Abstract
Angiopoietin (Ang) signaling plays a role in angiogenesis and remodeling of blood vessels through the receptor tyrosine kinase Tie2, which is expressed on blood vessel endothelial cells (BECs). Recently it has been shown that Ang-2 is crucial for the formation of lymphatic vasculature and that defects in lymphangiogenesis seen in Ang-2 mutant mice are rescued by Ang-1. These findings suggest important roles for Ang signaling in the lymphatic vessel system; however, Ang function in lymphangiogenesis has not been characterized. In this study, we reveal that lymphatic vascular endothelial hyaluronan receptor 1-positive (LYVE-1+) lymphatic endothelial cells (LECs) express Tie2 in both embryonic and adult settings, indicating that Ang signaling occurs in lymphatic vessels. Therefore, we examined whether Ang-1 acts on in vivo lymphatic angiogenesis and in vitro growth of LECs. A chimeric form of Ang-1, cartilage oligomeric matrix protein (COMP)-Ang-1, promotes in vivo lymphatic angiogenesis in mouse cornea. Moreover, we found that COMP-Ang-1 stimulates in vitro colony formation of LECs. These Ang-1-induced in vivo and in vitro effects on LECs were suppressed by soluble Tie2-Fc fusion protein, which acts as an inhibitor by sequestering Ang-1. On the basis of these observations, we propose that Ang signaling regulates lymphatic vessel formation through Tie2. (Blood. 2005;105:4649-4656)
Introduction
Several endothelial cell growth factors have thus far been identified as essential for vascular development, based primarily on gene-targeting approaches. Among these factors, members of the angiopoietin (Ang) family are ligands for the receptor tyrosine kinase Tie2.1,2 The first member of the family, Ang-1, activates Tie2 receptors expressed on vascular endothelial cells and functions as a positive regulator of angiogenesis and of remodeling and stabilization of blood vessels.3 In contrast, the second member of the Ang family, Ang-2, plays a role in the context of vessel regression as a negative regulator of angiogenesis by blocking Tie2 activation.4 Loss of function assays in mice have revealed that Ang-1 is essential for embryonic vascular development,3 whereas Ang-2 is dispensable for embryonic angiogenesis but required for normal postnatal vascular remodeling.5 These findings indicate different roles for Ang-1 and Ang-2 in blood vessel formation.
The recent discovery of lymphatic endothelial cell (LEC) markers and factors regulating the development of lymphatic vessels has shed new light on the molecular mechanisms underlying lymphangiogenesis.6,7 More recent findings from mice with targeted mutations in Ang-2 indicate that Ang-2 loss results in profound defects in patterning and function of the lymphatic vasculature, indicating that Ang-2 is crucial for lymphatic vessel development.5 Interestingly, defects in lymphatics seen in Ang--/- mice are completely rescued by Ang-1, suggesting a possible role for Ang in lymphangiogenesis. Moreover, this finding suggests an important role for Ang signaling in the formation of the lymphatic vessel system. Tie2 expression in LECs has not yet been demonstrated directly. Moreover, Ang-1 function in lymphatic vessels has not been clarified because Ang-1 homozygous mutants undergo early embryonic death.3
In this study, we focused our investigation on the role of Ang-1 in lymphatic vessels. We show that Tie2 is expressed on lymphatic vascular endothelial hyaluronan receptor 1-positive (LYVE-1+) LECs both embryonically and in the adult, suggesting a direct effect of Ang-1 on lymphatic vessels. Ang-1 enhances in vitro colony formation of CD45-CD31+CD34-/lowLYVE-1+ LECs on OP9 feeder cells. Furthermore, Ang-1 promotes in vivo lymphatic vessel formation in addition to angiogenesis in mouse cornea. On the basis of these findings, we report here that Ang-1 displays potent lymphangiogenic activity.
Materials and methods
Generation of anti-mouse LYVE-1 monoclonal antibodies
A 684-bp fragment of murine LYVE-1 cDNA (position 241-924 in GeneBank clone AJ311501) encoding the extracellular domain of LYVE-18 fused at the C-terminus to the FLAG epitope was subcloned into pCEP4 (Invitrogen, Groningen, the Netherlands). HEK293 cells were transfected with pCEP-LYVE-1-FLAG using FuGENE 6 (Roche Diagnostics, Mannheim, Germany), and cells were selected in 300 μg/mL Hygromycin B (Invitrogen, Carlsbad, CA) for 5 days. LYVE-1-FLAG fusion proteins were purified by an affinity gel and detected by Western blotting analysis with an anti-FLAG antibody (M2), as previously described.9 Rat monoclonal antibodies (mAbs) against the mouse LYVE-1-FLAG fusion protein were generated as described elsewhere.10 In brief, 8-week-old Wistar rats were first immunized subcutaneously with 100 μg LYVE-1-FLAG protein in complete Freund adjuvant (Difco, Detroit, MI) followed by 3 subcutaneous injections of 100 μg LYVE-1-FLAG protein in Freund incomplete adjuvant (Difco) in alternating weeks and a final intravenous boost of 150 μg LYVE-1-FLAG protein. Three days after the boost, spleen cells were harvested and fused with mouse myeloma X63Ag8 cells. Undiluted supernatants from hybridomas were screened by enzyme-linked immunosorbent assay (ELISA) plates coated with 2 μg/mL LYVE-1-FLAG protein. Positive hybridomas were cloned using the limiting dilution technique. ELISA plates were first incubated with undiluted conditioned medium from monoclonal hybridomas. To select monoclonal hybridomas reacting positively to LYVE-1-FLAG protein, binding was detected by horseradish peroxidase (HRP)-conjugated goat anti-rat IgG(H+L) antibody (Biosource, Camarillo, CA). Plate-bound enzymic activity was detected by tetramethylbenzidine (TMB) substrate-chromogen (Dako, Carpinteria, CA), and the absorbance of each well was measured using a microplate reader (model 550; Bio-Rad, Hercules, CA). Three clones of LYVE-1 mAbs were established: ALY2, 7, and 9.
