Abstract
Several distinct classes of surface receptors can, on ligand binding, transmit signals that modulate the survival, proliferation, and apoptosis of peripheral B, T, and natural killer (NK) cells. At the population level, dynamic changes in lymphocyte cell numbers are strictly regulated to maintain a steady state, a process referred to as homeostasis. Although several studies have investigated the signals that regulate B- and T-cell homeostasis, little is known about the mechanisms that control the survival and proliferation of peripheral NK cells. Using an adoptive transfer system, we have investigated the role of γc-dependent cytokines, in particular interleukin 7 (IL-7) and IL-15, and major histocompatibility complex (MHC) class I molecules in peripheral NK-cell homeostasis. We observed that IL-15 plays a dominant role in the survival of peripheral NK cells, via maintenance of the antiapoptotic factor Bcl-2. IL-15 availability, however, also plays an important role because endogenous NK cells in the recipient mice influence the behavior of adoptively transferred NK cells. Finally, although NK cells bear functional inhibitory Ly49 receptors for MHC class I molecules, the presence or absence of specific ligands on host cells did not influence the survival or homeostatic expansion of donor NK cells.
Introduction
Natural killer (NK) cells are lymphocytes of the innate immune system and play important roles in immune defense.1,2 They develop from precursor cells in the bone marrow and eventually emigrate to peripheral lymphoid and nonlymphoid tissues.3,4 NK cells express multiple cell surface receptors that enable them to scan the environment for cells with abnormal major histocompatibility complex (MHC) class I expression. For example, NK cells can detect and eliminate virus-infected or transformed cells5 according to the “missing self” hypothesis.6,7 The NK-cell receptors specific for MHC class I molecules include the human killer immunoglobulin-like receptors (KIRs) and mouse Ly49 receptor families, and the CD94/NKG2 complexes in both species.8,9 These inhibitory receptors act, for the most part, as negative regulators of NK-cell activity, through their cytoplasmic immunoreceptor tyrosine-based inhibitory motifs (ITIMs) sequences, which recruit tyrosine phosphatases.10 NK cells acquire a `repertoire' of inhibitory and activating receptors as they mature in the bone marrow, in a process of NK cell “self-tolerance” (for a review, see Raulet et al11 ).
Development of NK cells critically depends on (1) cell-cell interactions between hematopoietic precursors and bone marrow stromal cells, and (2) soluble growth factors and cytokines. Cytokines using the common cytokine receptor γ chain, including interleukin 2 (IL-2), IL-4, IL-7, IL-9, IL-15, and IL-21, play an essential role in NK development, because
Lymphocyte homeostasis is an active process that allows the steady-state number of peripheral B, T, and NK cells to be maintained at optimal levels.18 The overall number of lymphocytes is tightly controlled and is influenced by the generation of new cells, as well as the survival, proliferation, and apoptosis of already existing cells. The signals for homeostasis depend on the state of cellular activation. For naïve T cells, 2 types of cell surface receptors transmit signals for survival and proliferation: (1) MHC/peptide complexes that stimulate the T-cell receptor, and (2) cytokines, including IL-7, that act via specific receptors (for a review, see Jameson19 ). For memory T cells, MHC interactions appear redundant for survival; however, either IL-7 or IL-15 could support the survival of CD8+ memory T cells.20-22 Vα14+ NK-T cells, like memory CD8+ T cells, require IL-15 but not MHC molecules (including CD1) for their survival.23,24 How γc-dependent cytokines influence the peripheral homeostasis of NK cells is unclear, although recent results have implicated a role for IL-15.25
We have assessed the role of γc cytokines in peripheral NK-cell homeostasis using 2 complementary approaches. First, we have analyzed the effects of inducible gene targeting of a `floxed' γc allele on the steady-state numbers of peripheral NK cells. Second, using an adoptive transfer system, we have characterized the behavior of mature NK cells in recipient mice that vary with respect to expression of MHC molecules, cytokines (including IL-7 and IL-15), and the presence or absence of endogenous NK cells. These studies help to define critical parameters of peripheral NK-cell homeostasis.
