Abstract
Splenectomy has been a standard treatment for adult patients with idiopathic thrombocytopenic purpura (ITP) for more than 50 years. However, the durability of responses, the ability to predict who will respond, and the frequency of surgical complications with splenectomy all remain uncertain. To better interpret current knowledge we systematically identified and reviewed all 135 case series, 1966 to 2004, that described 15 or more consecutive patients who had splenectomy for ITP and that had data for 1 of these 3 outcomes. Complete response was defined as a normal platelet count following splenectomy and for the duration of follow-up with no additional treatment. Forty-seven case series reported complete response in 1731 (66%) of 2623 adult patients with follow-up for 1 to 153 months; complete response rates did not correlate with duration of follow-up (r = -0.103, P = .49). None of 12 preoperative characteristics that have been reported consistently predicted response to splenectomy. Mortality was 1.0% (48 of 4955 patients) with laparotomy and 0.2% (3 of 1301 patients) with laparoscopy. Complication rates were 12.9% (318 of 2465) with laparotomy and 9.6% (88 of 921 patients) with laparoscopic splenectomy. Although the risk of surgery is an important consideration, splenectomy provides a high frequency of durable responses for adult patients with ITP. (Blood. 2004; 104:2623-2634)
Introduction
Splenectomy was the primary treatment for idiopathic (immune) thrombocytopenic purpura (ITP) prior to the introduction of glucocorticoids more than 50 years ago.1 For the past 50 years, splenectomy has remained a standard treatment for adults with ITP who do not respond to glucocorticoid treatment or who continue to require glucocorticoids to sustain a safe platelet count.2-4 Yet even after decades of experience, important questions concerning splenectomy for ITP remain unresolved.
What is the durability of complete responses achieved with splenectomy? Although many case series describe complete remissions in about two thirds of patients,2-4 some studies have reported a continuing occurrence of relapses with long-term follow-up.5,6 It has even been suggested that relapse of ITP may occur in most patients if follow-up after splenectomy is sufficiently long.7 Therefore, the durability of responses to splenectomy is uncertain.
Can any preoperative characteristic predict the success of splenectomy? Multiple patient and disease characteristics have been reported to predict response to splenectomy, but the findings are inconsistent. Therefore, the clinical value of any preoperative characteristic is unknown.
What are the mortality and morbidity of splenectomy for ITP? Splenectomy has been considered to be a safe procedure,2,4 but, because death caused by bleeding in patients with ITP is uncommon, 2 (1.6%) of 134 patients8 and 1 (0.4%) of 245 patients9 in 2 case series, death caused by splenectomy must be low to be acceptable. Complications of splenectomy may be substantial; one case series reported surgery-related death in 1 (1.3%) and postoperative complications resulting in prolonged hospitalization or readmission in 20 (26%) of 78 patients.8 Therefore, the relative risks and benefits of splenectomy are uncertain.
To understand and interpret the large number of publications on these issues, a systematic review10,11 of all articles describing splenectomy for ITP since 1966 was performed. This review focuses on ITP in adults because spontaneous remissions may occur in many children with persistent thrombocytopenia12 ; therefore, splenectomy is rarely performed.13
Methods
Literature search
Ovid software was used to search the Medline database from January 1, 1966, to February 29, 2004. Case series published prior to 1966 were not retrieved, because they often included patients treated before 1950, for whom splenectomy was performed as the primary treatment, before glucocorticoids became available.1 Also, some current supportive care measures, such as platelet transfusions and intravenous immunoglobulin, were not available prior to 1966. All terms were keyword searched by using unlimited truncation, retrieving articles identified by “splenec:,” “spleen and remov:,” or “spleen and extract:” that were also identified by “thrombocytopenia,” “thrombocytopenic purpura,” “ITP,” or “AITP.” The search was limited to English-language articles. The bibliographies of all retrieved articles were searched for additional relevant articles.
