Abstract
Hexokinase deficiency is a rare autosomal recessive disease with a clinical phenotype of severe hemolysis. We report a novel homozygous missense mutation in exon 15 (c.2039C>G, HK [hexokinase] Utrecht) of HK1, the gene that encodes red blood cell–specific hexokinase-R, in a patient previously diagnosed with hexokinase deficiency. The Thr680Ser substitution predicted by this mutation affects a highly conserved residue in the enzyme's active site that interacts with phosphate moieties of adenosine diphosphate, adenosine triphosphate (ATP), and inhibitor glucose-6-phosphate. We correlated the molecular data to the severe clinical phenotype of the patient by means of altered enzymatic properties of partially purified hexokinase from the patient, notably with respect to Mg2+-ATP binding. These kinetic properties contradict those obtained from a recombinant mutant brain hexokinase-I with the same Thr680Ser substitution. This contradiction thereby stresses the valuable contribution of studying patients with hexokinase deficiency to achieve a better understanding of hexokinase's key role in glycolysis.
Introduction
Hexokinase (HK) catalyses the phosphorylation of glucose to glucose-6-phosphate (G6P) using adenosine triphosphate (ATP) as a phosphoryl donor. HK-I is the predominant HK isozyme in tissues that depend strongly on glucose use for their physiological functioning, such as brain, muscle, and erythrocytes. HK-I displays unique regulatory properties in its sensitivity to inhibition by physiological levels of the product G6P and, moreover, relief of this inhibition by inorganic phosphate (Pi).1 The recent determination of structures for human2 and rat3 HK-I has provided substantial insight into ligand binding sites and subsequent modes of interaction.2-7
Erythrocytes contain a specific subtype of HK-I (HKr)8that is encoded by the HK-I gene.9Erythroid-specific transcriptional control results in a unique red blood cell–specific cDNA that differs from HK-I cDNA at the 5′-end.10-12 Hexokinase deficiency is a rare disease with nonspherocytic hemolytic anemia as the predominant clinical feature. Seventeen families with hexokinase deficiency have been described to date,13 and only one patient has been characterized at the cDNA level.14 We now report on the molecular defect underlying hexokinase deficiency in a previously reported Dutch family.15
Study design
Patient Z62 was born from a consanguineous marriage, that is, from first cousins, and was originally reported in 1983.15 She presented with neonatal jaundice and transfusion-dependent hemolysis. Residual HK activity in the patient's erythrocytes, platelets, and lymphocytes was about 25% of normal,15 in agreement with a generalized HK deficiency caused by a mutation in HK1. DNA was isolated from peripheral white blood cells according to standardized procedures. The erythroid-specific promoter, red blood cell–specific exon 1, and exons 2 to 18 of HK1 of the patient and a healthy control subject were amplified by polymerase chain reaction (PCR) (primer sequences available on request). cDNA nucleotide and amino acid numbering starts at the HK-I start codon, and sequence variations are described according to the mutation nomenclature system.16 DNA sequence analysis was performed with the appropriate primers as described.17
Results and discussion
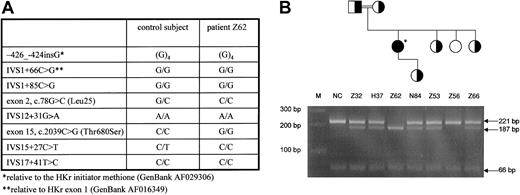
DNA sequence analysis of HK1 of the patient and a control subject revealed several base changes compared with the reference sequences (GenBank AF016349-016365) (Figure1A). Base variations for which both the patient and control subject were homozygous could represent either sequence discrepancies or polymorphisms. The c.78G>C and IVS15+27C>T are postulated dimorphic changes for Leu25 and an intronic polymorphism, respectively. The c.2039C>G missense mutation in exon 15, however, was a likely candidate to cause hexokinase deficiency because the consequent T680S substitution it encodes affects a critical residue in the active site2,3,7 18 that is, moreover, evolutionary conserved (Figure2A). Subsequent screening of the patient's family for c.2039C>G affirmed cosegregation of this mutation with lowered HK activity (Figure 1B), whereas in a healthy control population (n = 50), this mutation was not encountered (not shown). We designated the variant HK found in this family HK Utrecht.
Novel homozygous c.2039C>G (Thr680Ser) missense mutation in HK1 is associated with decreased HK activity.
