 vs SC + Sβ+, n (%) | 169 (80) vs 41 (20) | 0.14 (<.05) | 0.18 (<.05) | 0.17 (<.05) |
| Hb, mean (SEM) | 8.7 (0.1) | –0.15 (<.05) | –0.22 (<.01) | –0.20 (<.01) |
| MCV, mean (SEM) | 86.1 (1.0) | 0.08 (.29) | 0.13 (.11) | 0.11 (.17) |
| Hydroxyurea use (%) | 132 (63) | 0.10 (.07) | 0.13 (<.01) | 0.17 (<.05) |
| . | All (N = 210) . | ED visits, r (P value) . | Hospital admissions, r (P value) . | All encounters, r (P value) . |
|---|---|---|---|---|
| Age, mean (SEM), y | 9.7 (0.3) | 0.28 (<.0001) | 0.38 (<.0001) | 0.35 (<.0001) |
| Sex, males (%) | 102 (49) | 0.03 (.66) | 0.04 (.49) | 0.04 (.57) |
| Genotype | ||||
| SS + Sβ0 vs SC + Sβ+, n (%) | 169 (80) vs 41 (20) | 0.14 (<.05) | 0.18 (<.05) | 0.17 (<.05) |
| Hb, mean (SEM) | 8.7 (0.1) | –0.15 (<.05) | –0.22 (<.01) | –0.20 (<.01) |
| MCV, mean (SEM) | 86.1 (1.0) | 0.08 (.29) | 0.13 (.11) | 0.11 (.17) |
| Hydroxyurea use (%) | 132 (63) | 0.10 (.07) | 0.13 (<.01) | 0.17 (<.05) |
MCV, mean corpuscular volume; SEM, standard error of the mean.
The children were anemic with microcytosis, and only 63% were using hydroxyurea at the time of the PSG. There were 424 ED visits and 537 admissions, totaling 961 encounters for the study population. For the whole cohort, over the 4-year study period (2 years before and 2 years after the PSG), the average number of ED visits per participant was 2.1 ± 0.2, the number of admissions was 2.6 ± 0.3, and the total number of encounters was 4.6 ± 0.43.
Older age, severe genotype, and lower steady-state Hb levels were associated with increased ED visits, admissions, and total encounters. Hydroxyurea use was associated with increased admissions and total encounters but not ED visits (Table 1).
Sleep study results
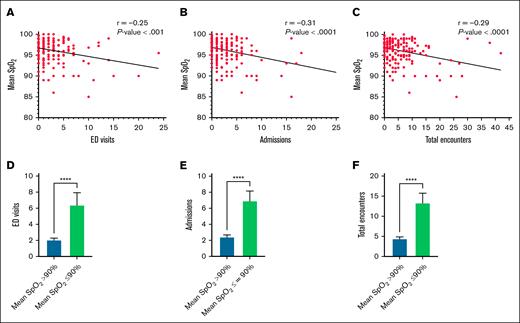
Markers of nocturnal hypoxemia were significantly associated with increased health care utilization. Worse nighttime oxygenation, denoted by lower mean SpO2 during the PSG, correlated with increased number of ED visits (r = –0.25; P < .001), admissions (r = –0.31; P < .0001), and total encounters (r = –0.29; P < .0001; Figure 1A-C; Table 2). The average SpO2 for the cohort was 96.2% ± 0.2%. Fifteen patients (7.1%) had nocturnal mean SpO2 ≤90%. Children with lower nocturnal mean SpO2 (≤90%) had significantly higher ED visits, admissions, and overall encounters than those with higher nocturnal mean SpO2 (>90%; ED visits, 6.4 ± 1.5 vs 2.1 ± 0.2 [P < .0001]; admissions, 6.9 ± 1.2 vs 2.4 ± 0.2 [P < .0001]; total encounters, 13.3 ± 2.4 vs 4.4 ± 0.4 [P < .0001]; Figure 1D-F).
Nocturnal mean SpO2. Scatterplots represent the correlation between the nocturnal mean SpO2 and each of the following: ED visits (A); hospital admissions (B); and total encounters (C). Column bar graphs represent the association of lower vs higher mean SpO2 with each of the following: ED visits (D); hospital admissions (E); and total encounters (F). ∗∗∗∗p < .0001.
