We have previously described a case of severe hypochromic microcytic anemia caused by a homozygous mutation in the divalent metal transporter 1 (DMT1 1285G > C). This mutation encodes for an amino acid substitution (E399D) and causes preferential skipping of exon 12 during processing of the DMT1 mRNA. To examine the functional consequences of this mutation, full-length DMT1 transcript with the patient's point mutation or a DMT1 transcript with exon 12 deleted was expressed in Chinese hamster ovary (CHO) cells. Our results demonstrate that the E399D substitution has no effect on protein expression and function. In contrast, deletion of exon 12 led to a decreased expression of the protein and disruption of its subcellular localization and iron uptake activity. We hypothesize that the residual protein in hematopoietic cells represents the functional E399D DMT1 variant, but because of its quantitative reduction, the iron uptake activity of DMT1 in the patient's erythroid cells is severely suppressed.
Introduction
Divalent metal transporter 1 (DMT1) protein (also called Nramp2, DCT1, and SLC11A2) plays a crucial role in intestinal iron (Fe2+) absorbtion1,2 and iron transport across the membrane of acidified endosomes.3 DMT1 is an integral membrane protein composed of 12 predicted transmembrane domains (TM) and containing 2 putative glycosylation sites in an extracytoplasmic loop, membrane targeting motifs, and a consensus transport motif.4,5 Recently, Touret et al6 demonstrated that the G185R DMT1 mutant protein found in microcytic anemia (mk) mice7 and the Belgrade (b) rats8 is abnormally processed, less stable, and displays decreased transport activity. We previously reported a Czech female with severe hypochromic microcytic anemia and iron overload caused by a homozygous mutation in the DMT1 gene (1285G > C) that changes Glu 399 to Asp (E399D).9,10 This single nucleotide substitution also causes preferential skipping of exon 12 during mRNA processing. As a consequence, there are 2 different DMT1 transcripts present in the patient's cells: a full-length transcript containing the point mutation and that missing exon 12; the later version comprising 90% of the total DMT1 mRNA. This shorter transcript is in low levels present also in normal control erythroid cells, but it was not detected in duodenum of control subjects.10 Here, we show that DMT1 protein levels in the patient's peripheral blood cells and in her burst-forming unit erythroid (BFU-E) colonies are markedly decreased. Additionally, we investigate the functional consequences of the patient's mutation by expressing different versions of DMT1 in cultured cells and thereby show that both the expression and function of DMT1 protein are disturbed by this mutation.
Study design
Processing of peripheral blood cells
Blood samples of the patient, her heterozygous parents, and healthy donors were obtained with informed consent. The ethics committee of the Palacky University Hospital approved the study. Peripheral blood was collected in heparinized tubes and processed for in vitro colony-forming assay9 and for Western blot analyses. Red blood cells were removed by treatment with NH4Cl. After 2 washes with phosphate-buffered saline (PBS), the protein extracts were prepared by resuspending the cells in 200 μL TNE buffer11 (100 mM NaCl; 10 mM Tris [tris(hydroxymethyl)aminomethane]-Cl, pH 7.0; 10 mM EDTA [ethylenediaminetetraacetic acid]) containing a cocktail of protease inhibitors (Sigma, Oakville, Canada) followed by centrifugation at 13 800g for 10 minutes. Protein concentration was determined by Bradford assay.
Cell culture and transfection
Chinese hamster ovary LR73 (CHO LR73) cells and the Caco-2 cells were grown as described.6,12 The human DMT1 cDNA (non-IRE isoform, generous gift from Dr J. T. Prchal, Baylor College of Medicine, Houston, TX) was tagged by C-terminal HA epitope and inserted into the expression plasmid pcDNA3.1(-) (Invitrogen, Burlington, Canada). Site-directed mutagenesis was performed using the QuikChange and ExSite kits (Stratagene, La Jolla, CA) and allele-specific oligonucleotides: 5′GGCCAGTTTGTCATGGACGGATTCCTGAACCTA3′, 5′TAGGTTCAGGAATCCGTCCATGACAAACTGGCC3′ to create E399D DMT1; 5′CCCTTTGTAGATGTCCACAGCCAGTGT3′,5′GGATTCCTGAACCTAAAGTGGTCACGC′3 to create DEL DMT1. Transfections of CHO cells with WT DMT1, E399D DMT1, DEL DMT1, and empty vectors were performed using lipofectamine reagent (Invitrogen). Clones of stable transfectants were selected as previously described.13
Immunoblot analysis
Total cell lysates from CHO-transfected cells were prepared as described for peripheral blood cells (see “Processing of peripheral blood cells”). Crude membrane fractions were isolated as previously described.11 Protein lysates were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) (10%) and transferred to nitrocellulose membranes (Bio-Rad, Hercules, CA). Immunoblots were incubated with primary antibodies: goat anti-Nramp2 antibody (N-20, Santa Cruz Biotechnology, Santa Cruz, CA) or mouse anti-HA antibody (F-7; Santa Cruz Biotechnology). The immune complexes were visualized using Super Signal West Dura Chemiluminescent Substrate (Pierce, Rockford, IL).