(A) DNA sequence variations in HK1 in both the patient and a healthy control revealed 5 base changes that represent either polymorphic substitutions or sequence discrepancies and, in addition, 2 postulated polymorphisms (c.78G>C and IVS15+27C>T). A homozygous c.2039C>G mutation is detected only in the patient, and the consequent Thr680Ser substitution affects a highly conserved and structurally important residue in the active site of HK-I. (B) In the family pedigree chart, individuals heterozygous or homozygous for the c.2039C>G mutation are indicated by half-filled and filled symbols, respectively. Lanes in the agarose gel for each family member are directly below the symbol for that individual. The agarose gel below shows a 301-bp PCR product encompassing exon 15, which was amplified from genomic DNA and subjected to AciI digestion. c.2039C>G creates an additional restriction site upon the 2 normally present in this fragment. Thus, digestion of the wild-type allele results in fragments of 14, 221, and 66 bp, whereas the extra AciI recognition sequence yields additional fragments of 34 and 187 bp. Digestion fragments are indicated by arrows on the right (14 and 34 bp, fragments not shown). Homozygosity for c.2039C>G is confirmed in patient Z62. The patient's father (Z32), mother (H37), sisters (Z53, Z56, and Z66.)15and daughter N84 were all heterozygous, whereas sister Z56 didn't carry the mutation. All family members heterozygous for the Thr680Ser substitution displayed reduced HK activity in their red blood cells, ranging from 0.64-0.89 U/gHb (reference value: 1.34 ± 0.42 U/gHb).15 HK activity for sister Z56. was 1.22 U/gHb. *indicates Z62; M, marker; NC, healthy control.
Novel homozygous c.2039C>G (Thr680Ser) missense mutation in HK1 is associated with decreased HK activity.
(A) DNA sequence variations in HK1 in both the patient and a healthy control revealed 5 base changes that represent either polymorphic substitutions or sequence discrepancies and, in addition, 2 postulated polymorphisms (c.78G>C and IVS15+27C>T). A homozygous c.2039C>G mutation is detected only in the patient, and the consequent Thr680Ser substitution affects a highly conserved and structurally important residue in the active site of HK-I. (B) In the family pedigree chart, individuals heterozygous or homozygous for the c.2039C>G mutation are indicated by half-filled and filled symbols, respectively. Lanes in the agarose gel for each family member are directly below the symbol for that individual. The agarose gel below shows a 301-bp PCR product encompassing exon 15, which was amplified from genomic DNA and subjected to AciI digestion. c.2039C>G creates an additional restriction site upon the 2 normally present in this fragment. Thus, digestion of the wild-type allele results in fragments of 14, 221, and 66 bp, whereas the extra AciI recognition sequence yields additional fragments of 34 and 187 bp. Digestion fragments are indicated by arrows on the right (14 and 34 bp, fragments not shown). Homozygosity for c.2039C>G is confirmed in patient Z62. The patient's father (Z32), mother (H37), sisters (Z53, Z56, and Z66.)15and daughter N84 were all heterozygous, whereas sister Z56 didn't carry the mutation. All family members heterozygous for the Thr680Ser substitution displayed reduced HK activity in their red blood cells, ranging from 0.64-0.89 U/gHb (reference value: 1.34 ± 0.42 U/gHb).15 HK activity for sister Z56. was 1.22 U/gHb. *indicates Z62; M, marker; NC, healthy control.
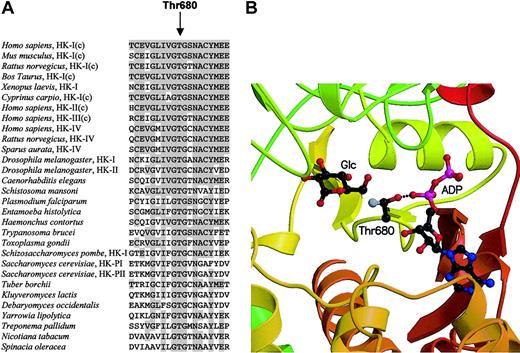
Thr680 is a critical residue in the active site of HK and is highly conserved among hexokinases from other species.
(A) Amino acid alignment of the region surrounding human HK-I residue Thr680 among 30 hexokinases. Where applicable, specific isozymes are denoted. Conserved residues compared with human HK-I are in shaded gray. The “c” in parentheses refers to the C-terminal domain of HK. (B) Schematic drawing of the active site of HK-I shows Thr680, bound glucose, and adenosine diphosphate (ADP) in ball-and-stick representation (atom coloring: carbon, black; oxygen, red; nitrogen, blue; and phosphor, magenta). The carbon atom that is absent in the Thr680Ser mutant is colored gray. Illustrated is the central position occupied by Thr680 in the active site where it is located at the tip of a loop and makes a hydrogen bond with the α-phosphoryl group of ADP. This figure was generated from the atomic coordinates of the HK-I ADP/glucose complex7 (protein data bank entry 1DGK) using computer programs Molscript22 and Raster3D.23
Thr680 is a critical residue in the active site of HK and is highly conserved among hexokinases from other species.
