MicroRNAs (miRNAs) are short, non-coding, single-stranded RNA molecules of 22 nucleotides that regulate an estimated 30 percent of mammalian genes, mostly by repressing target gene expression via either translational repression or mRNA degradation. MiRNAs are involved in a wide variety of normal and pathological cellular processes, including tumorogenesis. There is growing evidence that miRNAs also regulate mammalian hematopoiesis and that some miRNAs are specific for hematopoietic lineages. For example, down-modulation of two specific miRNAs, miR-221 and miR-222, takes place concomitantly with erythropoiesis maturation and is correlated with increased expression of c-Kit.
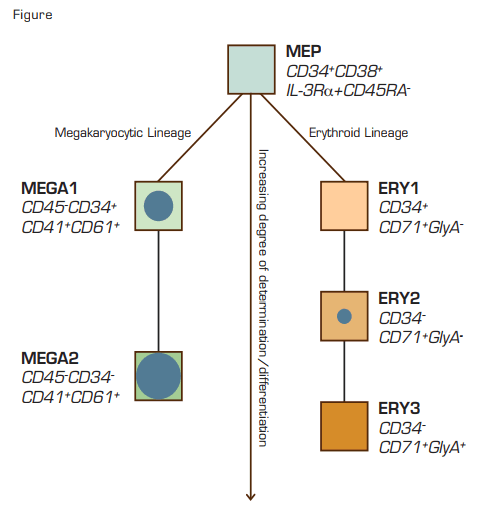
Lu et al., from Todd Golub’s laboratory at the Broad Institute in Boston, now report that the expression level of the miR-150 modulates lineage fate in megakaryocyte-erythroid progenitors (MEPs). They isolated six different populations of progenitor cells (defined by differential expression of lineage-specific surface markers) with varying degrees of commitment to megakaryocyte and erythroid development from human umbilical cord blood samples (see Figure). Using these sorted cells, a miRNA expression profile was performed with a method developed in their lab to identify specific miRNA species that were differentially expressed comparing megakaryocytic to erythroid populations. The most divergent was miR-150, which had a megarkaryocyte to erythroid expression ratio >15.
The investigators then tested the function of miR-150 using in vitro differentiation, mouse bone marrow transplantation, and colony formation assays. A lentiviral gene transfer construct encoding either miR-150 or an inhibitor of miR-150 was introduced into isolated human bone marrow derived multipotential CD34+ cells, and these were then differentiated into bi-lineage progenitors of megakaryocytes and erythrocytes. The miR-150-expressing cells yielded an 8-fold enrichment of megakaryocytes, while no change was seen from cells expressing the inhibitor. They then transplanted miR-150 transduced bone marrow cells into lethally irradiated mice. Compared to control recipients, mice transplanted with miR-150-transduced cells had >15-fold increased megakaryocytes in the bone marrow, 2- to 14-fold enrichment of donor platelets in the circulation, and >60 percent reduction of donor erythrocytes in the bone marrow. In the colony formation studies they found that miR-150 overexpression resulted in a significant increase in megakaryocyte CFU and a dramatic decrease in erythroid colony formation. Knockdown of miR-150 expression in MEP resulted in a 4-fold decrease in megakaryocyte CFU. Interestingly, the megakaryocyte colonies had normal morphology and acetylcholinesterase activity, suggesting that miR-150 is dispensable after MEP have committed to the megakaryocyte lineage. Mice treated with a hemolysis-inducing agent to stimulate erythropoiesis had significantly lower miR-150 expression in bone marrow progenitor cells.
The investigators then searched for the mRNA targets of miR-150 that mediate these cell-fate decisions. Previous reports demonstrated that the transcription factor Myb was reduced in hematopoietic progenitors in thrombocytosis and anemia.1 In the cell lines described above, miR-150 overexpression dramatically repressed Myb expression. Furthermore, knockdown of Myb expression in CD34+ human bone marrow cell cultures promoted megakaryocyte development, and forced expression of Myb reversed the megakaryocyte-promoting effect of miR-150.
Previously, two independent investigators demonstrated that miR-150 targets Myb and controls B-cell differentiation, and transplantation of miR-150 overexpressing bone marrow cells stimulated B-cell lymphopoiesis.2,3 This work now shows that a high level of miR-150 and its target Myb causes MEP to preferentially differentiate into megakaryocytes over erythroblasts. Thus the same miRNA and target gene regulate two different blood lineages (B lymphocytes and megakaryocytes). These results indicate that the maturation stage of the progenitor in which a given miRNA is expressed is also an important factor for cell-fate decision. These studies have important clinical implications. Our group reported that early erythroid progenitors from patients with polycythemia vera have lower levels of miR-150.4 This suggests that the aberrant expression of miR-150 at particular stages of progenitor differentiation may contribute to the phenotype of some blood disorders.
References
Competing Interests
Drs. Yoon and Prchal indicated no relevant conflicts of interest.