Immunofluorescence
Harvested BFU-Es were cytospun, and CHO-transfected cells were seeded in 60-mm plates containing coverslips. The cells were fixed and permeabilized using ice-cold methanol-acetone (1:1) solution. The protein expression was analyzed using goat anti-Nramp2 (N-20) or mouse anti-HA (F-7) antibody. For the subcellular localization experiments, these primary antibodies were used: rabbit anti-early endosomal antigen 1 antibody (EEA1; Alexis, Montreal, Canada) and rabbit anti-calnexin antibody (Stressgen, Victoria, Canada). The slides were analyzed by immunofluorescence or confocal microscopy.
59Fe uptake by CHO cells
The reduction of 59Fe(III) to 59Fe(II) was accomplished by addition of 56FeSO4 (10-fold excess) to 59FeCl3 in 0.1 M HCl. Thirty-six hours after transfection cells were washed twice with PBS prewarmed to 37°C and incubated in 2 mL of prewarmed incubation buffer (25 mM Tris, 25 mM 4-morpholineethanesulfonic acid [MES], 140 mM NaCl, 5.4 mM KCl, 5 mM glucose, 1.8 mM CaCl2, pH 6.0) containing 10 μM 59Fe(II) and 0.44 mM sodium ascorbate at 37°C for 5, 20, and 60 minutes. The 59Fe uptake was terminated by 3 washes with ice-cold PBS. Cells were detached and membrane-associated 59Fe was removed using 30 minutes' incubation (4°C) in 2 mL PBS containing 1 mg/mL pronase plus 5 mM EDTA.14 After 2 additional washes with PBS, the 59Fe radioactivities were measured in a gamma counter (Packard Cobra II auto-gamma; Perkin-Elmer, Wellesley, MA).
Results and discussion
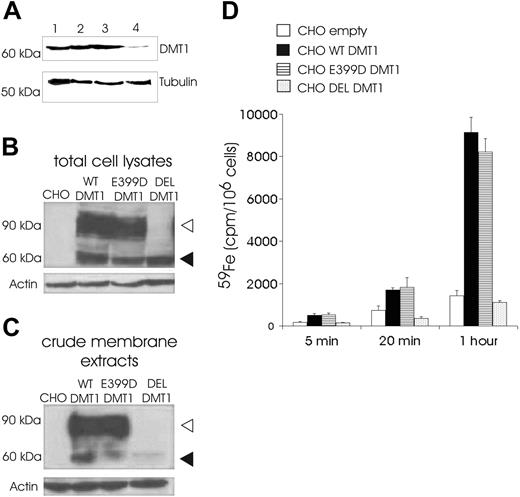
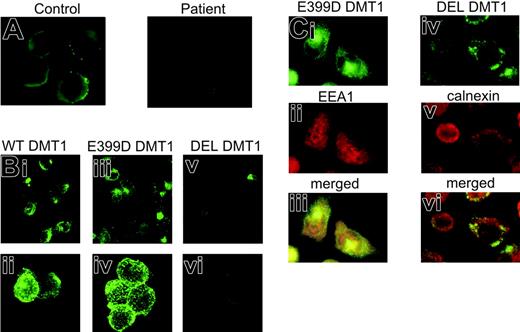
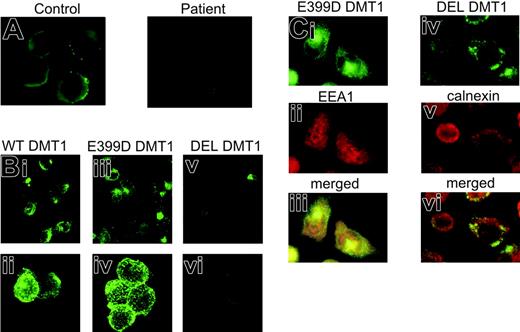
The effect of the DMT1 1285G > C mutation on the level of protein expression was initially studied in peripheral blood cells. Immunoblotting with an anti-Nramp2 antibody revealed a 64-kDa protein band (Figure 1A), which corresponded to a band seen in the protein extract of Caco-2 cells. The level of DMT1 expression in the patient's cells was clearly decreased compared to her heterozygous parents. Subsequent immunofluorescence analysis on cytospun BFU-Es revealed decreased abundance of DMT1 on the surface of the patient's erythroblasts compared to normal control BFU-E-derived erythroblasts (Figure 2A). These findings suggest that the mechanism of the patient's hypochromic microcytic anemia involves quantitative DMT1 protein reduction.