Transfections
To generate the murine pro-B BaF cell line expressing mouse LYVE-1 (BaF/LYVE-1), BaF cells were transfected with a full-length mouse LYVE-1 cDNA in the retroviral vector pMY-ires-GFP (kindly provided by Dr Toshio Kitamura, University of Tokyo, Japan). As controls, BaF cells were transfected with empty vector (BaF/GFP). Transfected cells were purified as green fluorescent protein-positive (GFP+) cells by sorting using fluorescence-activated cell sorting (FACS Calibur; Becton Dickinson, San Jose, CA). Retroviral infection, collection, and purification procedures were performed as previously described.11
Immunohistochemistry and LacZ staining
E14 mouse embryos were stained by whole-mount immunohistochemistry with 1:200 diluted anti-mouse LYVE-1 antibody (ALY7) as previously described.12 To detect lymphatic vessels in E14 mouse embryos, vascular endothelial growth factor receptor 3 (VEGFR-3LacZ/+) embryos13 were stained with X-gal as described previously.12 Intestine tissue from adult mice and the skin tissues from mouse embryos were fixed with 4% paraformaldehyde (PFA), embedded in optimal cutting temperature (OCT) compound (Sakura Finetechnical, Tokyo, Japan) to make frozen tissue specimens, and sectioned at 10 μm. Samples were stained with antiplatelet endothelial cell adhesion molecule 1 (PECAM-1)/CD31 (Mec13.3, rat antimouse mAb; PharMingen, San Diego, CA), anti-CD34 (RAM34; PharMingen), or anti-mouse podoplanin mAb (Cell Science, Canton, MA) at 4°C overnight, followed by staining with secondary antibodies, Alexa 488-conjugated goat anti-rat IgG for detecting anti-PECAM-1/CD31 and anti-CD34 mAb, and Alexa 488-cojugated goat anti-hamster IgG (Molecular Probes, Eugene, OR) for detecting podoplanin mAb. Isotype-matched rat and hamster immunoglobulins were used as control antibodies for ALY7 and podoplanin mAb, respectively. After washing with phosphate-buffered saline (PBS) 5 times, samples were stained with the biotinylated anti-LYVE-1 mAb, ALY7, followed by staining with Alexa 546-conjugated streptavidin. Stained samples were visualized using an FV1000 confocal microscope with UPlanApo 20×/0.70 (Olympus, Tokyo, Japan). Images were further processed with Adobe Photoshop (Adobe Systems, San Jose, CA).
To determine whether Tie2 is expressed in LYVE-1+ LECs, we stained sections from adult mouse intestine with ALY7 and with TEK4 anti-Tie2 mAb.14 To detect Tie2 expression in sections, we used a tyramide signal amplification (TSA) biotin system (Perkin-Elmer, Boston, MA) as previously reported.15,16 Stained samples were analyzed using a fluorescence microscope with UPlanApo 20×/0.70 (Olympus). When HRP-conjugated immunoglobulin was used as the secondary antibody, samples were incubated in PBS containing 0.3 mg/mL diaminobenzidine (DAB; Dojindo Laboratories, Kumamoto, Japan) in the presence of 0.8 mg/mL NiCl2 and 0.01% hydrogen peroxide at room temperature to visualize reactive samples. Samples were viewed using a BX50 microscope with UPlanApo 20×/0.70 (Olympus).
FACS analysis
To identify the characterization of LECs, we performed FACS analyses of mouse embryo cells by FACS Vantage (Becton Dickinson). After removing embryonic liver and spleen microscopically, E11, E13, and E15 mouse embryos were dissected and treated with 2.4 U/mL Dispase (Gibco, Carlsbad, CA) and 0.05% collagenase S-1 (Nitta Gelatin, Osaka, Japan) at 37°C for 30 minutes to produce a single-cell suspension. To exclude erythrocytes from this preparation prior to analysis with FACS Vantage, lymphoprep (Axis-Shield, Oslo, Norway) treatment was performed as previously described.17 Before staining, Fc receptors were blocked with an anti-mouse CD16/CD32 Fc receptor (FcR; PharMingen). First, all cells were stained with CD45-peridinin chlorophyll protein (PerCP) cyanine 5.5 (Cy5.5) to sort CD45- nonhematopoietic cells for further analysis. Subsequently, the cells were incubated with biotinylated ALY7 at 4°C for 30 minutes and then washed 3 times. Then, the cells were reacted with an allophycocyanin-conjugated streptavidin (PharMingen) to visualize LYVE-1+ cells. For double or triple staining, the cells were stained with CD31-phycoerythrin (PE)/FITC, CD34-PE (PharMingen), and TEK4-PE.14
RT-PCR characterization of sorted LECs
RNA was extracted from sorted LECs, blood vessel endothelial cells (BECs), and OP9 stromal cells using RNeasy Mini Kit (Qiagen, Hilden, Germany). RNA was reverse transcribed using an Advantage RT-for-PCR Kit (Clontech, Palo Alto, CA), and the product was used for further analysis. The sequences of primers for reverse transcription-polymerase chain reaction (RT-PCR) were as follows: (1) LYVE-1, sense: 5′-TTCCTCGCCTCTATTTGGAC-3′, antisense: 5′-ACGGGGTAAAATGTGGTAAC-3′; (2) Prox1, sense:5′-AAGTGGTTCAGCAATTTCCG-3′, antisense: 5′-TGACCTTGTAAATGGCCTTC-3′; (3) podoplanin, sense: 5′-GTGCCAGTGTTGTTCTGGGT-3′, antisense: 5′-TCTGTTGTCTGCGTTTCATCC-3′; (4) VEGFR-3, sense: 5′-GCTACCACTGCTACTACAAG-3′, antisense:5′-GATAATCCCAGTCGAAGGTG-3′; (5) secondary lymphoid chemokine (SLC), sense:5′-GATGATGACTCTGAGCCTCC-3′, antisense: 5′-CTCTTGAGGGCTGTGTCTGT-3′; (6) CD31/PECAM-1, sense: 5′-CAAACCGTATCTCCAAAGCC-3′; antisense:5′-TCTGTGAATGTTGCTGGGTC-3′; (7) Tie2, sense:5′-TTAGTTCTCTGTGGAGTCAG-3′, antisense: 5′-AGGCCCTGAGTTCTTCACTC-3′; (8) neuropilin-1, sense:5′-ACTGACAGCGCAATAGCAAAAGAAG-3′, antisense: 5′-TCGGACAAATCGAGTTATCAGTGGT-3′; (9) VEGFR-1 (Flt-1), sense: 5′-TGTGGAGAAACTTGGTGACCT-3′, antisense: 5′-TGGAGAACAGCAGGACTCCTT-3′; (10) VEGFR-2 (Flk-1), sense: 5′-AGAACACCAAAAGAGAGGAACG-3′, antisense: 5′-GCACACAGGCAGAAACCAGTAG-3′; (11) CD44, sense:5′-CACAGCAGCAGATCGATTTG-3′, antisense: 5′-GAGGAGCTGAGGCATTGAAG-3′; (12) Tie1, sense: 5′-CGAAGAAGCTGCCTACATCGGAG-3′, antisense: 5′-CTAGGCCTCCTCAGCTGTGGCAT-3′; and (13) glyceraldehyde phosphate dehydrogenase (GAPDH), sense: 5′-AATCCCATCACCATCTTCCA-3′, antisense: 5′-CCAGGGGTCTTACTCCTTG-3′. PCR products were separated on a 1.2% agarose gel and stained with ethidium bromide.