Materials and methods
Mouse strains
Alymphoid mice (lacking B, T, and NK cells) doubly deficient in the recombinase-activating gene (Rag)–2 and in the common cytokine receptor γ chain (Rag°γc° mice) as well as those deficient in Rag2 alone (Rag° mice) on the 11th backcross to the B6 background (H-2b) have been previously described.26 Rag°γc° mice bearing the H-2d haplotype were generated by backcrossing them to the B10.D2 background (B6.H-2d). Rag°γc° mice expressing the Ly5.1+ (CD45.1) allele were generated by crossing them to B6.Ly5a mice (CDTA, Orleans, France). Alymphoid mice deficient in MHC class I molecules associated with β2-microglobulin (Rag°γc°β2m°), in IL-7 (Rag°γc°7°) or IL-15 (Rag°15°) have been described.24 Mice transgenic for human Bcl-2 (Eμ-2-25)27 were crossed with B6.Rag° mice to generate mice with NK cells bearing the hBcl-2 transgene. The
Antibodies and reagents
Monoclonal antibodies (mAbs) directly conjugated to fluorescein isothiocyanate (FITC), phycoerythrin (PE), biotin, or allophycocyanin (APC) and specific for the following antigens (clone name in parentheses) were used in this study: CD11b (M1/70), CD45.1 (A1), CD45.2 (104), CD49b (DX5), CD69 (H1.2F3), CD94 (18d3), CD122 (TM-β1), CD127 (A7R34), CD132 (TUGm2), Ly49A (JR9), Ly49G2 (4D11), Ly49C/I (5E6), and NK1.1 (PK136). 5-(and 6-)Carboxyfluorescein diacetate succinimidyl ester (CFSE) was obtained from Molecular Probes (Eugene, OR). RPMI 1640 medium with Glutamax, fetal calf serum (FCS), and phosphate-buffered saline (PBS) were purchased from Gibco (Paisley, Scotland). Percoll was from Pharmacia (Stockholm, Sweden).
Isolation of lymphoid cells and flow cytometric analysis
Single-cell suspensions were generated from spleen by teasing the organs through a 100-μm nylon mesh followed by erythrocyte lysis. Single-cell suspensions were generated from liver by pressing the organs through a metal mesh. After 2 washes in PBS, liver lymphocytes were isolated by centrifugation using discontinuous 80%/40% Percoll gradients.30 For surface antibody staining, cells were incubated with saturating amounts of mAb for 20 minutes on ice. Biotinylated antibodies were revealed with peridinin chlorophyll protein (PerCP)– or APC-conjugated streptavidin (Pharmingen, San Diego, CA).
Bcl-2 expression was detected as described31 using directly conjugated FITC-labeled antibodies specific for human (6C8) or mouse (3F11) Bcl-2 and their isotype control antibodies from Pharmingen. Samples were analyzed using a FACScalibur flow cytometer with Cell Quest 3.3 software (Becton Dickinson, Rungis, France).
Adoptive transfer of CFSE-labeled NK cells
Splenic cell suspensions from Rag° mice were enriched by Ficoll and labeled with 5 μM CFSE. Cells were washed with ice-cold PBS and 106 NK cell equivalents were injected intravenously into nonirradiated recipient mice. At given times after transfer, splenic and hepatic lymphocytes were prepared, and CFSE+ cell frequency and phenotype were determined. Mature NK cells expressing CD122 and NK1.1 were followed. In some experiments, lymphokine-activated killer (LAK) cells were prepared using 5 day cultures of Rag° splenocytes in recombinant IL-2 (1000 U/mL).
For long-term NK-cell transfer studies, 106 splenic NK cell equivalents from B6 Rag° mice (CD45.2+) were injected into nonirradiated CD45.1+ Rag°γc° recipients. At 10, 20, or 30 days after transfer, donor CD45.2+ NK cells were quantitated in the spleen and liver of recipient mice.
Conditional deletion of a floxed γc allele in vivo
Interferon α/β (IFN-α/β; 106 U) was injected once intraperitoneally into
Statistics
P < .05 using the Mann-Whitney t test were considered significant.
Results
Peripheral NK cells can “homeostatically” expand after transfer to alymphoid hosts
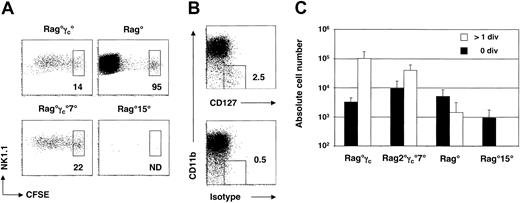
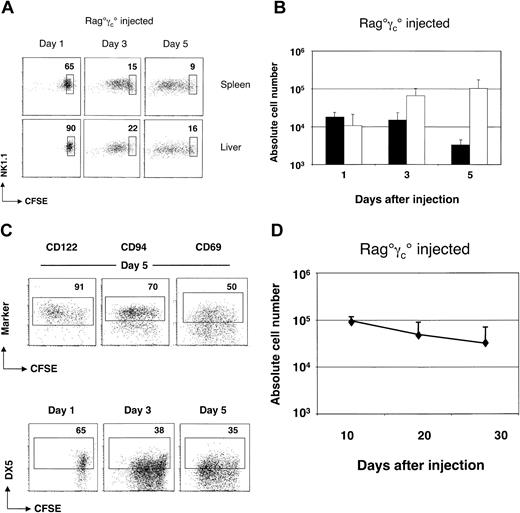
In this study, we have characterized the signals that regulate the homeostasis of peripheral NK cells. Mature splenic NK cells from Rag-2–deficient mice were used as a source of cells for adoptive transfer studies. Recipient mice were alymphoid mice (lacking B, T, and NK cells) bearing mutations in Rag-2 and common cytokine receptor γ chain genes (Rag°γc°). CFSE-labeled Rag° splenocytes containing the equivalent of 106 mature NK cells were injected intravenously into Rag°γc° mice. NK cells were identified as CD122+ (IL-2Rβ+) NK1.1+ cells. The total number of CFSE+ NK cells, as well as their CFSE dilution profiles, were assessed in the spleen and the liver of the recipient mice at day 1, 3, and 5 after transfer (Figure 1A-C).