Article selection criteria
Articles published in pediatric journals and articles describing splenic radiation, ultrasound, or embolization were not retrieved. Retrieved articles were selected for review if they reported 15 or more consecutive patients who had splenectomy for ITP and who were followed for at least 1 month after splenectomy, and if they contained data on 1 or more of the 3 outcomes of interest: (1) platelet count response, (2) predictors of response, or (3) surgical complications. Articles reporting fewer than 15 patients were excluded to avoid reports of exceptional patients; however, the bibliographies of these articles as well as the bibliographies of review articles with no patient data were searched to identify additional articles. Case series were not reviewed if it was clear that patient accrual was not consecutive. When multiple case series reported the same or a cumulative group of patients, only the most inclusive case series was selected. Articles describing only group data were selected only if it was clear that all reported patients had had a splenectomy for ITP. Articles were excluded if the data were insufficient to distinguish patients with ITP from patients with disorders other than ITP. When the original authors described their patients as having ITP, we accepted their diagnosis even though some investigators included patients with evidence for other autoimmune disorders within their definition of ITP.
Articles that reported data on children that could not be distinguished from data on adults were included only if it could be determined that 75% or more of the patients were 14 years old or older, or if the case series focused on adults and the mean or median age reflected the adult population, but the range of ages included children. Adults were defined as being 14 years old or older because this was the predominant age distinction for children and adults in the reviewed articles. To assess platelet count response, case series reporting only adults and case series that included both adults and children were analyzed separately. Case series with up to 25% children were not excluded from this review because they accounted for 38 (45%) of all 85 articles that could be analyzed for platelet count response.
Article assessment
In most articles, selection criteria were apparent. For articles in which criteria were unclear, the decision for selection was made by consensus among all authors. Each selected article was reviewed independently by 2 or more of the authors with the use of a standard form and a priori criteria for outcome assessments. Disagreements were resolved by consensus among all of the authors.
Assessment of platelet count response
The platelet count response of patients who survived splenectomy is described in relation to follow-up duration; therefore, patients were not included unless follow-up duration after splenectomy was reported. The platelet count determining a response was defined as the first count obtained after at least 1 month following surgery, to avoid the influence of perioperative treatment for ITP. (1) Complete response was defined as achievement and maintenance of a normal platelet count (> 150 × 109/L or as defined in the original report and at least 100 × 109/L) for all measurements 30 days or longer after splenectomy, and with no additional treatment for ITP, except for the tapering of perioperative glucocorticoids or other treatments. (2) Partial response was defined as achievement of a platelet count of 50 × 109/L (or 30 × 109/L in recent publications) or more for any measurement of 30 days or longer after splenectomy, with or without other treatment, excluding patients who qualify for complete response. Therefore, patients who relapsed after initially achieving a normal platelet count were considered to have a partial response. Some articles only described complete responses and did not describe partial responses. (3) No response was defined as failure to achieve a platelet count of 50 × 109/L (or 30 × 109/L in recent publications) for any measurement of 30 days or longer after splenectomy. If individual patient platelet counts were not reported, the investigators' description of the group response was accepted if it was clear that the responses were consistent with these criteria. Our definition of a complete response is clear but restrictive; other patients who are defined in this review as having a partial response or no response may have had substantial benefit from splenectomy. Although some patients defined as having a partial response may have had only a transient, trivial increase of their platelet count, others may have had a clinically important increase in their platelet count and required no further treatment. Also some patients defined as having no response may have had a substantially increased platelet count. These distinctions among patients defined as having a partial response or no response were not possible in most articles.
Relapse was defined as the recurrence of thrombocytopenia following initial achievement of a normal platelet count. It was not possible to distinguish patients who had a recurrence of only transient, mild thrombocytopenia from patients who had recurrent severe, symptomatic thrombocytopenia. Thirty-seven articles that could be evaluated for platelet count response could not be evaluated for relapse because only a single platelet count was reported, the time of relapse was not reported, or it was not clear whether the patients had ever achieved a normal platelet count.