Primary LEC coculture on OP9 feeder layers
To examine the effect of Ang-1 on LEC growth, sorted LECs were cultured on OP9 stromal cells in RPMI 1640 (Gibco) with 10% fetal calf serum (FCS) and 10-5 M 2-mercaptoethanol (Sigma, St Louis, MO) with or without COMP-Ang-1 (500 ng/mL), in which the N-terminal portion of Ang-1 is replaced with the short coiled-coil domain of cartilage oligomeric matrix protein (COMP). COMP-Ang-1 is a soluble, stable, and potent Ang-1 variant.18 To determine whether Ang-1 has an effect on LEC via Tie2 receptors, sorted LECs were cultured with COMP-Ang-1 (500 ng/mL) and recombinant soluble Tie2-Fc fusion protein4 (30 μg/mL), recombinant soluble VEGFR-3-Fc fusion protein (50 μg/mL; R&D Systems, Minneapolis, MN), or CD4-Fc fusion protein17 (50 μg/mL). Seven days later, the dishes were immunostained with anti-LYVE-1 mAb (ALY7) to detect LECs. To quantify the effects of Ang-1 signaling on the colony formation of LECs, we counted the number of LYVE-1+ endothelial cell colonies that formed the compact type.
Mouse corneal assay for in vivo lymphangiogenesis
A mouse corneal assay was performed as previously described.19,20 In brief, under sterile conditions, slow-release pellets were prepared incorporating COMP-Ang-1 (300 ng), PBS (4 μL), or COMP-Ang-1 (300 ng) together with either 500 ng of a soluble Tie2-Fc fusion protein (R&D Systems) or 500 ng soluble CD4-Fc protein in a casting solution of an ethynyl-vinyl copolymer (Elvax-40; DuPont, Wilmington, DE) in 10% methylene chloride. After anesthesia with sodium pentobarbital (Dainippon Pharmaceutical, Osaka, Japan), the pellet was implanted into the corneal micropocket of female 8-week-old C57BL/6 mice (SLC, Shizuoka, Japan). Erythromycin ophthalmic ointment was applied to each eye after implantation of the pellet. Seven days after implantation, enucleated eyes were fixed in 4% PFA, dehydrated, embedded in OCT compound, and sectioned radially in parallel to the growing limbal vessels. To visualize the lymphatic vessel formation in mouse cornea, sections were immunostained with biotinylated anti-LYVE-1 mAb (2 μg/mL, ALY7) and then incubated with HRP-conjugated streptavidin (Dako, Glostrup, Denmark). Peroxidase staining was performed by incubating sections in 0.3 mg/mL DAB (Dojindo Laboratories), 0.8 mg/mL NiCl2, and 0.05% hydrogen peroxide in PBS.
The lymphangiogenic grading scores were based on the distance of vessel growth from the limbus analyzed by immunohistochemistry as described elsewhere.21 In brief, sequential sections through the eyes were immunostained for LYVE-1. The grading was: 0, no corneal lymphatic vessels, including cases with growth of lymphatic vessels in the limbus only; 5, corneal lymphatic vessels penetrating into the pellet; and 1 to 4, the corneal area between the limbus and the pellet was divided into equal segments that were scored as 1 to 4 in the order of distance from the limbus. To visualize the COMP-Ang-1-induced lymphatic and blood vessel at the same time in mouse cornea, sections were stained with anti-PECAM-1/CD31 (Mec13.3) at 4°C overnight, followed by staining with a secondary antibody, Alexa 488-conjugated goat anti-rat IgG. After washing with PBS 5 times, samples were stained with biotinylated anti-LYVE-1 mAb (ALY7), followed by staining with Alexa 546-conjugated streptavidin.
Statistics
Data are expressed as the mean plus or minus SD or the mean plus or minus SEM. Statistical analysis was conducted using Student t test. Statistical significance was defined as P less than .05.
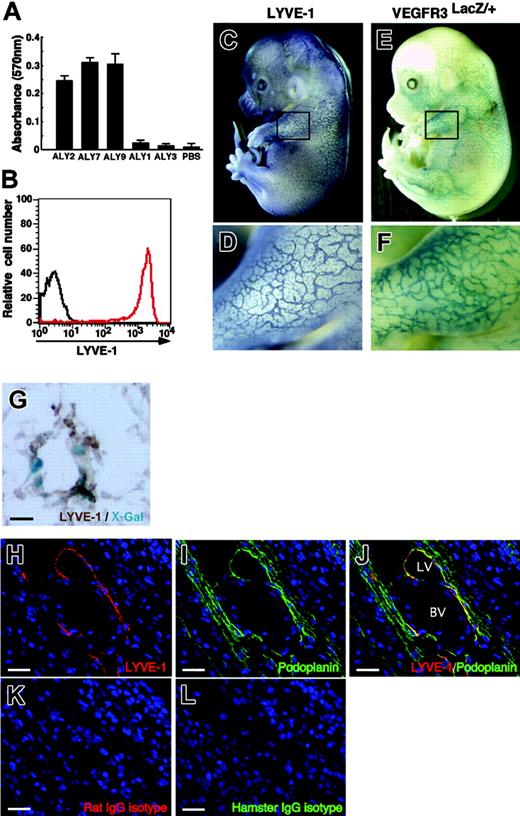
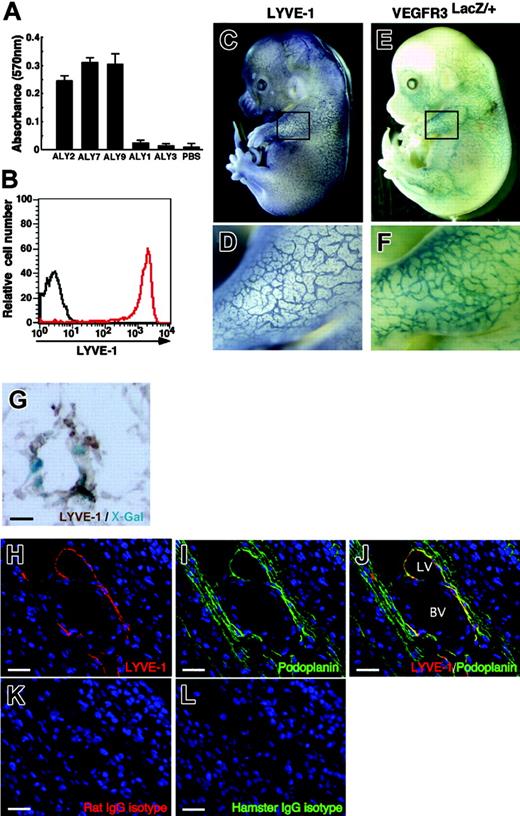
Specificity of novel antibodies (ALY) against mouse LYVE-1. (A) ALY2, 7, and 9 binding to LYVE-1-FLAG revealed by ELISA. Shown is absorbance at 570 nm. Each bar represents the mean ± SD of triplicate assays. (B) ALY7 binding to BaF/LYVE-1 cells was analyzed by FACS. The red line denotes binding of ALY7 to BaF/LYVE-1 cells. The black line denotes background staining seen when BaF/LYVE-1 cells were incubated with only secondary antibodies. (C-D) LECs in mouse embryos at E14.0 were analyzed by whole-mount immunohistochemical staining with anti-LYVE-1 mAb (ALY7). (E-F) The lymphatic endothelial network in E14.0 mouse embryos is visualized by VEGFR-3 expression. To detect VEGFR-3 expression, whole-mount X-gal staining of VEGFR-3LacZ/+ mouse embryos was performed. Panels D and F show higher magnification views of the black square in the upper limb region of panels C and E, respectively. (G) Section of lymphatic vessel in the skin tissues of the VEGFR-3LacZ/+ embryo. X-Gal staining shows that VEGFR-3+ (blue color) are identical to LYVE-1+ (brown color). (H-L) Section of the E14.0 mouse vascular system in body wall was stained with ALY7 (red color in panels H and J), anti-mouse podoplanin mAb (green color in panels I and J), isotype-matched rat IgG (red color in panel K), and isotype-matched hamster IgG (green color in panel L). LYVE-1+ lymphatic vessel (LV in panel J) expresses podoplanin, but blood vessel (BV in panel J) does not. Isotype-matched control antibodies showed no staining in any sections (K-L). Scale bars indicate 10 μm (G) and 20 μm (H-L), respectively.
