Comment on Biragyn et al, page 1961
Fusion proteins containing chemokine receptor ligands have been used to selectively deliver tumor antigens to the MHC class II processing pathway in dendritic cells, promoting tumor-specific CD4 T-cell responses and tumor-specific immunity. This novel strategy of vaccination, targeting the antigen-presenting cell (APC), may have broad application to T-cell immunotherapy.
There is considerable enthusiasm in cancer therapy for developing vaccinations using protein tumor antigens or DNA-encoding tumor antigens. In fact, targeting the clonally expressed “idiotypic” antigen (Id) on malignant B cells is currently considered one of the most promising approaches.1 However, potential lack of Id antigenicity and consequent tumor immunogenicity poses an obstacle to this approach. Tumor immunogenicity requires an effective T-cell (cellular) immune response. Previously, Biragyn et al2,3 developed an approach to enhance the Id-specific T-cell immune response by targeting delivery of weakly immunogenic antigens to chemokine receptors. They created recombinant proteins containing proinflammatory chemoattractant ligands (chemokines andβ-defensins) fused with nonimmunogenic B-cell lymphoma Id (sFv).Those chemokine-Id fusions or DNA constructs encoding the corresponding fusions, but not Id alone, induced protective and therapeutic antitumor immunity in vivo without the need for adjuvant. Results from those studies using recombinant proteins and their corresponding DNA constructs indicated that CD8 T cells participated in tumor protection, suggesting a role for major histocompatibility complex (MHC) class I–restricted antigen presentation.
In this issue, using additional fusion proteins containing chemokines whose receptors are preferentially expressed on immature dendritic cells (DCs), Biragyn and colleagues examined the role of both the class I pathway and the class II endocytic pathway in processing and presenting these proteins to T cells. Chemoattractant fusion proteins were processed and efficiently presented by immature DC populations via the MHC class II pathway to Id-specific CD4+ T cells in vitro and in vivo. Compounds that disrupted the endosomal/lysosomal compartment or vesicular transport of newly synthesized class II molecules inhibited antigen processing and presentation, whereas an inhibitor of proteosomes, a structure required for delivering antigens for class I–restricted antigen presentation, did not. In vivo administration of chemokine-Id led to Idspecific tumor protection that was associated with secretion of the Th1 cytokine interferon-γ.
Evidence has supported the importance of anti-Id antibodies and CD8 T cells in antitumor responses. The use of select chemokine receptors to direct proteins in to the class II process ing pathway in immature dendritic cells may provide greater tumor protection. CD4T cells are central inFIG1 importance in immuneresponses, not only participating as effector cells but also providing “help” for CD8 T-cell responses and humoral responses. The findings in this report raise certain questions regarding this approach. How do the internalized chemokine receptor and its ligand traffic through the endocytic pathway? Will the use of chemokine carriers elicit antihost chemokine autoimmune responses? Taking advantage of chemokine redundancy, the authors created viral chemokine-Id fusions and demonstrated that they provided equal tumor protection. However, the “non-self” carrier might be more immunogenic, limiting repeated clinical use. Clinical trials using Id vaccines have provided encouraging results.4 Targeting the MHC class II pathway via chemokine receptors may have wide application in immunotherapy. The report in this issue, providing a methodtoenhance T-cell responses against a weakly immunogenic tumor, is provocative and clearly supports testing chemokine-Id fusions in clinical tumor immunity.
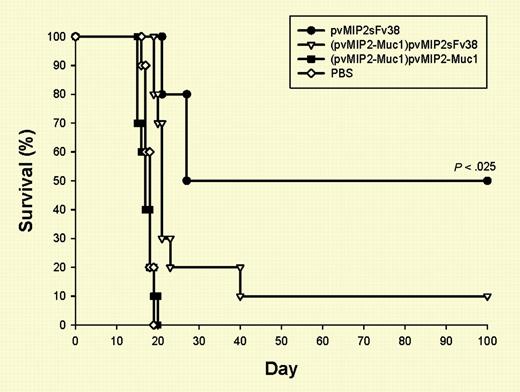
Chemokine-Id fusion proteins provide efficient in vivo tumor-specific immune response. See the complete figure in the article beginning on page 1961.
Chemokine-Id fusion proteins provide efficient in vivo tumor-specific immune response. See the complete figure in the article beginning on page 1961.