Langerhans cell histiocytosis (LCH) is a rare disease that is described in the literature; however, there are very few case reports chronicling the treatment of this disease in hematology/oncology clinics.
The Question
What is a typical management course for LCH from diagnosis through the completion of treatment, including interval scans and surveillance practices.
Case Report
A 29-year-old woman presented to our hospital with worsening complaints of neck stiffness and pain over a period of two weeks. She had an MRI of the brain that revealed mass-like enhancement of the tegmen tympani and reactive dural changes with multiple lytic foci present in the right temporal bone, first cervical vertebral body, and posterolateral wall of the right maxillary sinus wall and mandible. Surgical pathology samples were positive for histiocyte proliferation in a background of eosinophilic and lymphoplasmacytic inflammation consistent with LCH. Immunohistochemical stains demonstrated diffuse positivity for CD1a and S-100. Further staining was positive for Langerin (CD207). After evaluation at the hematology/oncology clinic, the patient underwent a skeletal survey and pan-computed tomography (CT) scan to assess for systemic disease. There was no evidence of lung, liver, or spleen abnormalities. Given her presentation with involvement of craniofacial bones, she was deemed to be at high risk for central nervous system involvement. Thus, the patient was offered outpatient treatment, which she declined. She was subsequently lost to follow-up.
Approximately three years later, the patient was seen in our hematology/oncology clinic for persistent intermittent headaches. CT scans of the head showed progression of Langerhans histiocytosis with increased burden of multifocal lytic calavarial lesions. At this point, the patient was agreeable to systemic therapy. Typically, LCH is treated with prednisone, vinblastine, or cytosine arabinoside (ARA-C). A review of relevant literature indicated the lowest rate of relapse for patients treated with ARA-C.1 Following these data, we recommended the patient undergo six cycles of treatment with ARA-C. Each cycle consists of five days of therapy via intravenous infusion, and the cycle repeats every 28 days.
This treatment lasted approximately nine months owing to cytopenias, and there was an extended delay because of port infection with methicillin-sensitive Staphylococcus aureus (MSSA) bacteremia. Overall, the patient tolerated the treatment well. In the literature, patients treated with prednisone/vinblastine had the highest incidence of grade 3-4 neuropathy and cytopenias. However, even with a regimen less frequently associated with these adverse events, our patient endured severe cytopenias, predisposing her to opportunistic infection such as MSSA.
We performed interval CT scans of the head that revealed interval healing changes in the calavarium at the site of previously seen multifocal aggressive lytic lesions. Additionally, the soft tissue components associated with the previously seen calavarial lytic lesions had decreased in size and were no longer clearly visible. Following cycle 6, we repeated the CT scan of the head, which revealed stable interval changes. There remained lucencies/groundglass calavarial lesions, but these did not progress in size. The patient reported feeling well and had no further headaches, neck pain, ear pain, or jaw pain.
At present, all of her cytopenias have long since resolved, and she reports that she is feeling well.
Surveillance and Follow-Up
In patients with LHC, current standard of care for initial chemotherapy is vinblastine/prednisone in those who require systemic treatment. Other therapies include observation or prednisone therapy alone. The patient described here initially declined chemotherapy but was willing to reconsider it three years after diagnosis owing to persistent pain from the disease. She underwent six cycles of ARA-C, and we continue to perform CT scans every three to six months initially, for a duration of two years or in case she develops recrudescence of symptoms. We attempted to obtain a positron emission tomography (PET)/CT scan, but this was denied by her insurance due to their lack of familiarity with the surveillance of this disease. PET/CT may be beneficial in assessing disease state and response to therapy.2
If the patient should have relapsed disease, we will consider salvage therapies, including cladribine, or clofarabine. These salvage treatments have resulted in high rates of cure among high-risk patients but were associated with prolonged hospitalization and treatment-related death.3
Vemurafenib, a BRAF V600E inhibitor, is another treatment modality to consider for patients with genomic testing that reveals BRAF V600E mutations. The VE-BASKET trial, an open-label, nonrandomized, multicohort study for patients with nonmelanoma cancers carrying the BRAF V600E mutation, showed promising outcomes with vemurafenib treatment.4 LCH patients treated with vemurafenib had improved two-year progression-free survival and overall survival rates. All patients evaluated in the study achieved a metabolic response confirmed on PET scan, and no patient had progression of disease while receiving therapy with vemurafenib.4 Cobimetinib, a MEK-inhibitor, is currently under investigation for patients harboring BRAF mutations. There have been some cases of aggressive LCH successfully treated with allogeneic hematopoietic stem cell transplantation, which could be considered as another treatment modality.5,6
Many patients studied longitudinally after treatment for LCH frequently endorse disabling chronic musculoskeletal pain, fatigue, and mental fogging that diminishes their ability to work and participate fully in their lives. For these patients, we refer them to symptom management with
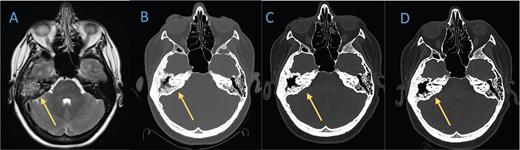
Axial T2 sequence of A) a brain MRI from 2019 demonstrates diffuse opacification of right mastoid air cells and lytic changes of the right temporal bone. A B) head computed tomography (CT) scan obtained in February 2021 shows erosion of the bony septate and partial opacification of the mastoid air cells. C) Nine months later, CT of the head reveals an interval decrease in the amount of fluid collection in the mastoid portion of temporal bone and reactive sclerotic changes. D) The most recent head CT in July 2022 shows stable changes.