Specificity of novel antibodies (ALY) against mouse LYVE-1. (A) ALY2, 7, and 9 binding to LYVE-1-FLAG revealed by ELISA. Shown is absorbance at 570 nm. Each bar represents the mean ± SD of triplicate assays. (B) ALY7 binding to BaF/LYVE-1 cells was analyzed by FACS. The red line denotes binding of ALY7 to BaF/LYVE-1 cells. The black line denotes background staining seen when BaF/LYVE-1 cells were incubated with only secondary antibodies. (C-D) LECs in mouse embryos at E14.0 were analyzed by whole-mount immunohistochemical staining with anti-LYVE-1 mAb (ALY7). (E-F) The lymphatic endothelial network in E14.0 mouse embryos is visualized by VEGFR-3 expression. To detect VEGFR-3 expression, whole-mount X-gal staining of VEGFR-3LacZ/+ mouse embryos was performed. Panels D and F show higher magnification views of the black square in the upper limb region of panels C and E, respectively. (G) Section of lymphatic vessel in the skin tissues of the VEGFR-3LacZ/+ embryo. X-Gal staining shows that VEGFR-3+ (blue color) are identical to LYVE-1+ (brown color). (H-L) Section of the E14.0 mouse vascular system in body wall was stained with ALY7 (red color in panels H and J), anti-mouse podoplanin mAb (green color in panels I and J), isotype-matched rat IgG (red color in panel K), and isotype-matched hamster IgG (green color in panel L). LYVE-1+ lymphatic vessel (LV in panel J) expresses podoplanin, but blood vessel (BV in panel J) does not. Isotype-matched control antibodies showed no staining in any sections (K-L). Scale bars indicate 10 μm (G) and 20 μm (H-L), respectively.
Results
Generation of anti-mouse LYVE-1 mAbs
After screening more than 600 clones by ELISA for the immunoreaction to LYVE-1-FLAG protein, 3 monoclonal clones, ALY2, ALY7, and ALY9 (isotype IgG1), were isolated (Figure 1A). To investigate whether these mAbs react with cells expressing mouse LYVE-1, we transfected BaF cells, which express no endogenous LYVE-1, with mouse LYVE-1 (BaF/LYVE-1) or empty vector (BaF/GFP) using a retroviral vector. All ALYs reacted with BaF/LYVE-1 but not with BaF/GFP (Figure 1B). Binding of ALYs to BaF/LYVE-1 was completely inhibited when BaF/LYVE-1 cells were preincubated with LYVE-1-FLAG recombinant protein (data not shown), suggesting that all 3 ALYs specifically detect the extracellular domain of mouse LYVE-1. We evaluated the specificity of ALYs to detect lymphatics in vivo in E14 embryos and compared the LYVE-1 staining patterns with the expression of VEGFR-3, a specific lymphatic endothelial marker at this stage,22 which was detected as LacZ expression in VEGFR-3LacZ/+ mouse embryos. ALYs detected lymphatic vessels in embryonic skin tissues and that expression pattern was similar to that of VEGFR-3, suggesting that ALYs specifically stain lymphatic vessels (Figure 1C-F). Immunohistochemistry with ALY antibody of LacZ-stained sections from VEGFR-3LacZ/+ mouse embryos revealed that VEGFR-3+ lymphatic vessels express LYVE-1 (Figure 1G). Furthermore, LYVE-1+ cells express podoplanin,23 which is selectively expressed within the lymphatic endothelial cells (despite expression in some other cells, such as in the kidney podocytes and lung alveolar type 1 cells) but not within the blood vessel cells (Figure 1H-J and Figure S1, available on the Blood website; see the Supplemental Figures link at the top of the online article). Taken together, we conclude that ALY2, 7, and 9 are specific to LYVE-1 and can be used for immunostaining and flow cytometric analysis as a markers for LECs. Because the 3 clones showed essentially equivalent outcomes, we used ALY7 as LYVE-1 mAb in the following studies.
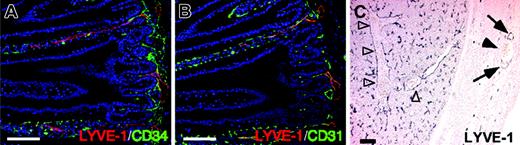
LYVE-1 expression in LECs and liver sinusoids. Immunostaining with ALY7 in tissue sections (A-B). Cross-sections were prepared from intestines of adult mice and stained with ALY7 (red color in panels A-B), CD34 mAb (green color in panel A), and CD31/PECAM-1 mAb (green color in panel B). Note that all LYVE-1+ cells are also CD31+ but CD34-. (C) Cross-sections were prepared from liver of E14 embryos and stained with ALY7. Note that ALY7 specifically detects lymphatic vessels (arrows) but not blood vessels (arrowheads). ALY7 also detects sinusoid blood vessels in liver (open arrowheads). Scale bars indicate 100 μm.
LYVE-1 expression in LECs and liver sinusoids. Immunostaining with ALY7 in tissue sections (A-B). Cross-sections were prepared from intestines of adult mice and stained with ALY7 (red color in panels A-B), CD34 mAb (green color in panel A), and CD31/PECAM-1 mAb (green color in panel B). Note that all LYVE-1+ cells are also CD31+ but CD34-. (C) Cross-sections were prepared from liver of E14 embryos and stained with ALY7. Note that ALY7 specifically detects lymphatic vessels (arrows) but not blood vessels (arrowheads). ALY7 also detects sinusoid blood vessels in liver (open arrowheads). Scale bars indicate 100 μm.