Because of the frequency of spontaneous remissions in children with chronic ITP,12 data from case series that included up to 25% children and in which data on children and adults could not be distinguished were analyzed separately from case series reporting only adult patients. Because the technique of splenectomy should not affect the platelet count response, data from case series reporting open laparotomy and laparoscopy were combined for this analysis.
Assessment of predictors of response
Articles were analyzed only if data were presented to support the conclusion that a variable did or did not predict a response to splenectomy. Therefore, articles that did not present a statistical analysis of their data, or did not present data from which we could calculate a P value, were excluded from this analysis. Application of uniform criteria or analysis of pooled data from different articles was not possible because case series assessed different demographic, clinical, and laboratory variables in different ways and used different definitions for a successful outcome. Also the methodology of techniques, such as determination of the site of platelet sequestration, was different among the articles. Variables reported in each article were categorized as predictive, not predictive, or not interpretable. We reported variables as predictive if (1) the observed difference was statistically significant and (2) the correlation was persistent for the duration of patient follow-up. If the original authors had performed a multivariate analysis, only the variables that were determined to independently correlate with response after adjustment for other variables were accepted as predictive. If no statistical comparison was made in the original article, we performed an appropriate statistical test to obtain the P value. If appropriate statistical evaluation of the presented data showed no significant correlation of a variable, but the original authors had reported the variable as predictive, we categorized the variable as not interpretable.
Because the technique of splenectomy should not affect the platelet count response, data from case series reporting open laparotomy and laparoscopy were combined for this analysis. Data from case series of adults only and adults plus children were also combined; although patient age may affect the response to splenectomy, age was analyzed as one of the prediction variables.
Assessment of surgical complications
Complications related to splenectomy were defined as those occurring within 30 days of splenectomy, or later if the complication occurred during the original hospitalization for splenectomy. Complications beyond the postoperative period, such as overwhelming sepsis14 and thrombosis15-17 that may be attributable to the absence of the spleen, were not analyzed. Even if an article did not explicitly address surgical complications, data were included in the analysis of surgical mortality if deaths were reported or if it was clear that no patients had died.
Because the technique of splenectomy may affect the risk for surgical complications, case series describing open laparotomy and laparoscopic procedures are described separately. For articles that accrued patients prior to 1991, the year of the first report of laparoscopic splenectomy for ITP,18 and the surgical technique was not defined, it was assumed that splenectomy was performed by open laparotomy. However, if patient accrual began during or after 1991 and the surgical technique was not defined, the article was not included in this analysis. If the article stated that both open laparotomy and laparoscopy were performed but the data did not distinguish these techniques, the article was not included in this analysis. Data from case series reporting adults only and adults plus children were combined for this analysis.
Statistical methods
All data were entered into a Microsoft Access (Redmond, WA) database. The correlation between duration of follow-up and the complete response rate and between duration of follow-up and the relapse rate were evaluated by the Spearman correlation coefficient; the corresponding graphs were produced with Microsoft Excel (Redmond, WA). When it was necessary to analyze data from the reviewed articles for correlation of prediction variables with response, we used the chi-square test of independence to evaluate differences between rates of platelet count response to splenectomy across categorical variables. Mortality rates with open laparotomy and laparoscopic techniques were compared by using Fisher exact test; morbidity rates were compared by using the chi-square test. A 2-sided P less than .05 was considered statistically significant.