Survival and homeostatic expansion of mature NK cells on transfer to alymphoid Rag°γc° recipient mice. (A) Kinetics of CFSE dilution of splenic and hepatic NK cells at various times after transfer. An electronic gate was set on CD122+ (IL-2Rβ+) cells. Boxes indicate percentages of CFSE+ NK cells that have not divided. (B) Absolute numbers of recovered splenic CD122+NK1.1+ NK cells at indicated times after transfer. Black bars (▪) indicate nondivided donor NK cells and open bars (□) indicate NK cells with more than one division. Error bars denote standard deviations. (C) Phenotype of adoptively transferred NK cells. An electronic gate was set on NK1.1+ NK cells. Boxes indicate the percentages of cells staining positive above background levels for the given marker. (D) Long-term survival of adoptively transferred NK cells. Absolute numbers of recovered splenic CD122+NK1.1+ NK cells at indicated times after transfer. Error bars denote standard deviations.
Survival and homeostatic expansion of mature NK cells on transfer to alymphoid Rag°γc° recipient mice. (A) Kinetics of CFSE dilution of splenic and hepatic NK cells at various times after transfer. An electronic gate was set on CD122+ (IL-2Rβ+) cells. Boxes indicate percentages of CFSE+ NK cells that have not divided. (B) Absolute numbers of recovered splenic CD122+NK1.1+ NK cells at indicated times after transfer. Black bars (▪) indicate nondivided donor NK cells and open bars (□) indicate NK cells with more than one division. Error bars denote standard deviations. (C) Phenotype of adoptively transferred NK cells. An electronic gate was set on NK1.1+ NK cells. Boxes indicate the percentages of cells staining positive above background levels for the given marker. (D) Long-term survival of adoptively transferred NK cells. Absolute numbers of recovered splenic CD122+NK1.1+ NK cells at indicated times after transfer. Error bars denote standard deviations.
The following observations were made: (1) About 3% of the total injected NK cells were recovered in the spleen of the Rag°γc° mice 1 day after injection (Figure 1B), and about 20-fold less in the liver (data not shown). (2) Mature CD122+NK1.1+ cells divided extensively in alymphoid recipients. By day 3, approximately 85% of the cells had divided at least once in the host spleen, and this proportion slightly increased by day 5. Similar expansion of NK cells occurred in the liver (Figure 1A and data not shown). (3) Absolute numbers of CD122+NK1.1+ cells increased from day 1 to day 5 after transfer, resulting in a 3.5-fold increase in NK-cell numbers in the spleen (Figure 1B) and a 10-fold increase in the liver (data not shown). Similar results were obtained with splenic NK cells from C57Bl/6 mice (data not shown). Collectively, this short-term adoptive transfer assay demonstrates that peripheral splenic NK cells have the capacity to homeostatically expand in lymphopenic Rag°γc° hosts.
The expressions of several NK-cell surface markers were analyzed during the period after adoptive transfer. We failed to detect modifications in the expression of CD122, NK1.1, CD69, or CD94 (Figure 1C). Overall expression of DX5 (CD49b) decreased with time after transfer, but this was not restricted to the dividing NK-cell population (Figure 1C).
