Abstract
CD20 has been reinvestigated in 66 patients with multiple myeloma (MM). Twelve of the patients (18%) expressed CD20, including 5 of 50 patients at diagnosis presenting 100% CD20+ cells. Seven (58%) of 12 CD20+ patients with MM had a small mature plasma cell morphology as opposed to 4 (7%) of 54 with CD20- MM (P = .0001). Of note, 10 (83%) of 12 patients with CD20+ MM had t(11;14) as opposed to 5 of 54 (9%) CD20- patients (P < .001). All the patients with 100% CD20+ cells presented with t(11;14) and 4 of 5 with a small mature plasma cell morphology. Thus, 66% of the patients with t(11;14) expressed CD20, whereas only 4% of the 51 patients lacking such translocation expressed CD20 (P < .0001). In conclusion, CD20 expression is associated with small mature plasma cell morphology and with t(11;14) in patients with MM.
Introduction
Multiple myeloma (MM) is a very heterogeneous disease. A large part of this heterogeneity is now related to chromosomal abnormalities, mainly 13q deletion and 14q32 translocations.1,2 The phenotype of MM cells could be also an important component of tumor heterogeneity and represents a specific target for new treatments. Because this is the case for CD20 in lymphomas, we have re-evaluated its expression in MM. MM cells were first described as lacking CD20 expression.3 Subsequent studies have shown that up to 22% of patients with MM had partial or total expression of CD20.4-7 However, no effort has been made to better characterize this subset of CD20+ patients with MM. In the current study, we show that the expression of CD20 is associated with a small mature plasma cell morphology and with t(11;14).
Study design
Patients
CD20 has been prospectively evaluated in 66 consecutive patients with MM, including 50 at diagnosis and 16 at relapse. They were 59% IgG MM, 25% IgA MM, and 15% Bence Jones–only MM. The diagnostic criteria and staging were those of the Southwest Oncology Group of the United States.8 The University Hospital of Nantes review board approved these studies. Informed consent was provided according to the Declaration of Helsinki.
Morphology
On stained bone marrow aspirates, 100 plasma cells were classified according to Greipp et al.9 We subclassified the mature and the intermediate types as lymphoplasmacytic when more than 30% plasma cells were characterized by a less abundant cytoplasm; the nuclear-to-cytoplasm ratio was less than 0.6 according to Goasguen et al.10 Morphology was reviewed in a blind fashion by 2 of us (R.G. and R.B.). Agreement was 89% on the mature morphology and on the lymphoplasmacytic type more particularly.
Immunophenotype using flow cytometry
After density separation (lymphocyte medium; Eurobio, les Ulis, France) 0.5 × 106 bone marrow mononuclear cells were incubated for 20 minutes at room temperature with fluorescein isothiocyanate (FITC) labeled with anti-CD45 and phycoerythrin-cyanine 5 (PE-Cy5), anti-CD138 (Coulter Immunotech, Miami, FL), allophycocyanin (APC), anti-CD38 phycoerythrin (PE) IgG control (Becton Dickinson, San Jose, CA), or PE–anti-CD20 (Pharmingen, San Diego, CA) monoclonal antibodies. The PE fluorescence was used to explore the phenotype of these myeloma cells for different markers (CD19, CD22, etc). After 2 washes, cells were fixed in 1% formaldehyde and 15 000 events/tube were counted using a flow cytometer (FACScalibur; Becton Dickinson). To increase the sensitivity of the analysis, in a second step, acquisition through a “live-gate” drawn on side scatter (SSC)/CD38 strongly positive cells was performed and data from at least 103 plasma cells were analyzed with CellQuest software (Becton Dickinson). Furthermore, accurate identification of plasma cells requires the use of a sequential gating. A second gate was set around cells expressing a high level of CD38 and CD138 as previously published.11,12 The apoptotic cells were excluded with a third region set on the light scatter of the CD38+/CD138+ cells.