Results
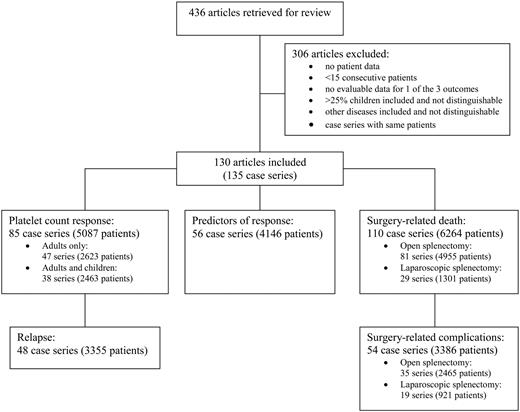
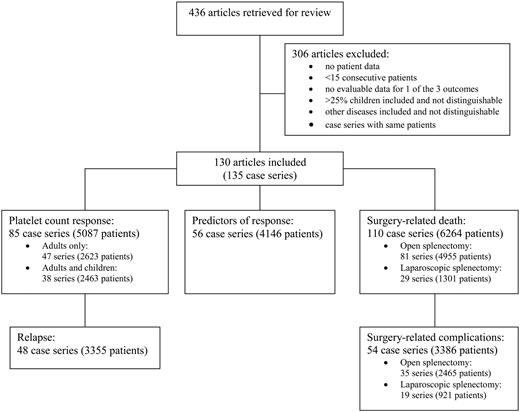
The literature search identified 436 articles (Figure 1); 306 articles did not meet our selection criteria and were not reviewed. We selected 130 articles that reported 15 or more consecutive patients who had splenectomy for ITP and that presented evaluable data on 1 or more of the 3 outcomes of interest: (1) platelet count response, (2) predictors of response, or (3) surgical complications. Patient accrual in these articles spanned 58 years, from 1944 to 2002, and the articles represent the experience of 29 countries (Table 1). These 130 articles contained 135 case series, as 5 articles describing surgical techniques reported separate case series for laparotomy and laparoscopic splenectomy.100,102,103,105,140 In 9 of the case series,9,45,59,88,94,115,130,134,141 patients were enrolled and analyzed prospectively for long-term platelet count responses. Two of the 9 prospective case series were randomized trials in which splenectomy was part of the treatment in both groups.59,141 No case series compared splenectomy with either a nonsurgical form of treatment or observation. In 4 other case series, only collection of perioperative data was performed prospectively91,114,120,139 ; the remaining 122 case series were retrospective analyses. The numbers of case series and patients analyzed for each of the different outcomes are presented in Figure 1.
Article and patient selection. Articles were retrieved for review if their journal, title, or abstract suggested that they contained evaluable data on eligible patients and indicated that the articles did not report primarily on children. Retrieved articles were selected for review if they reported 15 or more consecutive patients who had splenectomy for ITP and who were followed for at least 1 month after splenectomy, and if they contained data on 1 or more of the 3 outcomes of interest: (1) platelet count response, (2) predictors of response, or (3) surgical complications.
Article and patient selection. Articles were retrieved for review if their journal, title, or abstract suggested that they contained evaluable data on eligible patients and indicated that the articles did not report primarily on children. Retrieved articles were selected for review if they reported 15 or more consecutive patients who had splenectomy for ITP and who were followed for at least 1 month after splenectomy, and if they contained data on 1 or more of the 3 outcomes of interest: (1) platelet count response, (2) predictors of response, or (3) surgical complications.
Platelet count response
In 47 case series reporting only adults, 1731 (66%) of 2623 patients had a complete response with a median follow-up of 29 months (range, 1-153 months) (Table 2); 1853 (88%) of 2116 had a complete or partial response. When the median rate of complete response across the 47 individual case series was calculated, rather than combining all patients, the rate of complete response was 67% (range, 37%-100%). In 38 case series that included up to 25% children, the frequency of complete responses was slightly but significantly greater (Table 2): 1775 (72%) of 2463 adults and children had a complete response with a median follow-up of 23 months (range, 3-130 months), 1449 (88%) of 1640 adults and children had a complete or partial response. The median complete response rate across the 38 individual case series of adults and children was also 72% (range, 28%-96%). Platelet count responses were similar when only case series with a median or mean follow-up of at least 5 years after splenectomy were analyzed (Table 2). In 14 case series reporting only adults, 456 (64%) of 707 patients had a complete response with a median follow-up of 7.25 years (range, 5-12.75 years). In 7 case series reporting adults and children, 323 (71%) of 452 patients had a complete response with a median follow-up of 7 years (range, 5-10.83 years). Data from the 9 prospective case series were not different from the retrospective analyses and were, therefore, not reported separately. The frequency of complete responses was not different across the 58 years of patient accrual. The median rate of complete responses in the first 42 case series, published from 1968 to 1994 with patient accrual from 1945 to 1990, was 69% (range, 28%-88%); the median rate of complete responses in the second 43 case series, published from 1995 to 2004 with patient accrual from 1968 to 2001, was 67% (range, 37%-100%) (Table 1).
