STUDY TITLE: A Study of TAK-755 in Participants with Congenital Thrombotic Thrombocytopenic Purpura
CLINICAL TRIALS.GOV IDENTIFIER: NCT04683003
PARTICIPATING CENTERS: 27 sites throughout North America, Europe, and Asia
ACCRUAL GOAL: 77 patients
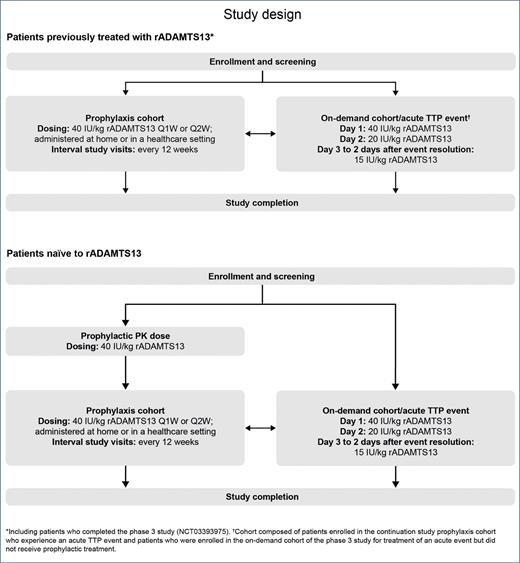
STUDY DESIGN: This phase IIIb, prospective, open-label, multicenter, single-arm, continuation study is evaluating the safety and efficacy of TAK-755 (recombinant ADAMTS13 [rADAMTS13]) in the prophylactic and on-demand treatment of patients with severe congenital thrombotic thrombocytopenic purpura (cTTP). This trial is a follow-up to the pivotal phase III Study 281102 (NCT03393975) and comprises two treatment cohorts (prophylactic and on-demand) that include treatment-naïve and non– treatment-naïve participants with cTTP. A maximum of approximately 77 participants will be enrolled, including roughly 57 non–treatment-naïve participants who have completed Study 281102 and at least 20 participants considered naïve to TAK-755. The study schema is shown in Figure.1
Study design elaborating the approach for patients with cTTP previously exposed to the study drug as well as that for treatment-naïve patients (Figure from Blood)
Study design elaborating the approach for patients with cTTP previously exposed to the study drug as well as that for treatment-naïve patients (Figure from Blood)
This trial has enrolled patients up to age 70 years with cTTP. To date, participants have not displayed any signs of severe thrombotic thrombocytopenic purpura (TTP; defined as a platelet count <100 × 109/L and elevation of lactate dehydrogenase [LDH] >2 times the upper limit of normal [ULN]) at screening in the prophylactic cohort only.
Patients who have been diagnosed with any other TTP-like disorder (or microangiopathic hemolytic anemia), including immune-mediated TTP, are excluded from the trial. Known life-threatening hypersensitivity reactions, including anaphylaxis, to the parent molecule ADAMTS13, hamster protein, or other constituents of TAK-755 also serve as exclusion criteria. Severe genetic or acquired immunodeficiency and organ dysfunction from cardiac, liver, renal, and neurologic events also lead to exclusion.
The main aim of the study is to assess the safety and efficacy profile of the drug in both on-demand and prophylactic settings. The study’s primary outcome measure is the long-term safety and tolerability of rADAMTS13, measured based on the incidence of related treatment-emergent adverse events (AEs) and serious AEs. Secondary outcome measures focus on the efficacy of rADAMTS13, assessed as the incidence of isolated TTP manifestations, along with laboratory parameters and pharmacokinetic and pharmacodynamic data. Researchers also aim to evaluate immunogenicity, health-related quality of life, and healthcare resource utilization in both the prophylactic and on-demand cohorts.
RATIONALE: cTTP, also known as Upshaw-Schulman syndrome, is a rare, life-threatening inherited thrombotic microangiopathy caused by pathogenic variants in the ADAMTS13 gene that leads to undetectable levels of the metalloprotease ADAMTS13.2 Patients with cTTP experience frequent flare-ups of thrombocytopenia and microangiopathic hemolytic anemia, particularly in the neonatal and peripartum periods. Severe exacerbations are infrequent but associated with high mortality without proper management. Traditionally, the cornerstone of treatment is the use of either fresh-frozen or solvent-detergent-treated plasma to provide circulating ADAMTS13.3 rADAMTS13 is being studied for use as a replacement therapy for patients with cTTP.
A phase I study (NCT02216084) in patients with severe cTTP has shown the pharmacokinetic parameters of rADAMTS13 to be comparable with those estimated in previous plasma infusion studies. A phase III study (NCT03393975) to assess the safety and efficacy of rADAMTS13 in patients with severe cTTP was initiated in October 2017 and is ongoing.
COMMENT: cTTP is a rare life-threatening disorder. Until now, therapy options have been limited to plasma replacement, which can be associated with issues with infusion volumes and reactions. This study will evaluate the long-term safety and efficacy of rADAMTS13 for the treatment of patients with severe cTTP. This is a promising approach because it minimizes the risk of exposure to donor plasma while still providing the deficient enzyme. Recently, the drug has also been brought into a trial for study in adults with immune-mediated thrombotic thrombocytopenic purpura (iTTP)(NCT05714969).
Competing Interests
Dr. Shah indicated no relevant conflicts of interest.