LYVE-1 expression in LECs and sinusoidal endothelial cells
To further characterize LYVE-1-positive cells, we performed double staining of LECs and BECs in intestines of adult mice with ALY7 and either CD34 as the BEC marker or CD31/PECAM-1 as the pan-endothelial cell marker. Immunohistochemistry revealed that ALY7 was detected in only intestinal lacteal lymphatics, and not in the blood vessel capillary network. ALY-stained lymphatic vessels express CD31 but not CD34 (Figure 2A-B). Sinusoid blood vessels, which contain many erythrocytes in liver and spleen, also strongly expressed LYVE-1 (Figure 2C and data not shown), which was consistent with a previous report.24
Tie2 expression in intestinal lymphatics
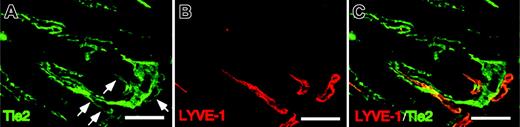
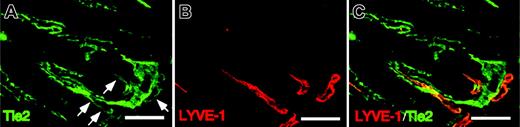
To determine whether the Tie2 receptor is expressed in LECs, we performed immunohistochemistry in intestinal lymphatic vessels using anti-Tie2 antibody. Immunohistochemistry in mouse intestine revealed that Tie2 receptors are expressed in the lacteal lymphatics (Figure 3A-C). Tie2 expression in LECs was significantly weaker than expression seen in BECs. Based on these findings, we speculate that Ang signaling through Tie2 receptors regulates the maintenance of lymphatics or lymphangiogenesis or both.
Tie2 expression in LYVE-1+LECs. (A-C) Cross-sections from intestines of adult mice were stained with Tie2 mAb (green color in panel A) and ALY7 (red color in panel B). Note the Tie2 immunoreactivity in BECs and the weaker staining seen in LECs (arrows). Double staining of LYVE-1 and Tie2 demonstrates that LYVE-1+ LECs are Tie2+ (merged staining in panel C). Scale bars indicate 100 μm.
Tie2 expression in LYVE-1+LECs. (A-C) Cross-sections from intestines of adult mice were stained with Tie2 mAb (green color in panel A) and ALY7 (red color in panel B). Note the Tie2 immunoreactivity in BECs and the weaker staining seen in LECs (arrows). Double staining of LYVE-1 and Tie2 demonstrates that LYVE-1+ LECs are Tie2+ (merged staining in panel C). Scale bars indicate 100 μm.
Isolation and characterization of LYVE-1+ LECs
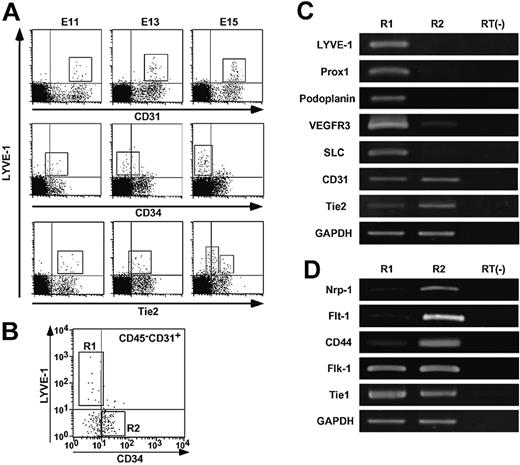
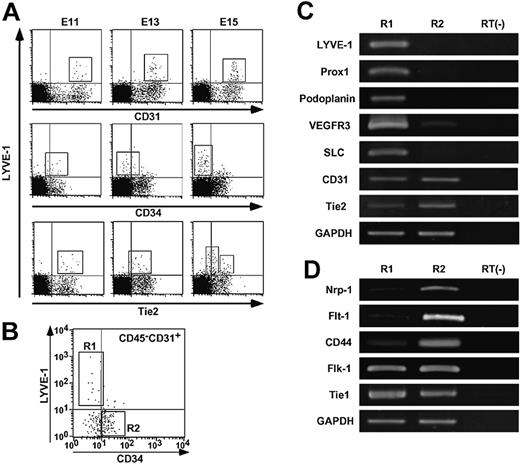
Because immunohistochemistry revealed that LYVE-1+ LECs express high CD31 and low Tie2 but not CD34 in adult mice, we examined the expression patterns of these 3 markers during the course of embryonic development. Based on previous reports that lymphatic vessel development occurs at mid-gestational stages,25,26 we examined the expression of CD31, CD34, and Tie2 in LYVE-1+ LECs derived from E11, E13, and E15 embryos by FACS analysis. LYVE-1+ LECs expressed CD31 as did LYVE-1- BECs throughout embryogenesis (upper panels in Figure 4A). By contrast, CD34 expression was not detected in LYVE-1+ LECs from E15 mouse embryos, whereas LYVE-1+ LECs and LYVE-1- BECs expressed CD34 at E11 (middle panels in Figure 4A). As for Tie2 expression, LYVE-1+ LECs from E11 embryos express Tie2 as do LYVE-1- BECs. LYVE-1+ LECs were further grouped into Tie2 high- or low-positive cells (lower panels in Figure 4A). Based on these findings, we sorted CD45-CD31+CD34-/lowLYVE-1+ cells derived from E14 mouse embryos as LECs (R1 in Figure 4B) and CD45-CD31+CD34+LYVE-1- cells as BECs (R2 in Figure 4B). To determine the purity of the sorted LECs and BECs, we examined the expression of the lymphatic endothelial markers Prox1, podoplanin, and SLC in sorted LECs compared with BECs by RT-PCR analysis (Figure 4C). We confirmed that LYVE-1 is expressed only in the R1 but not the R2 fraction, indicating that ALY7 specifically reacts to endogenous LYVE-1. The expression of Prox1, podoplanin, and SLC as lymphatic endothelial markers was restricted to cells in the R1 fraction. VEGFR-3 expression was detected in the cells in both the R1 and R2 fractions, and its expression in the cells in the R1 fraction was higher than that in the R2 fraction. These observations for the molecular characterization of CD45-CD31+CD34-/lowLYVE-1+ cells (R1) are consistent with previous reports27,28 describing the characterization of LECs, indicating the presence of high purity LECs in the R1 fraction. Immunohistochemistry revealed that LYVE-1+ LECs expressed CD31 as did LYVE-1- BECs, whereas Tie2 expression in LECs was significantly lower than that seen in BECs (Figures 2, 3). Therefore, we compared the levels of Tie2 expression in LECs in the R1 fraction with those of cells in the R2 fraction by RT-PCR analysis. CD31 expression was not different between them, whereas significantly low expression of Tie2 was detected in LECs. Next, we examined the expression of neuropilin-1, Flt-1, CD44, Flk-1, and Tie1, all of which were shown to be abundantly expressed in BECs27,28 by RT-PCR analysis (Figure 4D). LYVE-1+ LECs expressed Flk-1 and Tie1 as did LYVE-1- cells. Abundant expression of neuropilin-1, Flt-1, and CD44 was detected in the R2 fraction, whereas faint transcripts of these genes were detected in LECs in the R1 fraction. These results indicate that cells in the R1 and R2 fractions consist of a highly pure LECs and BECs, respectively.