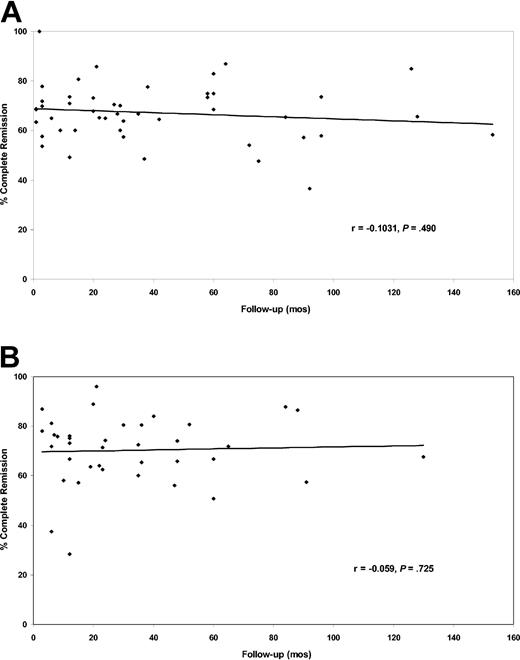
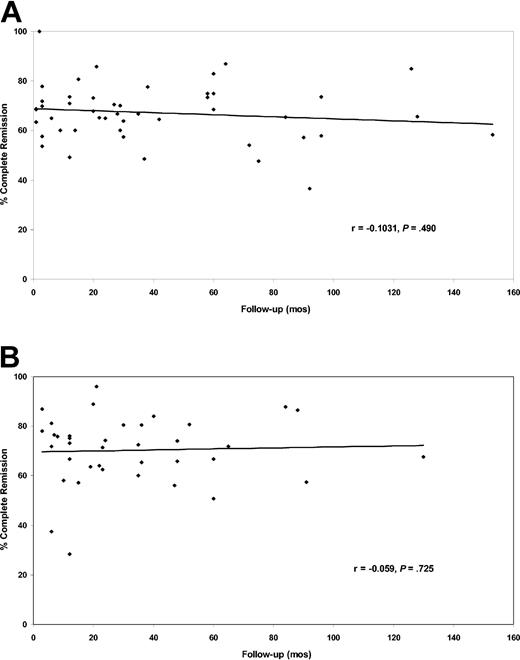
The complete response rates in case series of adults only and of adults and children did not correlate with the duration of follow-up (Figure 2). There was also no correlation between the complete response rates and duration of follow-up when all 85 case series, combining case series of adults only with case series of adults plus children, were analyzed together (rs = -0.074; P = .50).
Relationship of complete response rates with median or mean duration of patient follow-up. (A) Data for the 47 case series reporting adults only with follow-up for 1 to 153 months (median, 29 months). (B) Data for the 38 case series reporting adults and children with follow-up for 3 to 130 months (median, 23 months).
Relationship of complete response rates with median or mean duration of patient follow-up. (A) Data for the 47 case series reporting adults only with follow-up for 1 to 153 months (median, 29 months). (B) Data for the 38 case series reporting adults and children with follow-up for 3 to 130 months (median, 23 months).