Nocturnal mean SpO2. Scatterplots represent the correlation between the nocturnal mean SpO2 and each of the following: ED visits (A); hospital admissions (B); and total encounters (C). Column bar graphs represent the association of lower vs higher mean SpO2 with each of the following: ED visits (D); hospital admissions (E); and total encounters (F). ∗∗∗∗p < .0001.
Correlation of PSG results and health care utilization in the multicenter cohort of children with SCD who underwent PSG
| . | All (N = 210), mean (SEM) . | ED visits, r (P value) . | Hospital admissions, r (P value) . | All encounters, r (P value) . |
|---|---|---|---|---|
| oAHI | 7.6 (2.0) | 0.002 (.98) | –0.002 (.98) | –0.011 (.87) |
| Mean SpO2 (%) | 96.2 (0.2) | –0.25(<.001) | –0.31(<.0001) | –0.29(<.0001) |
| %TST90 | 7.0 (1.3) | 0.32(<.0001) | 0.26(<.001) | 0.31(<.0001) |
| TST >50 (%) | 1.9 (0.7) | –0.079 (.27) | –0.087 (.22) | –0.088 (.21) |
| PLMI mean | 4.2 (1.4) | 0.15(<.05) | 0.19(<.01) | 0.17(<.05) |
| Arousal index | 8.1 (0.6) | –0.089 (.21) | 0.022; (.75) | –0.030 (.67) |
| . | All (N = 210), mean (SEM) . | ED visits, r (P value) . | Hospital admissions, r (P value) . | All encounters, r (P value) . |
|---|---|---|---|---|
| oAHI | 7.6 (2.0) | 0.002 (.98) | –0.002 (.98) | –0.011 (.87) |
| Mean SpO2 (%) | 96.2 (0.2) | –0.25(<.001) | –0.31(<.0001) | –0.29(<.0001) |
| %TST90 | 7.0 (1.3) | 0.32(<.0001) | 0.26(<.001) | 0.31(<.0001) |
| TST >50 (%) | 1.9 (0.7) | –0.079 (.27) | –0.087 (.22) | –0.088 (.21) |
| PLMI mean | 4.2 (1.4) | 0.15(<.05) | 0.19(<.01) | 0.17(<.05) |
| Arousal index | 8.1 (0.6) | –0.089 (.21) | 0.022; (.75) | –0.030 (.67) |
TST>50, percentage of total sleep time with pCO2 >50 mm Hg.
Boldface indicates statistical significance.
A similar trend was noticed with percent TST90, with worse nocturnal oxygenation marked by higher percent TST90 positively correlating with each ED visits (r = 0.32; P < .0001), admissions (r = 0.26; P < .001), and total encounters (r = 0.31; P < .0001; Figure 2A-C; Table 2). The mean percent TST90 for the cohort was 7.0% ± 1.3%. Of the 210 children with SCD and PSG, 54 (25.7%) had nocturnal percent TST90 ≥5% of sleep time. Children with higher nocturnal percent TST90 (≥5% of sleep time) had significantly higher ED visits, admissions, and overall encounters than those with lower nocturnal percent TST90 (<5% of sleep time; ED visits, 4.0 ± 0.8 vs 1.8 ± 0.2 [P < .001]; admissions, 5.0 ± 0.7 vs 2.0 ± 0.2 [P < .0001]; total encounters, 8.9 ± 1.3 vs 3.8 ± 0.4 [P < .0001]; Figure 2D-F).
Percent TST90. Scatterplots represent the correlation between percent TST90 and each of the following: ED visits (A); hospital admissions (B); and total encounters (C). Column bar graphs represent the association of lower vs higher percent TST90 with each of the following: ED visits (D); hospital admissions (E); and total encounters (F). pTST, percentage of total sleep time. ∗∗∗p < .001; ∗∗∗∗p < .0001.
Percent TST90. Scatterplots represent the correlation between percent TST90 and each of the following: ED visits (A); hospital admissions (B); and total encounters (C). Column bar graphs represent the association of lower vs higher percent TST90 with each of the following: ED visits (D); hospital admissions (E); and total encounters (F). pTST, percentage of total sleep time. ∗∗∗p < .001; ∗∗∗∗p < .0001.