Lower expression level of Tie2 in LECs. (A) FACS analysis of the distribution of LYVE-1+ cells in embryogenesis. E11, E13, and E15 mouse embryos were dissected. Embryonic liver and spleen tissues were excluded microscopically, and other tissues were treated with collagenase and Dispase and used as a source of material for FACS analysis. These cells were double-stained with ALY7 and CD31/PECAM-1 mAb (upper panels), CD34 mAb (middle panels), and anti-Tie2 mAb (lower panels). Box in each panel indicates LYVE-1+ LEC fraction, respectively. (B) Expression of CD34 and LYVE-1 in CD45-CD31+ cells. mRNA was isolated from LYVE-1+ CD34low/- as LECs (R1) or LYVE-1- CD34+ as BECs (R2) from E14 embryos in which liver and spleen tissues were excluded. (C-D) RT-PCR analysis of genes served as markers for LECs/BECs. mRNA from nonsorted cells without reverse-transcriptase treatment was used as a negative control (RT(-)). (C) Note that expression of Prox1, podoplanin, and secondary lymphoid chemokine (SLC) was restricted to R1, and VEGFR-3 expression was very high in R1, whereas CD31/PECAM-1 was expressed equally in both R1 and R2. Tie2 expression in R1 was weaker than that seen in R2. (D) Note that expression of neuropilin-1 (Nrp-1), Flt-1, and CD44 was very high in R2, but faint in R1. In contrast, the transcript of both Flk-1 and Tie1 was equally expressed in R1 and R2, respectively. Expression of GAPDH was used as an internal control.
Lower expression level of Tie2 in LECs. (A) FACS analysis of the distribution of LYVE-1+ cells in embryogenesis. E11, E13, and E15 mouse embryos were dissected. Embryonic liver and spleen tissues were excluded microscopically, and other tissues were treated with collagenase and Dispase and used as a source of material for FACS analysis. These cells were double-stained with ALY7 and CD31/PECAM-1 mAb (upper panels), CD34 mAb (middle panels), and anti-Tie2 mAb (lower panels). Box in each panel indicates LYVE-1+ LEC fraction, respectively. (B) Expression of CD34 and LYVE-1 in CD45-CD31+ cells. mRNA was isolated from LYVE-1+ CD34low/- as LECs (R1) or LYVE-1- CD34+ as BECs (R2) from E14 embryos in which liver and spleen tissues were excluded. (C-D) RT-PCR analysis of genes served as markers for LECs/BECs. mRNA from nonsorted cells without reverse-transcriptase treatment was used as a negative control (RT(-)). (C) Note that expression of Prox1, podoplanin, and secondary lymphoid chemokine (SLC) was restricted to R1, and VEGFR-3 expression was very high in R1, whereas CD31/PECAM-1 was expressed equally in both R1 and R2. Tie2 expression in R1 was weaker than that seen in R2. (D) Note that expression of neuropilin-1 (Nrp-1), Flt-1, and CD44 was very high in R2, but faint in R1. In contrast, the transcript of both Flk-1 and Tie1 was equally expressed in R1 and R2, respectively. Expression of GAPDH was used as an internal control.
Enhanced in vitro colony formation of LECs in the presence of COMP-Ang-1
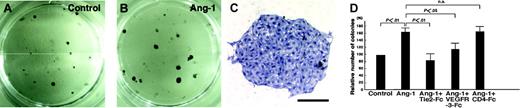
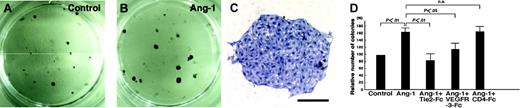
Because CD45-CD31+CD34-/lowLYVE-1+ LECs (R1 in Figure 4B) from E14 embryos express Tie2, we cultured them on OP9 stromal cells29 to determine whether Ang-1 signaling promotes in vitro growth of LECs. After 7 days in culture, colonies on OP9 stromal cells express LYVE-1, suggesting that colonies consist of LYVE-1+ LECs (Figure 5A and Figure S2A). Prox1 was also expressed in colonies expressing LYVE-1, indicating that colonies consist of lymphatic endothelial cells (Figure S2A-D). Notably, the addition of COMP-Ang-1 to the culture resulted in increases in LYVE-1+ colony formation, suggesting that Ang-1 signaling enhances the growth of LECs (Figure S2A-C). Furthermore, COMP-Ang-1 increases the number of LEC colonies forming on OP9 cells, and this enhancing phenomenon by COMP-Ang-1 was completely suppressed by the addition of recombinant soluble Tie2-Fc fusion protein,4 which contains only the Tie2 ectodomain and thereby acts as an inhibitor by sequestering Ang-1 (Figure 5D). RT-PCR analysis revealed that expression of Ang-2, but not Ang-1, was detected in sorted LECs, whereas expression of Ang-1, but not Ang-2, was detected in OP9 stromal cells. Whereas VEGF-D was expressed in OP9 stromal cells, the expression of VEGF-C in OP9 stromal cells was induced after a coculture with LECs (Figure S3A-C), suggesting that VEGFR-3/VEGF-C or -D signaling might support the growth of LECs in this coculture system. To examine the possibility that COMP-Ang-1 may induce secondary activation of the VEGF-C or -D/VEGFR-3 pathway, we investigated the effects of recombinant soluble VEGFR-3-Fc fusion protein,12 which acts as an inhibitor by sequestering both VEGF-C and VEGF-D, on COMP-Ang-1-induced growth of LECs. A partial inhibitory effect on the COMP-Ang-1-induced growth of LECs was observed by the addition of VEGFR-3-Fc protein.
Increases in the number of colonies of LECs by COMP-Ang-1. (A-B) Lower magnification photographs of ALY7-stained LEC colonies are shown. The number of colonies in cultures supplemented with COMP-Ang-1 (B) increased compared to controls (A). (C) Higher magnification photographs of ALY7-stained LEC colonies. Bar indicates 200 μm. (D) Quantitative analysis of ALY7-stained LEC colony formation. Columns represent the relative number of packed round colonies, which is expressed as a percentage against untreated cultures. LEC colonies in culture with COMP-Ang-1 were significantly increased in number compared to those cultured without COMP-Ang-1. The addition of soluble Tie2-Fc reduced the Ang-1-induced increase in the number of colonies to the basal value, whereas the inhibitory effect of VEGFR-3-Fc protein on the Ang-1-induced increase in the number of colonies was partial. The graphs represent the mean ± SEM (n = 6).
Increases in the number of colonies of LECs by COMP-Ang-1. (A-B) Lower magnification photographs of ALY7-stained LEC colonies are shown. The number of colonies in cultures supplemented with COMP-Ang-1 (B) increased compared to controls (A). (C) Higher magnification photographs of ALY7-stained LEC colonies. Bar indicates 200 μm. (D) Quantitative analysis of ALY7-stained LEC colony formation. Columns represent the relative number of packed round colonies, which is expressed as a percentage against untreated cultures. LEC colonies in culture with COMP-Ang-1 were significantly increased in number compared to those cultured without COMP-Ang-1. The addition of soluble Tie2-Fc reduced the Ang-1-induced increase in the number of colonies to the basal value, whereas the inhibitory effect of VEGFR-3-Fc protein on the Ang-1-induced increase in the number of colonies was partial. The graphs represent the mean ± SEM (n = 6).
