Editor’s Note
Those who were in attendance at the Late-Breaking Abstract Session that took place during the recent ASH annual meeting in New Orleans were treated to six outstanding presentations. Included in this session were two abstracts that reported the long-anticipated discovery of the gene that is somatically mutated in the majority ofJAK2 V617F-negative myeloproliferative neoplasms. The presentations of the two abstracts complemented each other beautifully, and the enthusiasm and sense of joy of the two presenters, Dr. Jyoti Nangalia and Dr. Thorsten Klampfl, reminded those in the audience of the unique rewards of discovery and the pride of accomplishment that naturally follows a notable achievement. Below, Dr. Mark Chao and Dr. Jason Gotlib review the two papers, recently published in theNew England Journal of Medicine, which reported the findings first presented by Dr. Nangalia and Dr. Klampfl at the annual meeting.
Activation of the JAK-STAT pathway is a cardinal feature of he Philadelphia chromosome-negative yeloproliferative neoplasms (MPNs). Dysregulation of this signaling axis can be attributed to the somatic JAK2 V617F mutation in ~98 percent of patients with polycythemia vera (PV), and 50 to 60 percent of patients with essential thrombocythemia (ET) or primary myelofibrosis (PMF). Less commonly, JAK-STAT activation may result from JAK2 exon 12 mutations in 1 to 2 percent of PV cases or mutation of the thrombopoietin receptor, MPL, in 1 to 5 percent of ET and 5 to 10 percent of PMF cases. Rarely, JAK-STAT activation is due to mutations in negative regulators of JAK2, including SH2B3 (LNK) and CBL. Although mutations in tumor suppressors or components of the epigenetic and spliceosome machinery (e.g., TET2, ASXL1, EZH2, DNMT3A, IDH1/2, U2AF1, RSF2, SF3B1, TP53) are found in the chronic or blast phase of MPNs, these mutations do not account for the constitutive JAK-STAT signaling observed in the 30 to 40 percent of MPN patients who are JAK2- or MPL-mutant-negative. With eight years passing since the discovery of JAK2 V617F, the question of whether another highly recurrent mutation would ever be found in MPN patients began to assume the mythical narrative of El Dorado, the Lost City of Gold.
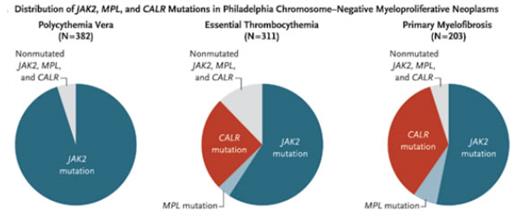
Distribution of JAK2, MPL, and CALR Mutations in Philadelphia Chromosome-Negative Myeloproliferative Neoplasms. From New England Journal of Medicine, Klampfl et al. 369, 2382 and 2383. Copyright © 2013 Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
Distribution of JAK2, MPL, and CALR Mutations in Philadelphia Chromosome-Negative Myeloproliferative Neoplasms. From New England Journal of Medicine, Klampfl et al. 369, 2382 and 2383. Copyright © 2013 Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
Recently, however, two groups of investigators laid claim to the discovery of this elusive treasure. Dr. Jyoti Nangalia and colleagues from the laboratory of Dr. Tony Green at Cambridge, and Dr. Thorsten Klampfl and coworkers from the laboratory of Dr. Robert Kralovics in Vienna, in conjunction with teams of collaborators, used whole-exome sequencing to identify somatic mutations in the calreticulin gene (CALR) in the majority of ET and PMF patients with non-mutated JAK2. From the results of sequencing samples from large numbers of patients with malignant diseases, two important findings emerged: 1) CALR mutations are predominantly found in ET, PMF, and the MDS/MPN overlap disorder, refractory anemia with ring sideroblasts with thrombocytosis (RARS-T), and they are rare or absent in other myeloid or lymphoid neoplasms or solid tumors; and 2) CALR mutations are mutually exclusive of JAK2 and MPL mutations (Figure A). In the study by Dr. Klampfl and colleagues, mutated CALR was found in 67 percent of ET and 88 percent of PMF without JAK2 or MPL mutations. In a similarly designed analysis by Dr. Nangalia and colleagues, 71 percent of ET, 56 percent of PMF, and 86 percent of post-ET MF patients exhibited CALR mutations. The latter study surveyed the genomic landscape of MPNs and found that the median number of somatic mutations in patients with PV, ET, and PMF was 6.5, 6.5, and 13, respectively. The finding of a more complex genotype in myelofibrosis is consistent with its heterogeneous and more aggressive clinical course compared with PV and ET.