Relapse rates following splenectomy were evaluable in 48 of the 85 case series reporting 3355 patients. When case series reporting only adults were analyzed together with case series reporting adults and children, relapses occurred in a median 15% of patients (range, 0%-51%) with a median follow-up of 33 months (range, 3-153 months). The relapse rate appeared to increase with duration of follow-up, but the correlation did not reach statistical significance (rs = 0.275, P = .059). Relapse rates were also not significantly correlated with duration of follow-up when case series reporting only adults and case series reporting adults and children were evaluated separately.
Predictors of response
Demographic, clinical, and laboratory variables that have been studied for their ability to predict response to splenectomy are distinguished as preoperative and postoperative prediction variables (Table 3).
Among variables that are available prior to splenectomy, age at the time of splenectomy most often correlated with response, with 14 case series reporting that younger age was associated with a better response. In 7 of these 14 case series, the mean or median age of the groups of patients with the better outcome was significantly less than the age of the groups of patients with the worse outcome. In these 7 studies there was no specific age cut point; the mean or median age of the patients with better outcomes was 32 to 51 years, compared with 40 to 73 years in the groups with less good outcomes. In the other 7 case series, responses of patients above and below specific ages—30 to 60 years in the different case series—were compared, and the younger group had a better outcome. In all 7 case series that analyzed multiple variables in a multivariate model,5,67,69,82,124,131,144 age was an independent variable for predicting response. Because of the different methods and different definitions of response used in these articles, no summary statement about the relation of age to response is possible. Data could not be pooled to provide estimates of response according to different age categories. Even though younger patients were demonstrated to have more frequent responses in these 14 studies, most of the older patients also responded to splenectomy. Seventeen other case series reported no correlation of age with response, or the data were not interpretable.
Previous response to glucocorticoids was correlated with response in 11 case series, but in all 7 case series that analyzed multiple variables in a multivariate model,5,67,69,82,124,131,144 previous response to glucocorticoids was not an independent variable for predicting response. Previous response to intravenous immunoglobulin correlated with response in only 3 of 7 case series. An influential report that described response to intravenous immunoglobulin as a sensitive (100%) and specific (82%) marker for response to splenectomy was not included in our review because 9 (30%) of the 30 patients were children.145 Only one article reported data on patients who failed both glucocorticoids and intravenous immunoglobulin: 7 of 75 patients failed both treatments; 6 of these 7 patients responded to splenectomy.112
The principal site of platelet sequestration, determined by different radioisotope techniques, correlated with response in 6 case series reporting that patients who had predominant splenic sequestration had a better response than patients whose platelet sequestration was predominantly nonsplenic. However, in the one case series that analyzed multiple variables in a multivariate model and included analysis of platelet sequestration,67 it was not an independent variable for predicting response after taking age into consideration. Also, 9 other case series reported no correlation of the site of platelet sequestration with response, or the data were not interpretable. There was no apparent difference between the reports describing a significant predictive value and those describing no predictive value regarding the year of the report, the isotope used (51Cr or 111In), the source of the platelets (autologous or homologous), or the measurement technique. Even among the reports describing a better response to splenectomy in patients with predominant splenic sequestration of labeled platelets, many patients with nonsplenic sequestration also responded.
Among postoperative prediction variables, the magnitude and rate of the platelet count increase within the first 4 weeks after surgery were often, but inconsistently, reported to correlate with the response at 30 days following splenectomy. No summary statement about the relation of postoperative platelet count recovery to response is possible because the studies reporting a predictive value used different criteria for platelet count levels and time after splenectomy.