To evaluate the consequences of the patient's mutation on the functional properties of the DMT1 protein, CHO cells were transiently transfected with HA-tagged expression constructs encoding WT DMT1, E399D DMT1, or DEL DMT1. Western blotting with mouse anti-HA antibody revealed 2 bands in CHO cells expressing WT or E399D DMT1 (Figure 1B), representing the mature complex-glycosylated form (90 to 100 kDa) and the core-glycosylated form (66 kDa) of DMT1 as it has previously been shown for HA-tagged mouse Nramp2.13 In CHO cells expressing the DEL DMT1, only the core-glycosylated form of the protein was detectable (Figure 1B). The absence of complex glycosylation of this protein product is not unexpected since deletion of exon 12 removes TM domain 8; the 2 putative glycosylation sites in DMT1 are present in an extracytoplasmic loop between TM7 and TM8.4 To assess the possibility that the mutation can interfere with the membrane targeting of the protein, crude membrane extracts from CHO-transfected cells were prepared. Subsequent immunoblot analysis revealed the same 2 DMT1 species as detected in total cell lysates (Figure 1C). These results demonstrate that the E399D substitution has no effect on protein expression, its glycosylation, or membrane targeting. A very weak band of core-glycosylated protein also was present in the membrane extract from CHO cells transfected with DEL DMT1. This could either indicate that a small fraction of the protein is targeted to the membrane, or it may represent a fraction of the immature protein present in the endoplasmic reticulum (ER).15 The cellular distribution of the proteins revealed the same pattern of DMT1 staining for WT and E399D DMT1 transfectants (Figure 2Bi-iv), suggesting that the E399D amino acid substitution has no effect on the protein cellular targeting. On the other hand, only a faint signal was observed in CHO cells transfected with DEL DMT1 (Figure 2Bv-vi). These expression patterns were not unique to CHO cells, as the same results were obtained using H1299 cells expressing WT, E399D, and DEL DMT1 (data not shown).
Expression of DMT1 and iron uptake study. (A) Western blot analysis of peripheral blood cell lysates (90 μg) and Caco-2 cells lysate (30 μg) with primary goat anti-Nramp2 antibody (1/1000, 16 hours, 4°C) and peroxidase-conjugated secondary antibody (Pierce; 1/1000, 90 minutes, room temperature). The 64-kDa band represents the DMT1 protein; lane 1, Caco-2; lane 2, patient's father; lane 3, patient's mother; lane 4, patient. (B-C) Western blot analysis of total cell lysates (30 μg) and crude membrane extracts (50 μg) from CHO cells expressing the empty vector (CHO), WT, E399D, and DEL forms of HA-tagged DMT1 with mouse anti-HA antibody (1/1000, 1 hour, room temperature) and peroxidase-conjugated secondary antibody (1/1000, 90 minutes, room temperature). Mature complex-glycosylated DMT1 form (90 to 100 kDa; ▹) and the core-glycosylated form of DMT1 (66 kDa; ◂) are indicated. Equal loading of proteins was assessed by probing with an antibody against β-actin or α-tubulin (1/1000, 1 hour, room temperature). Representative immunoblots of 3 separate experiments are illustrated. (D) Iron transport activities of WT, E399D, and DEL DMT1 incubated in pH 6.0 incubation buffer with 59Fe(II)-ascorbate (10 μM 59Fe). Iron uptake is expressed as intracellular 59Fe (cpm per 106 cells). “CHO empty” represents iron uptake by cells transfected with empty vector. Data shown are the means ± SD of duplicate determinations from a typical experiment that was performed 3 times.