We developed 4 models to determine the independent effect of each covariate and the combination of different covariates on health care utilization. Models 1 to 3 included age, genotype, and Hb level, respectively, whereas model 4 included all 3 covariates. Model 1 of the multivariate linear regression analysis showed a significant association between nocturnal mean SpO2 ≤90% and percent TST90 with all indicators of health care utilization (ED visits, admissions, and all encounters) after controlling for age, whereas PLMI did not correlate with the health care utilization anymore (Table 3). After controlling for age, mean SpO2 was negatively associated with total encounters, with an estimate of –0.44 (95% CI, –0.75 to –0.13), whereas percent TST90 was positively associated with total encounters, with an estimate of 0.09 (95% CI, 0.05-0.14). Model 2 (including the genotype) showed a consistently positive coefficient between the 3 significant sleep results and the 3 health care utilization outcomes. After controlling for genotype, mean SpO2 was negatively associated with total encounters, with an estimate of –0.68 (95% CI, –0.99 to –0.37), whereas percent TST90 was positively associated with total encounters, with an estimate of 0.11 (95% CI, 0.06-0.16), and PLMI was also positively associated with total encounters, with an estimate of 0.06 (95% CI, 0.02-0.11). Similar results were reported in model 3 (including Hb), except for the negative association between ED visits and the PLMI after adjusting for Hb. After controlling for steady-state Hb level, the mean SpO2 was negatively associated with total encounters, with an estimate of –0.51 (95% CI, –0.87 to –0.15), whereas percent TST90 was positively associated with total encounters, with an estimate of 0.10 (95% CI, 0.04-0.14), and PLMI was also positively associated with total encounters, with an estimate of 0.05 (95% CI, 0.002-0.10). When including all covariates (age, genotype, and Hb), only percent TST90 was significantly associated with all health care utilization indicators. Percent TST90 was still positively associated with total encounters, with an estimate of 0.07 (95% CI, 0.02-0.12; Table 3).
Multiple linear regression analysis of the association between the PSG results and health care utilization
| . | Model 1 (age), estimate (P value) . | Model 2 (genotype), estimate (P value) . | Model 3 (Hb), estimate (P value) . | Model 4 (all), estimate (P value) . |
|---|---|---|---|---|
| Mean SpO2 | ||||
| ED visits | –0.21(<.05) | –0.30(<.001) | –0.22(<.05) | –0.11 (.29) |
| Admissions | –0.24(<.01) | –0.39(<.0001) | –0.31(<.01) | –0.16 (.12) |
| All encounters | –0.44(<.01) | –0.68(<.0001) | –0.51(<.01) | –0.26 (.17) |
| %TST90 | ||||
| ED visits | 0.05(<.0001) | 0.06(<.0001) | 0.05(<.001) | 0.04(<.01) |
| Admissions | 0.04(<.01) | 0.05(<.0001) | 0.04(<.01) | 0.03(<.05) |
| All encounters | 0.09(<.001) | 0.11(<.0001) | 0.10(<.001) | 0.07(<.01) |
| PLMI | ||||
| ED visits | 0.02 (.23) | 0.02(<.05) | 0.02 (.11) | 0.02 (.11) |
| Admissions | 0.02 (.08) | 0.04(<.01) | 0.03(<.05) | 0.01 (.33) |
| All encounters | 0.04 (.09) | 0.06(<.01) | 0.05(<.05) | 0.02 (.33) |
| . | Model 1 (age), estimate (P value) . | Model 2 (genotype), estimate (P value) . | Model 3 (Hb), estimate (P value) . | Model 4 (all), estimate (P value) . |
|---|---|---|---|---|
| Mean SpO2 | ||||
| ED visits | –0.21(<.05) | –0.30(<.001) | –0.22(<.05) | –0.11 (.29) |
| Admissions | –0.24(<.01) | –0.39(<.0001) | –0.31(<.01) | –0.16 (.12) |
| All encounters | –0.44(<.01) | –0.68(<.0001) | –0.51(<.01) | –0.26 (.17) |
| %TST90 | ||||
| ED visits | 0.05(<.0001) | 0.06(<.0001) | 0.05(<.001) | 0.04(<.01) |
| Admissions | 0.04(<.01) | 0.05(<.0001) | 0.04(<.01) | 0.03(<.05) |
| All encounters | 0.09(<.001) | 0.11(<.0001) | 0.10(<.001) | 0.07(<.01) |
| PLMI | ||||
| ED visits | 0.02 (.23) | 0.02(<.05) | 0.02 (.11) | 0.02 (.11) |
| Admissions | 0.02 (.08) | 0.04(<.01) | 0.03(<.05) | 0.01 (.33) |
| All encounters | 0.04 (.09) | 0.06(<.01) | 0.05(<.05) | 0.02 (.33) |
Model 1 includes age; model 2 includes genotype; model 3 includes Hb; and model 4 includes all significant covariates (age, genotype, and Hb).