We next assessed the long-term survival capacity of purified splenic NK cells after transfer to alymphoid Rag°γc° recipients. We quantified numbers of donor-derived CD45.2+ NK cells in CD45.1+ Rag°γc° mice from 10 to 30 days after injection (Figure 1D). In these experiments, equivalent numbers of FACS-sorted CD122+NK1.1+ NK cells were injected as described in Figure 1A-C. Compared to the short-term assay (at day 5), the number of NK cells recovered at day 10 after transfer was similar to that recovered at day 5, suggesting that NK cells do not further expand from day 5 to day 10; cell cycle analysis using Hoechst labeling confirmed that only 2% of donor NK1.1+ cells were in cycle at any time point after day 10 (data not shown). The phenotype of recovered NK cells was similar to that observed at day 5; NK cells were CD122+NK1.1+ and expressed lower levels of DX5 (data not shown). Total numbers of donor-derived NK cells decayed from day 10 to day 30 after transfer (Figure 1D), with an estimated half-life of 10 days. Collectively, these adoptive transfer studies suggest that (1) a large fraction of peripheral NK cells can proliferate on transfer to lymphopenic hosts but that these cells have a limited capacity for self-renewal; (2) a subset of peripheral NK cells appears to remain in a resting state after adoptive transfer; and (3) nonproliferating NK cells have a life span of about 10 days.
NK cells, in which the γc chain is inducibly deleted, are rapidly lost in vivo
We next investigated a potential role for γc-dependent cytokines in the peripheral maintenance of mature NK cells. We used conditional gene targeting to inducibly delete a floxP-flanked γc allele (
Splenic NK cells were analyzed in groups of mice at 2, 7, and 14 days after induction of γc deletion. The absolute number of NK cells and the frequency of
Inducible deletion of the γc gene modifies mature NK-cell homeostasis. Deletion of a
Inducible deletion of the γc gene modifies mature NK-cell homeostasis. Deletion of a
Role of γc-dependent cytokines in the peripheral homeostasis of NK cells
To define which γc-dependent cytokine was involved in the maintenance of peripheral NK cells, we adoptively transferred CFSE-labeled splenic NK cells into cytokine-deficient Rag° or Rag°γc° recipient mice. CFSE+NK1.1+ cells were analyzed in the spleen of the hosts 5 days after transfer (Figure 3A-C). Considering the essential role of IL-15 in NK-cell development, we performed adoptive transfers into mice doubly deficient in Rag2 and IL-15 (Rag°15° mice). Compared to Rag°γc° mice, absolute numbers of NK cells recovered from Rag°15° mice at day 5 were dramatically reduced (100-fold decrease; Figure 3A,C). The few surviving NK cells recovered at day 3 in IL-15–deficient hosts had not diluted the CFSE label, indicating that they had not divided (data not shown). These results demonstrate the essential role for IL-15 in the survival (and perhaps for the “homeostatic” proliferation) of peripheral NK cells.
Peripheral homeostasis of mature NK cells is conditioned by cytokines and the presence of endogenous NK cells. (A) CFSE dilution profiles of splenic NK cells 5 days after adoptive transfer into the indicated hosts. An electronic gate was set on CD122+ (IL-2Rβ+) cells. Boxes indicate percentages of CFSE+ NK cells that have not divided. ND represents not detected. (B) CD127 versus CD11b expression on electronically gated NK1.1+ splenocytes from Rag° mice. Isotype control staining is shown for comparison. (C) Absolute numbers of recovered splenic CD122+NK1.1+ NK cells at 5 days after transfer. Black bars (▪) indicate nondivided donor NK cells and open bars (□) indicate NK cells with more than one division. Error bars denote standard deviations.
Peripheral homeostasis of mature NK cells is conditioned by cytokines and the presence of endogenous NK cells. (A) CFSE dilution profiles of splenic NK cells 5 days after adoptive transfer into the indicated hosts. An electronic gate was set on CD122+ (IL-2Rβ+) cells. Boxes indicate percentages of CFSE+ NK cells that have not divided. ND represents not detected. (B) CD127 versus CD11b expression on electronically gated NK1.1+ splenocytes from Rag° mice. Isotype control staining is shown for comparison. (C) Absolute numbers of recovered splenic CD122+NK1.1+ NK cells at 5 days after transfer. Black bars (▪) indicate nondivided donor NK cells and open bars (□) indicate NK cells with more than one division. Error bars denote standard deviations.
To assess the potential role of IL-7 in the maintenance of peripheral NK cells, we similarly injected CFSE+ NK cells into mice triply deficient in Rag2, γc, and IL-7 (Rag°γc°7° mice). At day 5 after transfer, significant proliferation of the transferred NK cells had occurred in the absence of IL-7, although the absolute number of NK cells in the spleen of Rag°γc°7° mice was 2-fold decreased compared to Rag°γc° controls (Figure 3C). Interestingly, the proportion of cells with high levels of CFSE (indicative of nondivided cells) was 3-fold increased in Rag°γc°7° mice compared to Rag°γc° controls (P = .02; Figure 3A,C). These results suggested that a subset of NK cells might express CD127 (IL-7Rα+). Further analysis revealed that a small subset of CD11blo (Mac-1lo) NK cells expressed CD127 (Figure 3B). These results suggest that IL-7 plays a minor role in the homeostatic proliferation of a subset of NK cells.