Expression of DMT1 and iron uptake study. (A) Western blot analysis of peripheral blood cell lysates (90 μg) and Caco-2 cells lysate (30 μg) with primary goat anti-Nramp2 antibody (1/1000, 16 hours, 4°C) and peroxidase-conjugated secondary antibody (Pierce; 1/1000, 90 minutes, room temperature). The 64-kDa band represents the DMT1 protein; lane 1, Caco-2; lane 2, patient's father; lane 3, patient's mother; lane 4, patient. (B-C) Western blot analysis of total cell lysates (30 μg) and crude membrane extracts (50 μg) from CHO cells expressing the empty vector (CHO), WT, E399D, and DEL forms of HA-tagged DMT1 with mouse anti-HA antibody (1/1000, 1 hour, room temperature) and peroxidase-conjugated secondary antibody (1/1000, 90 minutes, room temperature). Mature complex-glycosylated DMT1 form (90 to 100 kDa; ▹) and the core-glycosylated form of DMT1 (66 kDa; ◂) are indicated. Equal loading of proteins was assessed by probing with an antibody against β-actin or α-tubulin (1/1000, 1 hour, room temperature). Representative immunoblots of 3 separate experiments are illustrated. (D) Iron transport activities of WT, E399D, and DEL DMT1 incubated in pH 6.0 incubation buffer with 59Fe(II)-ascorbate (10 μM 59Fe). Iron uptake is expressed as intracellular 59Fe (cpm per 106 cells). “CHO empty” represents iron uptake by cells transfected with empty vector. Data shown are the means ± SD of duplicate determinations from a typical experiment that was performed 3 times.
Immunofluorescence analysis of DMT1 in BFU-E-derived erythroblasts and transfected CHO cells. (A) Day 14 healthy control and the patient's BFU-Es were harvested, cytospun, and stained using goat anti-Nramp2 antibody (1/50, 3 hours, room temperature) and fluorescein isothiocyanate (FITC)-conjugated secondary antibody (Molecular Probes, Eugene, OR; 1/1000, 90 minutes, room temperature). (B) Immunostaining of WT (i-ii), E399D (iii-iv) and DEL (v-vi) DMT1 in transiently transfected CHO cells with mouse anti-HA antibody (1/200, 1 hour, room temperature) and FITC-conjugated secondary antibody (1/1000, 1 hour, room temperature). (C) Subcellular localization of E399D DMT1 in early endosomes (i-iii) and DEL DMT1 in the endoplasmic reticulum (iv-vi) in stably transfected CHO cells. Cells immunostained with mouse anti-HA antibody and with FITC-conjugated secondary antibody were subsequently stained either with anti-EEA1 antibody (1/200, 1.5 hours, room temperature) or anti-calnexin antibody (1/200, 1.5 hours, room temperature) followed by incubation with red-fluorescent-conjugated secondary antibody (Alexa Fluor 594, Molecular Probes; 1/1000, 1.5 hours, room temperature). In panels A and C the cells were visualized on an Olympus BX 50 fluorescence microscope (Olympus, Hamburg, Germany) using a 100 ×/1.3 numeric aperture (NA) oil immersion objective. Digital images were acquired with an Olympus DP 50 camera driven by the software Viewfinder Lite version 1.0.135 (Pixera, Los Gatos, CA). Original magnifications, × 1000. In panel B the cells were examined on a Zeiss Pascal 5 confocal microscope (Carl Zeiss, Jena, Germany) using a 40 ×/0.75 NA objective (i, iii, v) and a 63 ×/1.4 NA oil immersion objective (ii, iv, vi). The Zeiss LSM Browser version 3.2.0 115 was used for handling pictures. Images were cropped, assembled, and labeled using Adobe Photoshop software (Adobe Systems, San Jose, CA).