Surgical complications
Data are presented separately for laparotomy and laparoscopic splenectomies; case series reporting only adults are combined with case series reporting adults and children. For laparotomy, 81 case series had data for surgical mortality and 35 case series had data for surgical complications. For laparoscopic splenectomy, 29 case series had data for surgical mortality and 19 case series had data for surgical complications. The frequency of death and complications was significantly greater for laparotomy than for laparoscopic splenectomy (Table 4). The earliest reported laparoscopic splenectomy was in 1991.18 To determine whether the decreased rate of death and complications may only reflect advances in surgical practice, the data for laparoscopic splenectomy were compared with the 5 case series reporting splenectomy by laparotomy that accrued patients beginning in 1991 or subsequently.100,102,105,129,140 In these 5 case series, 1 (0.75%) of 134 patients died, a rate of death that is not significantly different from laparoscopic splenectomy (P = .325). To determine whether the decreased rate of death and complications may only reflect patient selection, with laparotomy performed on the more critical patients, the 5 case series that reported results of both laparoscopy and open laparotomy100,102,103,105,140 were analyzed for patient characteristics. In 4 of these case series,100,102,105,140 there was no indication that the patient groups were different. However, in one case series,103 the data suggested that the ITP was more severe in the patients who had splenectomy by open laparotomy.
For laparotomy, the most common reported cause of death was bleeding, accounting for 11 (29%) of the 38 patients for whom a cause of death was reported (Table 4). Intraabdominal bleeding with stroke was the cause of death of 1 of 3 patients who died with laparoscopic splenectomy. Perioperative platelet counts were reported for 5 of the 12 patients who died from bleeding and all were described as less than 16 × 109/L103 or less than 20 × 109/L.38 The clinical importance of complications, such as prolonged hospitalization, readmission to the hospital, or requirement for additional intervention, could not be assessed in most articles.
Discussion
This systematic review documents that splenectomy is an effective treatment for ITP, with two thirds of patients achieving durable complete responses. These results are consistent across 58 years and the 29 countries contributing case series to this review. Because our definitions of partial response and no response could have included patients who had increased platelet counts and required no further treatment, the data on complete responses may underestimate the benefit of splenectomy.
The durability of the responses is supported by the lack of correlation between the rate of complete responses and the duration of follow-up in 85 case series with follow-up durations of 1 month to more than 12 years (Figure 2). Because relapses of ITP following response to splenectomy do occur, yet the rate of complete responses did not change over time in these case series, the occurrence of relapses may be balanced by the occurrence of late remissions, perhaps related to splenectomy or to other treatments, or perhaps occurring spontaneously. The influence of other treatments could not be assessed in these articles. Two case series suggesting that responses to splenectomy are not durable had follow-up durations of more than 7 years in selected patients,5,6 longer than most case series that we reviewed. Therefore, it is possible that publication of more case series with longer follow-up will demonstrate a decreasing frequency of complete remissions over time. However, at this time, the published patient data, including analysis of 21 case series with follow-up of more than 5 years, suggest that the response to splenectomy is durable. Although a continuing occurrence of relapses was suggested in many of these case series, our data did not clearly demonstrate increasing rate of relapse with longer follow-up. Perhaps this unexpected observation is related to the fact that each case series is a single point in time and may reflect different methods of follow-up and different definitions of relapse used in the different studies.
Among all of the prediction variables tested that are available before splenectomy, younger age was most often found to be associated with response (Table 3). In all 7 studies that analyzed variables in a multivariate model, younger age was an independent variable for predicting response. Younger age is also suggested as a predictor for better response by the greater frequency of complete remissions in case series that included children than in case series reporting only adults (Table 2). An equal number of studies demonstrated no correlation of age with response to splenectomy. Furthermore among the studies that did demonstrate a correlation, there was no consistent age that distinguished responders from nonresponders.
Six studies measuring the site of platelet sequestration reported a correlation of predominant splenic sequestration with response; however, 8 other studies reported no correlation, and one study was not interpretable (Table 3). Because these studies used different techniques, because the investigators may have had different levels of experience, and because patients with different clinical characteristics may have been studied, it may not be appropriate to consider these 15 reports as equivalent. However, because these assessments are often qualitative, rather than quantitative, reproducibility among different institutions, even with the same technique, may be difficult.
Although it is possible that some combination of preoperative characteristics may better predict the response to splenectomy, many patients without positive predictive characteristics also respond. Because most patients have a good response to splenectomy, the ability to predict the response may be more difficult and less important.