Boldface indicates statistical significance.
The degree of OSA, represented by the oAHI, was not correlated with ED visits, admissions, or total encounters (P = .98, .98, and 0.87, respectively; Table 2). There was no difference between patients with OSA and those without OSA regarding the number of ED visits (no OSA, 2.3 ± 0.37; OSA, 1.9 ± 0.26; P = .30), SCD-related hospital admissions (no OSA, 2.3 ± 0.31; OSA, 2.8 ± 0.38; P = .35), or total encounters (no OSA, 4.6 ± 0.65; OSA, 4.5 ± 0.59; P = .94). Increasing total sleep time spent with CO2 >50 mm Hg, denoting worse ventilation, did not correlate with increased ED visits (r = –0.079; P = .27), admissions (r = –0.087; P = .22), or total encounters (r = –0.088; P = .21; Table 2).
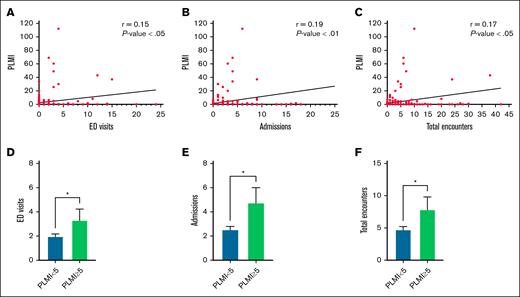
A higher PLMI correlated with more ED visits (r = 0.15; P < .05), admissions (r = 0.19; P < .01), and total encounters (r = 0.17; P < .05; Figure 3A-C; Table 2). The mean PLMI for the cohort was 4.2 ± 1.4 per hour. Of the cohort, 22 (10.5%) had PLMI of ≥5 per hour. Children with higher PLMI of ≥5 per hour had significantly higher ED visits, admissions, and overall encounters than those with lower PLMI of <5 per hour (ED visits, 3.3 ± 0.9 vs 2.0 ± 0.2 [P < .05]; admissions, 4.8 ± 1.2 vs 2.6 ± 0.3 [P < .05]; total encounters, 7.9 ± 2.0 vs 4.8 ± 0.5 [P < .05]; Figure 3D-F). The arousal index did not correlate with ED visits (r = –0.089; P = .21), admissions (r = 0.022; P = .75), or total encounters (r = –0.030; P = .67; Table 2).
PLMI. Scatterplots represent the correlation between the PLMI and ED visits (A), hospital admissions (B), and total encounters (C). Column bar graphs depict the association of lower vs higher PLMI with each of the following: ED visits (D); hospital admissions (E); and total encounters (F). ∗p < .05.
PLMI. Scatterplots represent the correlation between the PLMI and ED visits (A), hospital admissions (B), and total encounters (C). Column bar graphs depict the association of lower vs higher PLMI with each of the following: ED visits (D); hospital admissions (E); and total encounters (F). ∗p < .05.