Immunofluorescence analysis of DMT1 in BFU-E-derived erythroblasts and transfected CHO cells. (A) Day 14 healthy control and the patient's BFU-Es were harvested, cytospun, and stained using goat anti-Nramp2 antibody (1/50, 3 hours, room temperature) and fluorescein isothiocyanate (FITC)-conjugated secondary antibody (Molecular Probes, Eugene, OR; 1/1000, 90 minutes, room temperature). (B) Immunostaining of WT (i-ii), E399D (iii-iv) and DEL (v-vi) DMT1 in transiently transfected CHO cells with mouse anti-HA antibody (1/200, 1 hour, room temperature) and FITC-conjugated secondary antibody (1/1000, 1 hour, room temperature). (C) Subcellular localization of E399D DMT1 in early endosomes (i-iii) and DEL DMT1 in the endoplasmic reticulum (iv-vi) in stably transfected CHO cells. Cells immunostained with mouse anti-HA antibody and with FITC-conjugated secondary antibody were subsequently stained either with anti-EEA1 antibody (1/200, 1.5 hours, room temperature) or anti-calnexin antibody (1/200, 1.5 hours, room temperature) followed by incubation with red-fluorescent-conjugated secondary antibody (Alexa Fluor 594, Molecular Probes; 1/1000, 1.5 hours, room temperature). In panels A and C the cells were visualized on an Olympus BX 50 fluorescence microscope (Olympus, Hamburg, Germany) using a 100 ×/1.3 numeric aperture (NA) oil immersion objective. Digital images were acquired with an Olympus DP 50 camera driven by the software Viewfinder Lite version 1.0.135 (Pixera, Los Gatos, CA). Original magnifications, × 1000. In panel B the cells were examined on a Zeiss Pascal 5 confocal microscope (Carl Zeiss, Jena, Germany) using a 40 ×/0.75 NA objective (i, iii, v) and a 63 ×/1.4 NA oil immersion objective (ii, iv, vi). The Zeiss LSM Browser version 3.2.0 115 was used for handling pictures. Images were cropped, assembled, and labeled using Adobe Photoshop software (Adobe Systems, San Jose, CA).
To evaluate the iron uptake properties of the E399D and DEL DMT1 proteins, CHO transfectants were exposed to 59Fe(II) and the internalized radioactivity evaluated. The capacity of WT DMT1 and E399D DMT1 to transport iron was comparable (Figure 1D). In contrast, DEL DMT1-transfected CHO cells exhibited no increase in iron uptake, suggesting that deletion of exon 12 abolishes the iron transport function of DMT1.
The absence of the complex-glycosylated form of DEL DMT1 together with the immunofluorescence analysis and the iron uptake experiments in the transiently transfected cells suggested that the cellular trafficking of DEL DMT1 could be erroneous. To determine the subcellular protein localization and to overcome the DEL DMT1 borderline level of expression, stable transfectants expressing WT, E399D, and DEL DMT1 were selected and subjected to immunofluorescence analyses. The observed punctate intracellular staining for WT (not shown) and E399D DMT1 is consistent with the localization of these proteins in early endosomes, which was confirmed using an antibody against EEA1 (Figure 2Ci-iii). In contrast, the DEL DMT1 subcellular localization exhibited overlapping pattern with calnexin, an ER marker (Figure 2Civ-vi). These data show that disrupted complex glycosylation of DEL DMT1 results in retention of the immature form of this protein in the ER. The observed quantitative DMT1 protein reduction in the patient's bone marrow erythroblasts9 and in the patient's BFU-Es indicates that the stability of DEL DMT1 protein might be reduced. As only 10% of the total DMT1 transcript in the patient's erythroid cells encodes for a fully functional E399D protein, it is likely that the erythroid iron use is severely suppressed. The definite cause of the patient's concomitant liver iron overload remains elusive. Our group has previously shown that the total mRNA level of DMT1 in the patient's duodenum are probably not affected and that DMT1 protein expression might even be increased.10 However, it is difficult to make definitive conclusions as both intra-individual and inter-individual differences in the level of DMT1 expression in the duodenum, reflecting the body iron stores and recent iron content in the diet, may exist. Nevertheless, we believe that the liver hemosiderosis evolved as a result of the alteration of a few processes, including activation of heme absorption9 and increased activity of the basolateral iron transport.10
Prepublished online as Blood First Edition Paper, August 9, 2005; DOI 10.1182/blood-2005-04-1550.
Supported by the Canadian Institutes for Health Research (M.P., G.N., A.D.S., and P.P.) and by the Czech Republic Ministry of Health grant NR/7799-3 and Ministry of Education grant MSM 6198959205 (M.P., D.P., and V.D.).
M.P. conducted and designed the experiments and wrote the report; G.N. initiated and designed a part of the experiments; A.D.S. helped with confocal microscopy and the iron uptake study and contributed to the editing of the report; D.P. was responsible for the treatment of the patient and collection of the samples and contributed to the editing of the report; V.D. conceived the study, contributed to ideas in this report, and contributed to the writing of the report; P.P. conceived and designed the study and contributed to the writing of the report.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.