Axial T2 sequence of A) a brain MRI from 2019 demonstrates diffuse opacification of right mastoid air cells and lytic changes of the right temporal bone. A B) head computed tomography (CT) scan obtained in February 2021 shows erosion of the bony septate and partial opacification of the mastoid air cells. C) Nine months later, CT of the head reveals an interval decrease in the amount of fluid collection in the mastoid portion of temporal bone and reactive sclerotic changes. D) The most recent head CT in July 2022 shows stable changes.
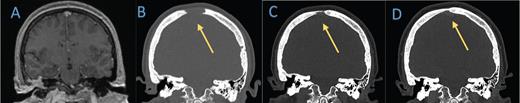
Coronal computed tomography (CT) head bone window demonstrates lytic lesion along the frontal cranium near the vertex which is not evident on the A) brain MRI from 2019. B-D) Subsequent head CTs obtained nine months later show significant interval decrease in size of the lytic lesions. Follow-up head CT in eight months demonstrates further evidence of healing.
Coronal computed tomography (CT) head bone window demonstrates lytic lesion along the frontal cranium near the vertex which is not evident on the A) brain MRI from 2019. B-D) Subsequent head CTs obtained nine months later show significant interval decrease in size of the lytic lesions. Follow-up head CT in eight months demonstrates further evidence of healing.
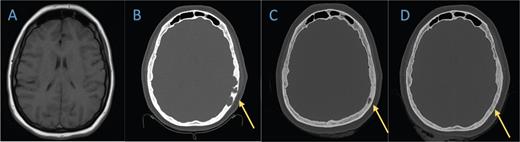
Axial bone window of (B) the head CT in February 2021 shows lytic lesions in the left temporal cranium that is not present on A) the brain MRI from 2019. C) Head CT in November 2021 demonstrates marked improvement of the lytic process. D) The last head CT obtained in July 2022 shows near resolution of the known lesion. palliative care. Early referral for psychological support and symptom management has been a beneficial strategy in many patients.7
Axial bone window of (B) the head CT in February 2021 shows lytic lesions in the left temporal cranium that is not present on A) the brain MRI from 2019. C) Head CT in November 2021 demonstrates marked improvement of the lytic process. D) The last head CT obtained in July 2022 shows near resolution of the known lesion. palliative care. Early referral for psychological support and symptom management has been a beneficial strategy in many patients.7
Discussion
LCH is a rare disease characterized by a clonal proliferation of myeloid dendritic cells.8 These histiocyte cells can accumulate creating inflammatory infiltrates in any organ in the body, though it has a predilection for bones, skin, the lung, and the pituitary gland. It most commonly causes osteolytic bone lesions. Diagnosis is confirmed with histology and immunophenotyping expression of CD1a, S-100, and CD207 (Langerin).9 LCH primarily affects children and is most common in children aged one to three years. LCH is exceedingly rare in adults, with an incidence as miniscule as 0.002%.10 There have been several randomized trials and new treatments in children that have led to improved outcomes; however, there are few studies for therapeutic options in adults.11 There is no well-defined treatment for adults with LCH, and research is currently ongoing for adult treatment option outcomes. Due to the rarity of cases, it is important to follow adult LCH cases to describe diagnosis, treatment, and follow-up outcomes of therapeutic management. Hopefully with more case studies like ours, we can help to reform standard of care for LCH in adults and lead to better overall outcomes.
Competing Interests
Dr. Krishnan, Dr. Kiamos, Dr. Wang, Dr. Dodd, and Dr. Quan indicated no relevant conflicts of interest.