Competition between adoptively transferred NK cells and host NK cells
The adoptive transfer studies were made into recipient mice that were deficient in endogenous NK cells. To address whether endogenous NK cells could influence the behavior of the injected NK-cell cohort, adoptive transfers were compared between NK-deficient (Rag°γc°) and NK-replete (Rag°) recipients. These recipient mice appear to provide a similar environment capable of supporting the short-term survival and proliferation of B and T cells.21,31 At day 5 after transfer, we recovered CFSE+ donor NK cells from both recipients (Figure 3A,C), but the NK cells behaved differently in the 2 hosts: (1) We recovered 16-fold fewer NK cells from Rag° compared to Rag°γc° hosts (although this was still 6-fold more than what was recovered from Rag°15° recipients at the same time point) and (2) only a minority (5%) of the donor NK cells in the Rag° hosts had divided compared to 86% in the Rag°γc° hosts (Figure 3A,C). These observations suggest that the adoptively transferred NK cells in the NK-replete Rag° recipients competed with endogenous NK cells (the CFSE-NK1.1+ population; Figure 3A), whereas this did not occur in the Rag°γc° hosts. Moreover, these results also suggest that the peripheral NK cell niches are limited, and that competition for IL-15 within these niches will condition NK-cell survival or expansion or both.
Role of Bcl-2 in mediating the survival effects of IL-15 on NK cells
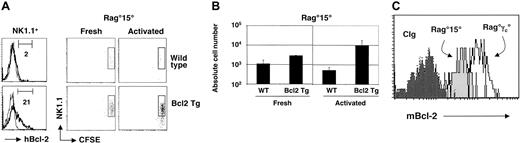
Bcl-2 is an antiapoptotic factor that can counteract certain types of cell death. We therefore assessed whether Bcl-2 was a molecular target of IL-15–mediated survival of peripheral NK cells using 2 independent approaches. First, NK cells or LAK cells from Eμ-2-25 mice (transgenic for human Bcl-227 ) or from control littermates were transferred into Rag°15° recipients. As shown in Figure 4A-B, adoptive transfer of the Bcl-2 transgenic NK cells could partially counteract a deficiency in IL-15. Absolute numbers of resting NK cells recovered were 2.5-fold increased compared to nontransgenic NK cells injected in Rag°15° recipients (P = .02); for LAK cells, 10-fold more cells were recovered. Still, the absolute numbers of NK cells remained reduced compared to those recovered from control Rag°γc° mice at the same time point (Figures 3C and 4B). This incomplete effect of the Bcl-2 transgene may result from its low expression level in NK cells (Figure 4A).
Bcl-2 is a downstream target of IL-15 in NK cells. (A) Left panels show expression of human Bcl-2 transgenic protein (hBcl-2) in gated NK1.1+ NK cells from Rag° and Rag°Eμ-2-25 mice. Percentages of Bcl-2+ NK cells is indicated. CFSE dilution profiles of splenic NK cells 5 days after adoptive transfer into the indicated hosts. An electronic gate was set on CD122+ (IL-2Rβ+) cells. (B) Absolute numbers of recovered splenic CD122+NK1.1+ NK cells at 5 days after transfer. Error bars denote standard deviations. (C) Endogenous Bcl-2 expression is shown in gated NK1.1+ splenocytes 4 days after adoptive transfer into the indicated hosts. Control isotype staining (CIg) is indicated by the dark gray histogram.
Bcl-2 is a downstream target of IL-15 in NK cells. (A) Left panels show expression of human Bcl-2 transgenic protein (hBcl-2) in gated NK1.1+ NK cells from Rag° and Rag°Eμ-2-25 mice. Percentages of Bcl-2+ NK cells is indicated. CFSE dilution profiles of splenic NK cells 5 days after adoptive transfer into the indicated hosts. An electronic gate was set on CD122+ (IL-2Rβ+) cells. (B) Absolute numbers of recovered splenic CD122+NK1.1+ NK cells at 5 days after transfer. Error bars denote standard deviations. (C) Endogenous Bcl-2 expression is shown in gated NK1.1+ splenocytes 4 days after adoptive transfer into the indicated hosts. Control isotype staining (CIg) is indicated by the dark gray histogram.
Second, we analyzed endogenous Bcl-2 expression in wild-type NK cells after adoptive transfer to Rag°γc° or Rag°15° mice. Bcl-2 levels were clearly decreased in NK cells recovered from Rag°15° recipients (Figure 4C). This result demonstrates that Bcl-2 is a direct molecular target for IL-15 action in vivo. Collectively, these results confirm and extend observations from Cooper et al25 demonstrating an important role for Bcl-2 in protection of NK cells from cell death in the absence of IL-15.