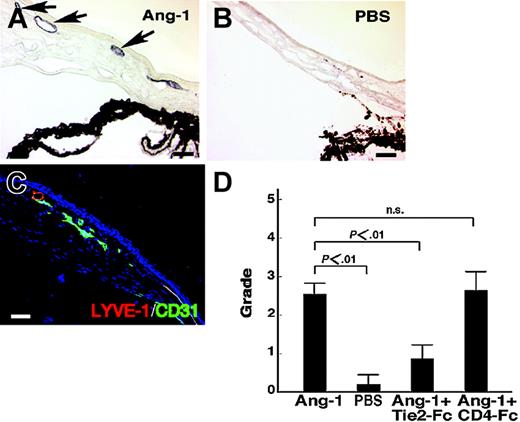
In vivo lymphatic vessel formation in mouse cornea induced by COMP-Ang-1
We next examined whether COMP-Ang-1 promotes chemotaxis of the CD45-CD31+CD34-/lowLYVE-1+ LECs (R1 in Figure 4B) from E14 embryos by a migration assay.9 We found that COMP-Ang-1 promoted the migration of LECs through a microchemotactic membrane. The number of migrated LECs due to induction by COMP-Ang-1 increased approximately 1.4-fold compared with the control that included no COMP-Ang-1 (data not shown). Given this in vitro finding that COMP-Ang-1 might promote the chemotactic activity of LECs, we examined whether COMP-Ang-1 acts on in vivo lymphatic vascular formation using a corneal neovascularization assay. In this assay, COMP-Ang-1 induced neovascularization in the mouse cornea, whereas a pellet containing PBS alone did not promote neovascularization, as previously reported.18 Because the lymphatic vessels are not visible by intra vitam observation, we performed immunohistochemistry and observed in sectioned cornea that a pellet containing COMP-Ang-1 induced lymphatic vessels composed of LYVE-1+ LECs (Figure 6A) and that a pellet containing PBS alone did not (Figure 6B). Double-staining of migrated endothelial cells on the mouse cornea with ALY7 and CD31 revealed that both lymphangiogenesis and angiogenesis were induced by COMP-Ang-1 (Figure 6C). Quantitative estimation of corneal lymphangiogenesis revealed that COMP-Ang-1 induced lymphatic vessel formation on mouse cornea. The soluble Tie2-Fc fusion protein suppressed COMP-Ang-1-induced lymphatic angiogenesis on mouse cornea, whereas the soluble CD4-Fc fusion protein could not inhibit it (Figure 6D). These findings suggest that Ang-1-activated Tie2 signaling may act on lymphatic vessel formation on mouse cornea.
In vivo lymphatic vessel formation in mouse cornea induced by COMP-Ang-1. (A) ALY7 immunostaining of lymphatic vessels of the cornea. COMP-Ang-1-induced migrated endothelial cells on the cornea show positive LYVE-1 immunoreactivity (arrows), indicating that lymphatic vessels were induced by COMP-Ang-1. (B) No obvious LYVE-1+ lymphatic vessel is seen on the cornea with a pellet containing PBS alone. (C) Double-staining of newly formed vessels with ALY (red) and anti-CD31 (green) antibodies. Both of the lymphatic (merged) and blood vessel (green) endothelial cells migrated into the cornea induced by the pellet containing COMP-Ang-1. (D) Quantitative estimation of corneal lymphangiogenesis by immunostaining with ALY7. The lymphangiogenic grading score was determined by a previously described method.10 Recombinant soluble Tie2-Fc protein suppressed the COMP-Ang-1-induced lymphatic vessel formation on mouse cornea, whereas soluble CD4-Fc protein could not inhibit corneal lymphangiogenesis. The graphs represent the mean ± SEM (n = 6); scale bars indicate 50 μm.
In vivo lymphatic vessel formation in mouse cornea induced by COMP-Ang-1. (A) ALY7 immunostaining of lymphatic vessels of the cornea. COMP-Ang-1-induced migrated endothelial cells on the cornea show positive LYVE-1 immunoreactivity (arrows), indicating that lymphatic vessels were induced by COMP-Ang-1. (B) No obvious LYVE-1+ lymphatic vessel is seen on the cornea with a pellet containing PBS alone. (C) Double-staining of newly formed vessels with ALY (red) and anti-CD31 (green) antibodies. Both of the lymphatic (merged) and blood vessel (green) endothelial cells migrated into the cornea induced by the pellet containing COMP-Ang-1. (D) Quantitative estimation of corneal lymphangiogenesis by immunostaining with ALY7. The lymphangiogenic grading score was determined by a previously described method.10 Recombinant soluble Tie2-Fc protein suppressed the COMP-Ang-1-induced lymphatic vessel formation on mouse cornea, whereas soluble CD4-Fc protein could not inhibit corneal lymphangiogenesis. The graphs represent the mean ± SEM (n = 6); scale bars indicate 50 μm.
Discussion
In the present study we generated anti-mouse LYVE-1 mAbs and found that LYVE-1+ mouse primary LECs express the Tie2 receptor, and that Ang-1 enhances in vitro colony formation of LECs and induces in vivo lymphatic vessel formation in mouse cornea. Based on these findings, we propose that Ang-1 is a factor that promotes lymphangiogenesis.
In contrast to the rapid progress made in determining the molecular mechanisms underlying blood vessel formation, the factors controlling the normal development of lymphatic vessels have only recently been identified.6 Among these factors, the homeobox gene Prox126 is a key regulator of the commitment of endothelial cells to differentiate as LECs, and VEGF-C is essential for sprouting of the first lymphatic vessels from embryonic veins.30 Furthermore, VEGF-C/VEGFR-3 signaling plays a pivotal role in the process of developmental and pathologic lymphangiogenesis.6,7 Alitalo et al have shown that VEGFR-3-Ig transgenic mice, in which endogenous VEGFR-3 signaling is inhibited by sequestering the VEGFR-3-specific ligands VEGF-C and -D, exhibit severe defects in lymphatic vessel development during the embryonic and neonatal stages, although the lymphatic vessel networks in adult mice are partially developed.31 This finding suggests the presence of other signaling molecules in later processes of lymphatic vessel formation, such as the remodeling and maturation of lymphatic vessels. Angs are known to be key regulators of BEC functions required for angiogenesis, remodeling, and maturation of blood vessels, but not for vasculogenesis.32 More recent findings from mice with targeted mutations in Ang-2 showed that loss of Ang-2 results in profound defects in the development and function of the lymphatic vasculature, which can be completely rescued by replacing Ang-2 with Ang-1.5 These findings suggest that Ang signaling acts in proper remodeling and maturation of the lymphatics, as has already been observed for Angs in the blood vessel system. Furthermore, the expression of Tie2 in LECs indicates that Angs directly affect lymphatic vessel formation as they do blood vessel formation. Ang-1-induced angiogenic activity is regulated by Tie2-induced activation of the phosphatidylinositol-3-kinase (PI-3K)/Akt pathway,33 which results in activation of the focal adhesion kinase (FAK) and production of matrix metalloproteases (MMPs).34 We hypothesize that such signaling pathways could activate positive chemotactic and antiapoptotic signals in lymphatic vessels.