Discussion
This study highlights the association between nocturnal hypoxemia, as measured by mean SpO2 and percent TST90 during PSG, and increased health care utilization in children with SCD. Our results demonstrate that lower mean SpO2 levels and higher percent TST90 during PSG significantly correlated with more ED visits, hospital admissions, and total clinical encounters. Children with SCD with mean SpO2 levels <90% have more than triple health care encounters than those with higher levels, and those with percent TST90 >5% have more than double health care encounters than those with lower levels. Interestingly, increased PLMI during sleep also correlated with increased health care utilization (those with PLMI >5 have >60% increase in total encounters compared to those with PLMI of <5), whereas OSA, total sleep time spent with CO2 >50 mm Hg, and the arousal index were not. Age, SCD genotype, and baseline Hb level also correlated with health care utilization in our cohort. Adjusting for each of these 3 covariates individually did not significantly affect the association of PSG results and health care utilization, whereas including all of them in a multivariate linear regression showed that higher percent TST90 is the only PSG outcome associated with health care utilization.
Our findings are consistent with previous research showing that nocturnal hypoxemia has a significant impact on clinical outcomes in SCD. For example, Nourani et al demonstrated a direct association between nocturnal hypoxemia and acute chest syndrome (ACS), which is one of the most common causes of hospitalization and death in children with SCD.23 Similarly, in a small study by Campbell et al, the authors identified nocturnal hypoxemia as a risk factor for severe complications, including pulmonary hypertension and central nervous system events, in pediatric patients with SCD. Both studies report that nocturnal hypoxia is associated with increased ED visits related to vaso-occlusive episodes and ACS.24 Further evidence of this relationship is provided by Hargrave et al, who, in a cohort study of 95 pediatric patients, found a significant association between low nocturnal oxygen saturation and the frequency of painful sickle cell episodes (P < .0001).25
Interestingly, although our study did not find a correlation between OSA severity, as represented by the oAHI, and health care utilization, these findings suggest that low nocturnal SpO2 plays a critical role in acute health care use, irrespective of OSA severity. This aligns with findings from Nourani et al, who also reported no significant association between ACS and other sleep-related variables, such as AHI and hypercapnia during sleep.23 In our study, hypercapnia (defined by the percentage of total sleep time with CO2 >50 mm Hg) was not associated with increased health care utilization. On the contrary, Takahashi et al found that OSA was a significant comorbidity associated with more severe complications, specifically the need for mechanical ventilation in children with ACS.26 These contrasting findings suggest that OSA may significantly affect more severe respiratory complications than general health care utilization. OSA may exacerbate the clinical picture under more acute conditions, such as severe ACS, in which mechanical ventilation becomes necessary. Another feasible explanation is that continuous hypoxemia secondary to lung disease may be more harmful in SCD than intermittent hypoxemia seen in OSA.
Additionally, a prospective cohort study of 140 pediatric patients with SCD by Willen et al reported that higher obstructive AHI and oxygen desaturation index were not associated with increased severe pain episodes. Higher nocturnal SpO2 was unexpectedly linked to more pain episodes.27 These findings point to the complexity of the relationship between OSA, sleep-related hypoxemia, and pain episodes in SCD, suggesting that other factors may modulate this interaction and warrant further investigations, because nocturnal oxygen saturation appears to be a crucial factor in the pathophysiology of SCD. One possible mechanism linking hypoxemia to an increased risk of vaso-occlusive pain may involve elevated levels of circulating von Willebrand factor and a higher proportion of high-molecular-weight von Willebrand factor multimers. High-molecular-weight von Willebrand factor, which has procoagulant and proadhesive properties, may contribute to the pathogenesis of pain episodes in patients with nocturnal hypoxemia.28
A novel finding in our study is the significant association between periodic limb movements in sleep and health care utilization, which has yet to be extensively covered in prior literature on pediatric SCD. Periodic limb movements, defined by repetitive, stereotyped movements of the legs or arms during sleep, were measured using the PLMI, which reflects the number of movements per hour of sleep.29 Periodic limb movement disorder is a clinical diagnosis that requires a PLMI of >5 per hour, causing significant sleep disturbances or impairments in daily functioning.29,30 Previous studies found that children with elevated PLMI had a higher prevalence of several comorbid conditions, such as restless legs syndrome, insomnia, mood disorders, and chronic kidney disease.31 Other studies have linked increased PLMI to conditions such as seizure disorders, neurodevelopmental delays, and attention-deficit/hyperactivity disorder.32-34 We found that higher PLMI was correlated with more ED visits, hospital admissions, and total clinical encounters. Lin et al reported an elevated PLMI in 42% of pediatric patients with SCD, whereas we reported 10.5%, which are both higher than the prevalence rates of 1.2% to 8.0% in the general pediatric population.35 Periodic leg movement disorder can be caused by multiple conditions, including mineral deficiencies, medications, neurologic disorders, organ dysfunctions, and sleep disorders.36 Further research is needed to explore the role of sleep disturbances in SCD, particularly considering our finding that the arousal index was not associated with any health care utilization measures, including ED visits, hospital admissions, or total encounters.