Role of MHC class I molecules in peripheral NK cell homeostasis
During their development, NK cells acquire a repertoire of MHC class I–specific receptors, which allow them to detect changes in the expression of class I molecules on targets cells (for a review, see Raulet et al11 ), and eliminate those cells that have altered or decreased MHC class I.7 The CD94/NKG2 and the Ly49 families of activating and inhibitory receptors recognize different MHC class I molecules; the inhibitory receptor Ly49A recognizes H-2Dd and H-2Dk, whereas Ly49G2 binds to H-2Dd and Ly49C/I ligands include H-2Db. On ligand recognition, inhibitory receptors prevent NK-cell activation through coupling to tyrosine phosphatases, including SHP-1, which counters the action of intracellular kinases.10 Whether engagement of inhibitory MHC class I receptors influences or regulates the peripheral homeostasis of NK cells remains unanswered.
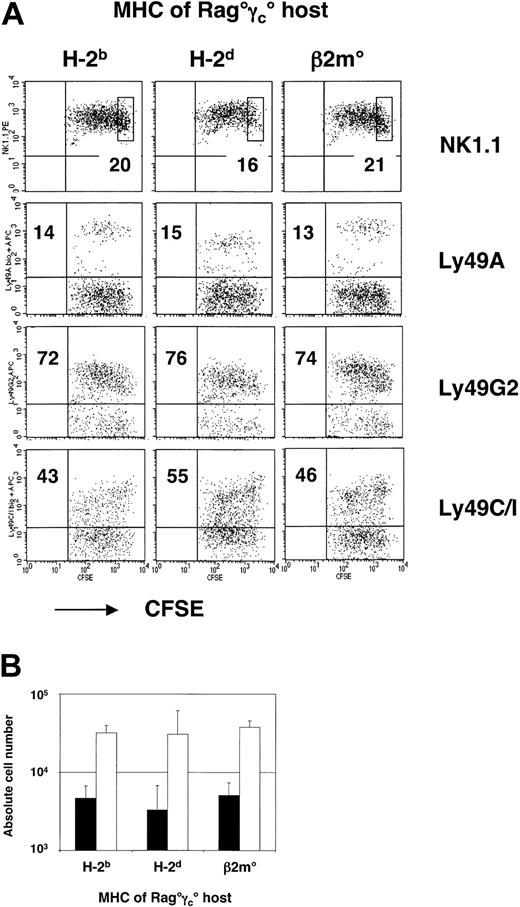
To assess the ability of MHC-specific inhibitory receptors to modify NK-cell homeostatic expansion, we performed adoptive transfer experiments of B6 Rag° NK cells (H-2b) into Rag°γc° recipients bearing MHC molecules of the H-2b or H-2d haplotype or lacking MHC class I molecules (Rag°γc°β2m° mice). We analyzed CFSE dilution profiles on splenic NK1.1+ cells coexpressing Ly49 receptors that were known to react with specific H-2b or H-2d ligands. Cells were analyzed 3 days after transfer into these different hosts to discriminate accelerated proliferation profiles.
Surprisingly, neither the absolute numbers of recovered NK cells nor the CFSE-dilution profiles of NK cells bearing different inhibitory Ly49 receptors varied between the recipient mice that expressed or did not express the specific ligands for those receptors (Figure 5A-B). Thus, NK Ly49A+ or Ly49G2+ NK cells survived and proliferated equally well in H-2b and H-2d recipients. However, the reduced Ly49A levels on donor NK cells recovered from recipients expressing the H-2Dd ligand (mean fluorescence intensity [MFI] = 300 ± 130 compared with 830 ± 120 in H-2b recipients) clearly demonstrated that the inhibitory receptors had been engaged and subsequently down-modulated. Furthermore, NK cells did not appear to divide more rapidly or to a greater extent in class I/β2m-deficient hosts (Figure 5A). These observations demonstrate that interactions of inhibitory receptors with MHC class I ligands have a very limited role (if any) in the regulation of short-term peripheral NK cell homeostasis.
The MHC background does not modify the behavior of adoptive transferred mature NK cells in alymphoid Rag°γc° recipient mice. (A) CFSE dilution profiles of splenic NK cells 3 days after adoptive transfer into the indicated hosts. An electronic gate was set on CD122+ (IL-2Rβ+) cells. Boxes in NK1.1 profiles correspond to CFSE+ NK cells that have not divided. For different Ly49s, percentages of the relevant subset are indicated. (B) Absolute numbers of recovered splenic CD122+NK1.1+ NK cells at 3 days after transfer. Black bars (▪) indicate nondivided donor NK cells and open bars (□) indicate NK cells with more than one division. Error bars denote standard deviations.