The decision for splenectomy must be carefully balanced by consideration of the potential risks, because the rate of complications following splenectomy is relatively great. Mortality rates of 0.2% and 1.0%, with laparoscopy and open laparotomy, respectively, are similar to the mortality due to bleeding estimated from large case series of patients with severe ITP followed for 5 to 10 years: 2 (1.6%) of 124 patients8 and 1 (0.4%) of 245 patients.9 However, this comparison may not be appropriate because patients with ITP sufficiently severe to require splenectomy may have a greater risk of death from bleeding than the overall rates reported in these large series. The mortality rate for laparoscopic splenectomy may be more representative of current surgical practice, because assessment of the 5 case series of laparotomy that accrued patients since 1991, the year of the first report of laparoscopic splenectomy, demonstrated no significant difference in mortality from laparoscopy. Although the complication rates of 9.6% and 12.9%, with laparoscopy and open laparotomy, respectively, may seem high, they are consistent with a case series8 that described serious complications, resulting in prolonged hospitalization or readmission, in 20 (26%) of 78 patients. These relatively high rates of death and complications, despite advances in anesthesia and surgical care, may be due to the increasing recognition of ITP among older persons,9,146 the greater risk of surgical complications in older patients,8 and the willingness of surgeons to perform surgery in older patients. It is possible that complications with open laparotomy are greater because it may be performed more often in more severely affected patients, when direct visualization of operative bleeding is preferred.
The risks of splenectomy may be greater than described in this systematic review, because we did not evaluate long-term risks of sepsis and thrombosis. The risk of fatal infection attributed to the absence of a spleen has been estimated to be 0.73 per 1000 patient-years14 ; in this study of patients with hereditary spherocytosis, 3 of the 4 deaths occurred 18 to 30 years following splenectomy,14 well beyond the follow-up time for most patients who were analyzed in this review. An increased risk of thrombosis has also been reported for patients following splenectomy,15-17 but these complications, similar to severe sepsis, may be rare, may be related to multiple risk factors, and may only become apparent many years after splenectomy.15-17
Because of the risks of splenectomy, intermittent glucocorticoid treatment is often continued with the hope that a remission will eventually occur, or other therapies are considered as an alternative to splenectomy. But the risks of these therapies may be substantial and their benefit is uncertain. Glucocorticoid treatment of even short duration may increase the risk of opportunistic infections, such as aspergillosis.147 In a large case series,8 deaths from infections related to immunosuppressive treatment were more frequent than death as a result of bleeding. For none of these therapies has efficacy been established by prospective controlled studies with clinical outcome measures and long-term follow-up.11 Therefore, alternatives to splenectomy may have similar risk but less benefit.
This systematic review has several weaknesses. The reviewed articles used diverse criteria to evaluate patient characteristics and to report outcomes; therefore, averaging data across studies may not be appropriate. Even the comparison of the results of individual studies, as in our description of predictors of response, may give an inaccurate impression because of different methodologies. Children may have been included in some of the case series that we described as reporting only adults and could have biased descriptions of predictors of response and the durability of responses. The severity of surgical complications could not be quantitatively described. Follow-up duration was often difficult to estimate because of inability to account for all patients in the reported case series. The duration of follow-up may not have been long enough to provide a valid estimate of the rate of relapse and was certainly not long enough to evaluate the risks of overwhelming sepsis and thrombosis that may occur many years after splenectomy.14-17
The strength of this review is that the comprehensive and critical analysis of all published reports on splenectomy for ITP over 38 years using a defined and reproducible methodology achieves a balance that is not possible in individual case series and selective reviews. These data provide the best current estimate for the benefits and risks of splenectomy. Although surgical risks are important, splenectomy provides effective long-term benefit for adult patients with ITP who do not achieve durable remissions with initial glucocorticoid treatment.
Prepublished online as Blood First Edition Paper, June 24, 2004; DOI 10.1182/blood-2004-03-1168.
Supported by the Daisy Foundation, Glen Ellen, CA