FISH analysis
All the patient samples have been analyzed by fluorescence in situ hybridization (FISH), using probes specifically detecting the following abnormalities: del13, t(11;14), t(4;14), and other 14q32+ rearrangements. The technique and the probes have been extensively reported in previous publications.1,2
Results and discussion
Twelve (18%) of 66 patients had more than one third of their tumor cells expressing CD20 and 5 patients (7%) expressed CD20 on 100% of their tumor cells. These 5 patients were observed only at diagnosis and represent 5 (10%) of 50 patients. CD20+ MM expressed neither CD19 nor CD22. We have looked for correlations between CD20 and morphology. Seven (58%) of 12 patients with CD20+ MM had a small mature plasma cell morphology (ie, > 30% small plasma cells) versus 4 (7%) of 54 patients with CD20- MM (Fisher exact test, P = .0001; Figure 1). Four of 5 patients with 100% CD20+ MM had a small plasma cell type morphology In addition, none of the CD20+ MM cases showed any plasmablastic cells nor any significant amount of immature myeloma cells (ie, < 6% of cells). The 14q32 chromosomal abnormalities were normally distributed (data not shown) in the 66 patients. A strong correlation was found between CD20 and t(11;14). Indeed, 10 (83%) of 12 patients with CD20+ MM had a t(11;14) versus 5 (9%) of 54 CD20- MM patients (P < .0001). Of note, 5 of 5 MM patients with 100% CD20+ MM cells had a t(11;14). Conversely, 10 (66%) of 15 MM patients with a t(11;14) had CD20+ MM versus 2 (4%) of 51 MM patients lacking t(11;14) (P < .0001). In addition, all 6 cases with t(4;14) and cases with other 14q32+ rearrangements were restricted to the patient population with CD20- MM.
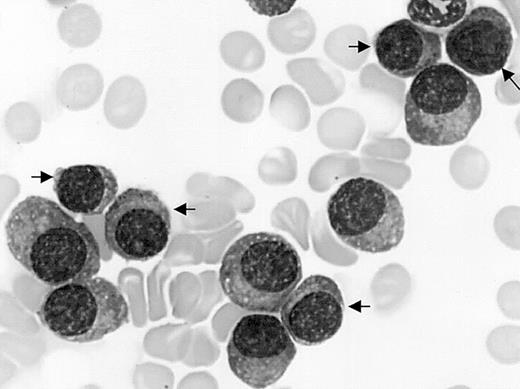
Mature plasma cell morphology. Bone marrow myeloma cell infiltrate by mature malignant plasma cells including 45% of small mature plasma cells (arrows) in a patient with CD20+ MM and t(11,14). May-Grünwald-Giemsa staining; original magnification, × 1000.
Mature plasma cell morphology. Bone marrow myeloma cell infiltrate by mature malignant plasma cells including 45% of small mature plasma cells (arrows) in a patient with CD20+ MM and t(11,14). May-Grünwald-Giemsa staining; original magnification, × 1000.
Because CD20 is now a therapeutic target in lymphomas, we have re-evaluated its expression in MM to better characterize the subset of patients expressing CD20. Eighteen percent of patients had CD20+ MM in agreement with the most recent evaluations (16%-22%).4-7 The CD20 phenotype was significantly associated with a mature morphology and more significantly with a small plasma cell (ie, lymphoplasmacytic) morphology. Indeed, the small mature plasma cell MM, observed in about 10% of MM series, represents 1 of the 2 mature types of MM, the other one being the Marschalko type.13 Both types are characterized by a low-grade malignancy.13 Incidentally, we discovered that data illustrating the strong correlation between the expression of CD20 with the small mature plasma cell type had been already described but ignored 10 years ago by Bartl et al.13 Indeed, this group5 evaluated CD20 phenotype and morphology in 45 patients with MM. From the data available in their tables, we found that 4 (66%) of 6 CD20+ MM patients had a small mature plasma cell morphology as opposed to 2 (8%) of 25 CD20- MM patients (P = .006).5 Another major correlation we found is that with t(11;14). This is in complete agreement with the data recently published by the Mayo Clinic group showing that 41% of their patients with a t(11,14) had a small mature plasma cell morphology.14,15 We have noted that the CD20 gene is mapped in 11q13.16 However, 11q13 breakpoints involved in MM are far from the CD20 gene location (> 10 Mb). Furthermore, one third of patients with MM with t(11;14) do not express CD20. Thus, we can exclude the involvement of t(11;14) in the aberrant expression of CD20 by some MM. Could CD20+ MM take advantage of anti-CD20 monoclonal antibody therapy? Our data suggest that patients expressing CD20 on 100% of myeloma cells at diagnosis could be ideal targets. To support this point is the observation that the unique MM patient who responded previously to CD20 therapy (partial response) had the most extended expression (65%) of CD20.17
Prepublished online as Blood First Edition Paper, April 17, 2003; DOI 10.1182/blood-2002-11-3333.
Supported by la Ligue Nationale Contre le Cancer (equipe labellisee, 2001).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked ”advertisement” in accordance with 18 U.S.C. section 1734.