The MHC background does not modify the behavior of adoptive transferred mature NK cells in alymphoid Rag°γc° recipient mice. (A) CFSE dilution profiles of splenic NK cells 3 days after adoptive transfer into the indicated hosts. An electronic gate was set on CD122+ (IL-2Rβ+) cells. Boxes in NK1.1 profiles correspond to CFSE+ NK cells that have not divided. For different Ly49s, percentages of the relevant subset are indicated. (B) Absolute numbers of recovered splenic CD122+NK1.1+ NK cells at 3 days after transfer. Black bars (▪) indicate nondivided donor NK cells and open bars (□) indicate NK cells with more than one division. Error bars denote standard deviations.
Discussion
Lymphocyte homeostasis is achieved at multiple levels; modifications in the survival, proliferation, and apoptosis of B, T, and NK cells affect the overall composition of the peripheral lymphocyte pool and have an impact on the ability of the organism to respond with an effective immune response. In this study, we have used 2 experimental systems (adoptive cell transfer or inducible γc gene targeting) to identify the signals that condition peripheral NK-cell homeostasis. Our findings identify essential factors for NK-cell survival but also reveal the capacity for NK-cell homeostatic proliferation. Finally, we provide evidence that the peripheral NK-cell pool is not homogeneous in its survival and proliferative characteristics.
Cytokines using the common γ chain, γc (including IL-2, -4, -7, -9, -15, and -21), are essential for the generation of NK cells, and IL-15 signaling appears to represent the dominant γc cytokine in this regard.12-14 Our results using conditional deletion of a
We enriched mature, peripheral NK cells from the spleen of Rag° mice and injected them into a series of recipient mice lacking either cytokines or MHC class I molecules associated with β2m, and in other experiments into NK-replete or NK-deficient hosts. One major finding from these studies was the essential role for cytokines, including IL-15, in the maintenance of mature NK cells; when adoptive transfers were made into IL-15–deficient hosts, NK cells were rapidly lost. Thus in addition to its nonredundant role in the generation of NK cells,13,14 IL-15 appears essential for their maintenance. Similar observations have been recently obtained by Cooper et al.25 In addition, these authors provided evidence that enforced Bcl-2 expression could counteract a deficiency in IL-15,25 a finding that we confirm using a different human Bcl-2 transgenic mouse system. We further demonstrate that endogenous Bcl-2 levels are reduced in the absence of IL-15 in vivo. Surprisingly, we also found a minor role for IL-7 in the homeostasis of peripheral NK cells in that 2-fold fewer NK cells were recovered after transfer into IL-7–deficient hosts. Because both IL-7 and IL-15 can up-regulate Bcl-2 levels in lymphoid cells,17,25 one could consider that survival of NK cells would also be compromised in the absence of IL-7. This appears not to be the case, however, because IL-7–deficient NK cells in vivo express normal levels of Bcl-2 and show no signs of increased apoptosis (C. A. J. V., T. R., J. P. D., manuscript in preparation). The lower NK-cell recovery in the absence of IL-7 may result from a lower rate of entry into the cell cycle of the CD11blo subset of CD127+ NK cells.
Our studies provide evidence that peripheral NK cells have the capacity for homeostatic proliferation after transfer into alymphoid (B–,T–,NK–) hosts. Analysis of the kinetics and characteristics of the proliferating NK-cell population revealed that NK cells can rapidly expand (resulting in 8 or more divisions within 5 days), without major changes in cell surface phenotype. NK cells remained CD122+, NK1.1+, DX5+ and did not up-regulate CD69, CD94, or CD25. The proliferative expansion observed by the adoptively transferred NK cells was not modified in alymphoid hosts expressing different MHC backgrounds or lacking MHC class I–associated β2m molecules. Murine NK cells express inhibitory Ly49 receptors with specificity for different MHC class I ligands.11 Despite productive interactions between the Ly49 receptors on donor NK cells and their specific ligands in the host, there were no obvious consequences for NK-cell survival or expansion under our experimental conditions. It is known that NK cells participate in the rejection of β2m-deficient bone marrow cells and can lyse β2m-deficient targets in vitro.2,6-9 We did not observe any obvious pathologic effects of transferring mature NK cells into a β2m-deficient host; perhaps the donor cells are not sufficiently activated under these conditions. Collectively, our results indicate that homeostatic NK-cell expansion can be clearly distinguished from NK-cell activation, and that these 2 processes are under different modes of regulation.
