Allogeneic hematopoietic stem cell transplantation (AHSCT) provides a curative option for high-risk hematologic malignancies as well as many immune disorders and bone marrow failure syndromes. Currently, allogeneic transplantation involves pretransplant conditioning chemotherapy, infusion of donor stem cells, and a recovery period characterized by profound immune suppression, due to both conditioning-related neutropenia and graft-versus-host disease (GVHD) prophylaxis, which primarily targets T-cell function. Febrile episodes in the early post-transplant period are near-universal; therefore, broad-spectrum antibiotic therapy is an inevitable component of allogeneic transplantation. This carries the consequence of significant disruption of the normal intestinal flora.1 Reduced gut microbiome diversity under this selective pressure, combined with dietary changes, prolonged hospitalization, and, typically, antibiotic prophylaxis can lead to the emergence of single-taxon dominance, which has been shown to increase risk of bacteremia and subsequently mortality.2
Earlier work at the Memorial Sloan Kettering Cancer Center identified that microbiota disruption associated with the early use of broad-spectrum antibiotics correlated with worse outcomes in allogeneic transplantation. The occurrence of single-taxon domination, particularly enterococcus, can also be favored by intestinal injury, such as that caused by conditioning and GVHD, and subsequent alterations in lactose metabolism.3 The purpose of this study was to validate the earlier findings in a multicenter conformational study and to prove definitively that despite regional variations in transplantation protocols, fecal microbiome diversity has an impact on transplant mortality.
In this study, Dr. Jonathan Peled and colleagues examined two cohorts from four separate transplant centers: cohort 1 from New York, and cohort 2 from North Carolina, Japan and Germany. They obtained fecal samples from patients prior to transplantation and weekly during the transplantation period, to obtain a median of four samples per patient. The cohort included a total of 1,362 patients for evaluation. The microbiome of fecal samples was analyzed using amplification and sequencing of the genomic 16S ribosomal RNA gene V4-V5 variable region. The intestinal microbiota diversity was established using alpha diversity as calculated by an inverse Simpson index at the level of operational taxonomic units.
The primary outcome was established in both cohorts with patients who had higher (above the median) microbial diversity at the periengraftment period, demonstrating improved survival compared with patients who had lower (below the median) microbiota diversity. The subgroup analysis of cohort 1 showed that the improved in survival in higher diversity group was owing to lower transplant-related mortality and lower GVHD-related mortality. The most common reason for transplantation was for leukemia, and relapse rates were similar between patients with high and low microbiome diversity.
Furthermore, the group then examined pretransplant fecal microbiome diversity in cohort 1 and showed similar results, with better survival outcome seen in patients with higher microbiome diversity. Compared to healthy controls, the pretransplant microbiome diversity is significantly reduced in both cohorts. This reflects the patient population referred for transplantation, as many patients with malignant disorders may have already incurred microbiota damage due to prior chemotherapy, poor oral intake, mucositis, and significant prior broad-spectrum antibiotic exposure. Lower microbiota diversity was associated with an abundance of pathogenic genera including Enterococcus, Klebsiella, Escherichia, Staphylococcus, and Streptococcus. This highlights a strong link between lower microbiome diversity, antibiotic exposure, and antibiotic-resistant organism colonization.
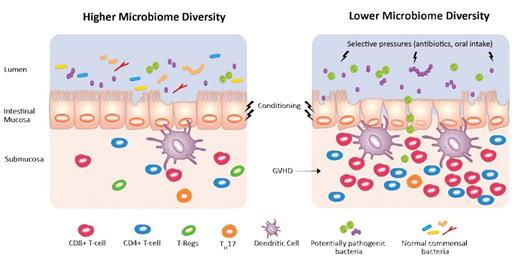
Lower microbiome diversity can be induced by selective pressures including broad-spectrum antibiotics and reduced oral intake. The low diversity state correlates with reduced survival after allogeneic bone marrow transplantation and increased graft-versus-host disease (GVHD) mortality. This is potentially mediated through intestinal inflammation and immune factors such as alloreactivity and T-cell infiltration.
Lower microbiome diversity can be induced by selective pressures including broad-spectrum antibiotics and reduced oral intake. The low diversity state correlates with reduced survival after allogeneic bone marrow transplantation and increased graft-versus-host disease (GVHD) mortality. This is potentially mediated through intestinal inflammation and immune factors such as alloreactivity and T-cell infiltration.
Interestingly, the link between GVHD-related mortality and poor microbial diversity was only seen in recipients of T-cell–replete grafts (compared with CD34-selected stem-cell-only grafts, where no donor T cells are infused). This supports the hypothesis that there is an interplay between the gut microbiome and early T-cell–driven alloreactivity (Figure). Previous studies have shown microbiome diversity may mitigate intestinal inflammation.4
In Brief
This study raises numerous questions. For example, can microbiome diversity be enhanced either prior to or during transplantation? So far, probiotic-based therapies have failed to improve microbiome diversity,5 and while fecal transplantation has been explored as a treatment for the eradication of multidrug resistance bacteria and recurrent Clostridium difficile colitis, including in immunocompromised patients,6,7 it is only in its infancy as a peritransplant therapy.8 Alternatively, a more pragmatic solution may lie in judicious antibiotic selection. Piperacillin/tazobactam- or meropenem-based antibiotic regimens seem to have the most profound effect on microbial diversity, and it may be that avoidance of highly selective regimens may relatively preserve microbiome diversity Altogether, these data provide provocative evidence that low patient microbiome diversity may represent a modifiable risk factor that can be targeted to improve AHSCT outcomes.
Acknowledgment: We thank Dr. Kate Markey for helpful suggestions and Daniela Kolakowski for the illustration.
References
Competing Interests
Drs. Grabek and Lane indicated no relevant conflicts of interest.