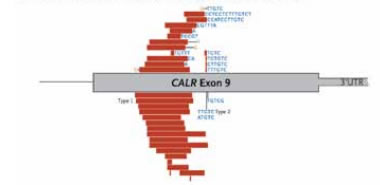
Genomic Position of the 36 Mutation Types Detected in CALR. Genomic positions of mutations in the CALR gene; red bars indicate deletions, blue letters indicate insertions, and orange letters highlight substitutions. From New England Journal of Medicine, Klampfl et al. 369, 2382 and 2383. Copyright © 2013 Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
Genomic Position of the 36 Mutation Types Detected in CALR. Genomic positions of mutations in the CALR gene; red bars indicate deletions, blue letters indicate insertions, and orange letters highlight substitutions. From New England Journal of Medicine, Klampfl et al. 369, 2382 and 2383. Copyright © 2013 Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
Why did it take so long for these scientific explorers to make this seminal discovery? Like the fabled lost city of El Dorado, mutant CALR remained hidden from view. A combination of factors, including low coverage and the presence of a repeat element in the CALR region, and the finding that almost all mutations are insertions or deletions (indels) as opposed to nucleotide substitutions, made CALR genetic abnormalities challenging to find. All CALR indels are localized to exon 9 and generate a 1-base pair (bp) frameshift, resulting in a mutant protein with a novel C-terminus. Within the C-terminus, a large proportion or almost all negatively charged amino acids and calciumbinding sites are lost, and there is also complete loss of the KDEL endoplasmic reticulum retention sequence. More than 80 percent of the more than 30 unique indels that were identified involved either the type I 52-bp deletion or the type II 5-bp insertion (Figure B). Patients with rare homozygous CALR mutations (all with the 5-bp insertion) were also identified. CALR mutations could be identified in the hematopoietic stem cell and progenitor cell compartments, consistent with an early event in MPN pathogenesis. Compared with JAK2 V617F-positive ET and PMF, CALR-mutant ET and PMF are clinically correlated with lower leukocyte and hemoglobin concentrations, higher platelet counts, and a reduced risk of thrombosis. Patients with PMF and mutated CALR exhibited longer survival than JAK2- or MPL-mutated PMF patients; among ET patients, having a CALR mutation was associated with a statistically significant increase in survival compared with JAK2-mutated, but not MPL-mutated, patients.
While CALR mutations represent a prevalent and distinct MPN subtype, the mechanism by which mutant CALR drives neoplastic transformation remains unexplained. In the endoplasmic reticulum, CALR has roles in calcium homeostasis and protein chaperoning. In the cytosol and on the cell surface, CALR has multiple functions including cell adhesion, gene transport to the nucleus, programmed cell removal, and immunogenic cell death.1,2,3 Although mutant CALR has been linked to the pathobiology of various cancers, it is currently unknown if alteration of the above physiologic functions of the protein are relevant to the MPN phenotype. Notably, over-expression of mutant (52-bp deleted) CALR in the lymphoid cell line Ba/F3 resulted in cytokine-independent proliferation and increased STAT5 phosphorylation, suggesting that CALR mutations may function similarly to JAK2 mutations by inducing constitutive STAT signaling. The two reports are mixed with respect to whether there is abnormal localization of mutant CALR to the cytosol and/or cell membrane, the latter of which could have implications for susceptibility to phagocytosis, a process that is partly mediated by a dynamic balance between the pro-phagocytic function of CALR and the anti-phagocytic function of CD47.3,4
In Brief
The discovery of CALR mutations will rapidly be incorporated into clinical practice as a diagnostic marker for ET and PMF and will assist particularly with clarifying the etiology of ambiguous cases of thrombocytosis. While many mutations in CALR have been identified, they all lead to a frameshift that changes the amino acid sequence of the protein product of the mutant gene. Consequently, cells with mutant CALR express a neo-epitope. Targeting of this tumor-specific epitope and inhibition of abnormal CALR function provide attractive therapeutic opportunities. However, caution is warranted when considering CALR inhibition as CALR is essential for normal cellular functions. Indeed, homozygous knockout of CALR in mice is embryonic lethal. Nevertheless, targeted CALR inhibition represents an important and anticipated area of investigation.
References
Competing Interests
Drs. Chao and Gotlib indicated no relevant conflicts of interest.