(A) Amino acid alignment of the region surrounding human HK-I residue Thr680 among 30 hexokinases. Where applicable, specific isozymes are denoted. Conserved residues compared with human HK-I are in shaded gray. The “c” in parentheses refers to the C-terminal domain of HK. (B) Schematic drawing of the active site of HK-I shows Thr680, bound glucose, and adenosine diphosphate (ADP) in ball-and-stick representation (atom coloring: carbon, black; oxygen, red; nitrogen, blue; and phosphor, magenta). The carbon atom that is absent in the Thr680Ser mutant is colored gray. Illustrated is the central position occupied by Thr680 in the active site where it is located at the tip of a loop and makes a hydrogen bond with the α-phosphoryl group of ADP. This figure was generated from the atomic coordinates of the HK-I ADP/glucose complex7 (protein data bank entry 1DGK) using computer programs Molscript22 and Raster3D.23
Structural changes due to a conservative threonine-to-serine substitution are likely to be limited since serine can form the same hydrogen bonds as threonine. However, the consequent removal of the methyl group could affect hydrogen bond formation by increasing the rotational freedom of the side chain or, alternatively, by introducing a water molecule at the site previously occupied by the methyl group. Small conformational changes could thus have a significant effect on catalysis, because enzymes typically require a delicate balance of interactions for optimal activity.
In view of its central position in the active site (Figure 2B), Thr680 has been proposed to hydrogen bond to the β- and γ-phosphoryl group of ATP during catalysis.7 Thus, altered binding of phosphate-containing ligands is likely an important factor in deficient enzyme function. Indeed, enzymatic properties of partially purified mutant red cell HK from the patient showed near normal affinity for glucose15 but a 2-fold decrease in affinity for Mg2+-ATP and a markedly (3- to 9-fold) decreased affinity for inhibitor glucose-1,6,-diphosphate.15 Surprisingly, these results contradict with kinetic data obtained from a recombinant human brain HK-I Thr680Ser mutant as expressed in Escherichia coli.18 The kcat value of this mutant decreased only 2.5-fold and was, in fact, characterized solely by a slightly higher affinity for glucose, whereas theKm(ATP), Ki(ATP), andKi for the G6P analog 1-5-anhydroglucitol 6-phosphate were similar when compared to wild-type HK-I.18 These unexpected discrepancies must be attributed either to the different sources of the enzyme, that is, E coli versus human red blood cells, or to the different N-terminal ends of HKr and HK-I. It highlights the differences in fate and function of mutant enzyme in vivo as underscored by a recent case of glucose-6-phosphate dehydrogenase deficiency.19
The 2-fold higher Km for Mg2+-ATP15 reflects a relatively modest effect of the Thr680Ser substitution on ATP binding to the active site. This change in Km may not affect in vitro HK activity, which is measured at excess Mg2+-ATP (5 mM),20 but may have a significant effect in vivo because intracellular ATP concentrations are much lower (0.6 mM)21 and similar to the Km. In the study conducted by Ouwerkerk et al21, blood from patient Z62 was used to measure intracellular Mg2+-ATP concentrations in oxygenated and deoxygenated red blood cells. In oxygenated red blood cells, Mg2+-ATP concentrations were slightly higher in the patient (1.04 mM) when compared with the healthy control (0.61 mM) and equal to the reticulocyte control (1.11 mM).21 Likewise, in deoxygenated red blood cells, comparable Mg2+-ATP concentrations were measured in the patient (0.57 mM) and healthy control (0.62 mM),21 which, in fact, denotes a strongly decreased Mg2+-ATP concentration when compared to a reticulocyte control (1.79 mM)21 and considering the high degree of reticulocytosis (40%-50%) in the patient. Therefore, we postulate that at physiological concentrations of Mg2+-ATP, the 2-fold increase in Km for Mg2+-ATP results in a significantly decreased HK activity in the patient's red blood cells, which is reinforced by deoxygenation. These considerations are in agreement with the severe clinical phenotype as observed in patient Z62. and even true if HK is expressed and maintained at a normal level. The residual (in vitro) activity of 25%, however, suggests an additional effect on either protein expression or enzyme stability.
The identification of a homozygous c.2039C>G (Thr680Ser) missense mutation in patient Z62 places previously determined enzymatic properties of the mutant enzyme into perspective and stresses the valuable contribution of studying patients with HK deficiency toward a better understanding of the key role played by HK in the glycolytic pathway and, in particular, in red cell metabolism.
Prepublished online as Blood First Edition Paper, August 8, 2002; DOI 10.1182/blood-2002-06-1851.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Wouter W. van Solinge, University Medical Center Utrecht, Department of Clinical Chemistry, G03.550, PO Box 85500, 3508 GA, Utrecht, The Netherlands; e-mail:wsolinge@lab.azu.nl.