Consistent with former studies showing severe SCD genotypes (HbSS and HbSβ0) cause more significant risks for ACS and other vaso-occlusive complications,37,38 we report a strong association between health care utilization and severe SCD genotype. We also report a positive association between older age and increased health care utilization, which seems consistent with reports of increased painful episodes with increasing age.21 Still, these results are inconsistent with the incidence of ACS, which peaks between age 2 and 4 years and generally declines after age 20 years.39 In addition, we found that lower Hb levels are associated with increased health care utilization, which is also consistent with former studies correlating this with disease severity in SCD.40
The multivariate linear regression analysis confirmed the association of each of the 3 PSG results with health care utilization after controlling for genotype and Hb level. However, no significant association between PLMI and health care utilization remained after adjusting for age. When we included all 3 covariates (age, genotype, and baseline Hb level) in the model, only percent TST90 correlated with health care utilization. Therefore, higher percent TST90 correlates best with SCD-related ED visits, admissions, and all encounters. There is an estimated increase by one of each ED visit, admission, and total encounters by increasing percent TST90 by 0.04, 0.03, and 0.07 points, respectively, after controlling for age, genotype, and steady-state Hb level.
Lastly, 63% of the patients in our study were prescribed hydroxyurea. We observed an association between hydroxyurea use, increased admissions, and total clinical encounters. Given the abundant evidence supporting hydroxyurea's role in reducing the frequency of vaso-occlusive episodes, ACS, and the need for blood transfusions, this likely reflects the greater severity of SCD in the patients prescribed hydroxyurea.41-43 In addition, data related to medication adherence and the timing of hydroxyurea initiation were not collected.
This study is limited by its retrospective design, which may introduce biases in data recording and patient selection, because children with SCD referred to the PSG have symptoms of sleep-disordered breathing. In addition, medication adherence and other environmental factors were not assessed. Furthermore, ED visits and admissions of the main 3 centers may not have been counted. Despite the demonstration of a significant association between nocturnal hypoxemia and health care utilization in pediatric patients with SCD, causality cannot be definitively established. Prospective studies with larger sample sizes and randomized interventions targeting nocturnal hypoxemia will be essential to confirm these findings. Because the association between periodic leg movement syndrome and health care utilization lacks a feasible mechanistic underpinning, type 1 error is a possible explanation.
Our study underscores the association of nocturnal hypoxemia and PLMI with health care utilization in pediatric patients with SCD. We demonstrated that lower nocturnal SpO2 levels, mainly higher percent TST90 level, are significantly associated with increased ED visits, hospital admissions, and total clinical encounters after adjusting for confounding covariates. Although periodic leg movement disorder was associated with increased health care utilization, the severity of OSA did not appear to correlate with health care use in this cohort. Further research is warranted to confirm the findings and explore the underlying mechanisms of sleep-related complications in SCD and the potential benefits of targeted treatments. These findings suggest that targeting nocturnal hypoxemia and related sleep disturbances, such as periodic leg movement disorder, through appropriate screening and interventions may help reduce health care utilization and improve outcomes for patients with SCD.
Authorship
Contribution: A.S.A., D.E., and T.A.-D. designed and performed research, collected and analyzed data, and wrote the manuscript; A.R.N. designed research and collected data; and all authors reviewed the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Ammar Saadoon Alishlash, Division of Pediatric Pulmonary and Sleep Medicine, Department of Pediatrics, Heersink School of Medicine, The University of Alabama at Birmingham, 1600 7th Ave, Lowder 620, Birmingham, AL 35233; email: ammarsaadoon@uabmc.edu.
References
Author notes
D.E. and T.A.-D. are joint senior authors.
Original deidentified data are available upon request from the corresponding author, Ammar Saadoon Alishlash (ammarsaadoon@uabmc.edu).