Does homeostatic NK-cell proliferation occur under physiologic conditions? Peripheral NK cells have a low steady-state level of proliferation; only 2% to 4% of splenic NK cells are in cycle.32,33 The homeostatic proliferation that we observed may have a physiologic correlate in newborn and young animals, when the peripheral NK cell compartment is “filling up.” However, NK-cell proliferation can be observed in vivo after infection and by administration of inducing agents such as polyinosinic/polycytidylic acid.32,33 In these cases, it appears that IL-15 may be involved, because IL-15 transcripts are induced in responding macrophages and dendritic cells.34,35 The resultant increases in local IL-15 could provide a cytokine source for peripheral NK-cell expansion. Similarly, overexpression of IL-15 via transgenes15,16 or IL-15 administration14 has been shown to increase numbers of peripheral NK cells. Thus IL-15 availability appears to act as a dynamic determinant of NK-cell homeostasis. Because Rag°γc° mice cannot “consume” IL-15, it is possible that steady-state levels of IL-15 are elevated in alymphoid mice, a factor that could facilitate the homeostatic NK-cell expansion observed when using these mice as recipients.
Transfer of mature NK cells into NK-replete or NK-deficient hosts revealed that competition between NK cells for cellular resources can occur, and this competition alters the behavior of the transferred cell population. Unlike the extensive proliferation seen in NK-deficient hosts, NK cells transferred into NK-replete hosts survived, but did not proliferate. Cooper and coworkers also found that NK cells transferred into wild-type mice had the same behavior.25 These observations suggest that the signals required for survival versus proliferation of mature NK cells are different; this could be related to quantitative or qualitative differences in the signals received by the adoptively transferred cells. Because IL-15 appears to be required for both survival and proliferation of NK cells, one possibility is that endogenous NK cells occupy a niche where a non–IL-15–dependent signal resides, which is necessary for NK-cell proliferation. Alternatively, the relative abundance of IL-15 could condition the NK-cell response (survival versus proliferation).
Another important observation made in these studies involves the heterogeneity of NK-cell responses after adoptive transfer. At day 3 after transfer, about 85% of NK cells had entered the cell cycle and proliferated; by day 5 no additional NK cells were recruited to proliferate. These results suggest the existence of a nondividing or perhaps “resting” NK-cell subset. The phenotype of the nondividing NK cells was similar to that of the original NK starting population and further studies indicated that this subset lacked self-renewal capacity and had a lifespan of about 10 days. The inability of these cells to enter the cell cycle is probably not due to a dearth of niches or a limited source of cytokines, because these cells persist in alymphoid mice in the absence of obvious competition. The further characterization of these “nondividing” NK cells could provide new insights into the regulation of peripheral NK-cell pools.
Prepublished online as Blood First Edition Paper, February 13, 2003; DOI 10.1182/blood-2002-11-3392.
Supported by grants from the INSERM, the Association pour la Recherche sur le Cancer, the Ligue National Contre le Cancer, the Fondation pour la Recherche Medicale, and the Pasteur Institute.
T.R. and C.A.J.V. contributed equally to this work.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We would like to thank Jacques Peschon (Immunex), Paulo Vieira, and Jacques Roland (Institut Pasteur) for providing mice and reagents. We thank Sandrine Samson, Anouk Caraux, Simona Zompi, and Francesco Colucci for stimulating discussions and comments on the manuscript.

![Figure 2. Inducible deletion of the γc gene modifies mature NK-cell homeostasis. Deletion of a \batchmode \documentclass[fleqn,10pt,legalpaper]{article} \usepackage{amssymb} \usepackage{amsfonts} \usepackage{amsmath} \pagestyle{empty} \begin{document} \({\gamma}_{\mathrm{c}}^{\mathrm{flox}}\) \end{document} allele12 was induced using IFN-α/β in mice bearing the Mx-cre transgene.28 NK cell numbers as percentages of control (▵) and percentages of γc+ NK cells (○) at the indicated time points are presented on left and right y-axes, respectively. Small symbols indicate individual mice.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/101/12/10.1182_blood-2002-11-3392/6/m_h81234461002.jpeg?Expires=1763521757&Signature=fmBXcw4LgDzX6Y7wtlBOLIdSC11PlY2graHWCyz~vviqeErgkGCvzhwDh86APjoJIQcFSzI421RtAw02VkjFsUPft6Pql74wDSxe0G1YQ6HVdNJC99cEzKNk5YxuLU-4xoTPtFcf16cBAXJVkkGndqT1Nr0lWKc9hL8WFiVfX7WTU7IcUaY8UkXzlVP6dokJn8s4cbZ5nDt3YRzqWPRwOoVPJabYxt9HbiKJNtSguT-o6Un0SoeaYVi98pnl06tdWRZt~XYpq4DzBYaS4uetMiqbqafzow0t8Xod-phjbnSXxsw-21mmtyqVO-WUkZlP99taK-YEMDbQ3ppAUzoLag__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)