Recently, Carlson et al35 demonstrated that Ang-1 can serve as a substrate for cell adhesion by binding to integrin β1, even in the absence of Tie2 receptors. Moreover, Huang et al36 reported that integrin α9β1 is required for normal development of the lymphatic system by analyzing mice lacking integrin α9. We also showed that Ang-1 stimulates cell adhesion to fibronectin mediated by integrin β1.37 Based on these findings, we speculate that Ang signaling in lymphatics might be regulated by 2 signaling systems, Tie2 and integrins.
Ang-1/Ang-2 signaling through the Tie2 receptor and VEGF-A signaling through VEGFR-1/VEGFR-2 and the neuropilin-1 (Nrp-1) coreceptor work in a complementary fashion during development of the blood vasculature.2,32,38 In contrast to VEGF-A signaling in blood vessel formation, VEGF-C/VEGF-D signaling via VEGFR-3 plays a critical role in lymphangiogenesis.39,40 In this study, we found that COMP-Ang-1 increased the number of LYVE-1+ endothelial cell colonies in an in vitro coculture with OP9 feeder cells, suggesting that Ang-1 signaling might be involved in the growth of LECs. These findings prompted us to speculate that there is some collaboration between Angs and VEGF-C/VEGF-D signaling in lymphangiogenesis. Ang-1 alone does not affect the proliferation of BECs, whereas Ang-1 potentiates VEGF, which is a critical vascular regulator.41 Because OP9 cells are known to secrete several growth factors, including VEGF-C and VEGF-D,42 it is possible that our observed COMP-Ang-1-induced LEC proliferation in the OP9 coculture system could depend on VEGFR-3 signaling. To examine this possibility, we investigated whether recombinant soluble VEGFR-3-Fc fusion protein, which acts as an inhibitor by sequestering VEGF-C and VEGF-D, suppressed COMP-Ang-1-induced LEC proliferation. The enhanced effect on COMP-Ang-1-induced growth of LECs was partially suppressed by the addition of VEGFR-3-Fc protein. In contrast, recombinant soluble Tie2-Fc fusion protein, which acts as an inhibitor by sequestering Ang-1, completely suppressed the COMP-Ang-1-induced LEC growth. These findings suggest that both a direct effect of Ang-1 signaling on the growth of LECs and secondary activation of the VEGF-C or VEGF-D signaling pathway enhanced the growth of LECs seen following the addition of COMP-Ang-1. More recent findings suggesting that Ang-1-induced LEC proliferation occurs partially by increasing the expression of VEGFR-3 is particularly noteworthy in regard to cross-talk among growth factors in lymphangiogenesis.43 Based on our findings, although the molecular mechanisms by which Ang-1/Tie2 and VEGF-C/VEGF-D/VEGFR-3 induce lymphangiogenesis may be regulated by different sets of genes, the mechanisms may partially overlap in some settings. Taken together with recent findings derived from animal models that the gene transfer of VEGF-C resolves lymphedema through direct augmentation of lymphangiogenesis,44 we hypothesize that Ang-1 is also involved in transporting extravasated tissue fluid through its promotion of lymphangiogenesis. Mice defective in neuropilin-2 (Nrp-2), which is highly expressed in LECs, show defects in the development of lymphatic vessels.45 These findings indicate that Ang's, VEGFs, and Nrp's act together in the development and regulation of the lymphatic vasculature.
In a preliminary study, we found that a caspase inhibitor46 increased the colony formation of LYVE-1+ endothelial cells in vitro in a coculture with OP9 feeder cells. Because Ang-1 activates Akt kinase, which promotes antiapoptotic signaling, Ang-1-induced colony formation of LECs might be dependent on the Ang-1-induced antiapoptotic signaling.
In this study, we established mAbs against LYVE-1, a specific LEC marker. LYVE-1, like CD44, is a hyaluronic acid receptor.47 Although the function of LYVE-1 is unknown, it may provide good clues for clarifying the original function of LECs, such as cell movement and transcytosis of leukocytes and cancer cells in the interstitial tissues. LYVE-1 mAb enables us to determine whether Tie2 is expressed in lymphatics. Immunohistochemistry in adult mice revealed a low level of Tie2 expression in LECs in contrast to the high levels seen in BECs. Moreover, FACS analysis revealed that LYVE-1+ LECs could be grouped as Tie2 high- or low-positive cells. It would be interesting to subdivide endothelial cells according to the Tie2 expression level and determine whether the Ang-1 effect correlates with receptor expression. At present, we hypothesize that Tie2 expression levels in LECs are dependent on a particular biologic context or activity, such as sprouting, remodeling, and antiapoptosis. Conditional mutations in mice only in lymphatic vessels should reveal the precise role of Ang signaling through Tie2, given that mice lacking Ang-1 and Tie2 show defects in angiogenesis and early embryonic death prior to development of lymphatic vessels. Moreover, we observed that LYVE-1 is exclusively expressed in LECs, but only occasionally in sinusoidal endothelial cells. It has been reported that sinusoidal endothelial cells exhibit different molecular characteristics from blood endothelial cells in other tissues.24 For example, the common blood endothelial cell antigen CD34 is not expressed on sinusoidal cells in liver, spleen, and bone marrow,48 although the biologic significance of this observation is not known. We also showed that lymphatic endothelial cells do not express CD34. Therefore, it will be interesting to clarify the relationships between LECs and sinusoidal endothelial cells.
In summary, our results show that Ang-1 displays potent lymphangiogenic activity based on the following 3 observations: (1) the expression of Tie2 in LECs, (2) COMP-Ang-1 increases in vitro colony formation of LECs and it is completely suppressed by soluble Tie2, and (3) COMP-Ang-1 promotes in vivo lymphatic angiogenesis in mouse cornea and is inhibited by the addition of soluble Tie2. Ang-1 strongly suppresses blood vascular permeability and its related tissue edema,49,50 because it strongly decreases plasma leakage in blood vessels. Based on the findings of this study, we speculate an increase in lymphatic vessel networks also contributes to an Ang-1-induced suppressive effect on tissue edema because of lymphatic vessels drainage of leaked plasma components and excess protein-rich interstitial fluid from the extracellular spaces within the organs. Further clarification of the role of Ang-1 in lymphatic vessels may be important for the control of tissue edema.
Prepublished online as Blood First Edition Paper, February 10, 2005; DOI 10.1182/blood-2004-08-3382.
Supported by Grants-in-Aid for Scientific Research on Priority Areas and Grant-in-aid for COE Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan, by a Research Grant from the Human Frontiers Science Program Organization, and by a Keio University Grant-in-Aid for Encouragement of Young Medical Scientists.
The online version of the article contains a data supplement.
An Inside Blood analysis of this article appears in the front of this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Ms Kana Fukushima and Ayami Ono for their expert assistance in the experiments and Dr Fumio Arai for generous assistance with FACS analysis.