Abstract
The function of α4β1 and α4β7 integrins in hematopoiesis is controversial. While some experimental evidence suggests a crucial role for these integrins in retention and expansion of progenitor cells and lymphopoiesis, others report a less important role in hematopoiesis. Using mice with a deletion of the β1 and the β7 integrin genes restricted to the hematopoietic system we show here that α4β1 and α4β7 integrins are not essential for differentiation of lymphocytes or myelocytes. However, β1β7 mutant mice displayed a transient increase of colony-forming unit (CFU-C) progenitors in the bone marrow and, after phenylhydrazine-induced anemia, a decreased number of splenic erythroid colony-forming units in culture (CFUe's). Array gene expression analysis of CD4+CD8+ double-positive (DP) and CD4–CD8– double-negative (DN) thymocytes and CD19+ and CD4+ splenocytes did not provide any evidence for a compensatory mechanism explaining the mild phenotype. These data show that α4β1 and α4β7 are not required for blood cell differentiation, although in their absence alterations in numbers and distribution of progenitor cells were observed.
Introduction
The development and maintenance of hematopoietic stem cells (HSCs), which can self-renew and differentiate into all hematopoietic blood cell lineages, is thought to depend on their interactions with the microenvironment. Integrins expressed on HSCs are capable of mediating several of those interactions as they can bind extracellular matrix components such as fibronectin and laminin, but also cellular receptors like VCAM-1 expressed on bone marrow (BM) stroma cells. Integrins are a family of heterodimeric cell-surface receptors consisting of an α and a β subunit.1 Integrins provide mechanical support by connecting the extracellular matrix (ECM) with the cytoskeleton, but are also capable of transducing chemical signals upon ligand binding. This signaling results in cytoskeleton reorganization and changes in gene expression affecting proliferation, differentiation, and survival of cells.2 Molecules inside the cell, on the other hand, can modulate the affinity and avidity of integrins, called inside-out signaling, which is, for example, crucial for the extravasation of leukocytes.3
In vitro and in vivo experiments suggests an important role of α4β1 and α5β1 integrins for the adhesion of HSCs and hematopoietic progenitor cells (HPCs) to fibronectin in the bone marrow matrix.4,5 Integrin α4β1 was additionally shown to mediate binding to VCAM-1, which is expressed on BM stroma cells.6 Injection of fibronectin fragments and blocking antibodies against α4β1 and VCAM-1 led to a release of HSCs/HPCs into the blood, supporting the proposed importance of these interactions in vivo.5,7 Conditional deletion of the VCAM-1 gene resulted in an early exit of B-cell precursors into the blood.8 Finally, it was shown that α4β1–mediated attachment of HPCs to fibronectin promotes proliferation and survival,9,10 suggesting a crucial role for self-renewal and survival of HSCs.
In vivo studies with β1 integrin–deficient somatic chimeric mice, which are generated by injecting β1-null embryonic stem (ES) cells into wild-type host blastocysts, demonstrated that β1 integrin is not required for the formation of HSCs, but is essential for their migration to the fetal liver.11 Additionally, β1 integrin–deficient HSCs failed to engraft lethally irradiated mice.12 Altogether, these data pointed to a key role of α4β1 integrin in hematopoiesis. This notion was corroborated by the analysis of α4-null somatic chimeric mice, which have almost no mature B cells, T cells, or erythroblasts derived from α4-null ES cells.13,14 In vitro experiments with cells derived from the α4-null chimeric mice suggested that both erythroid and B-cell precursors are less able to transmigrate through the stroma, which may result in reduced cell proliferation.14 Also, the number of α4-deficient myeloid cells was reduced compared with control chimera. Since β7 integrin constitutive null mice displayed normal hematopoiesis,15 it was suggested that α4β1 integrin might be the pivotal integrin during hematopoiesis, as α4 can dimerize only with β1 and β7 integrins. Therefore, it was unexpected when β1 mutant BM chimeras showed no defects in blood cell development.16 The simplest explanation at that time was that α4β1 and α4β7 integrin might have redundant functions in blood cell development and that only the absence of both receptors leads to the described hematopoietic defects. However, further experiments showed that inducible deletion of the α4 integrin gene has only subtle effects on hematopoiesis.17 These mutants showed only a partial reduction of the B220+ B-cell and CD4+ T-cell populations in BM. Monocytes (Mac-1+) and erythroblasts (Ter119+) were reported to occur in normal amounts in the BM. In this study, however, the α4 integrin gene was not only deleted in hematopoietic cells but also in many nonhematopoietic cells such as hepatocytes, endothelial cells, and so on, which could contribute to the phenotype. An alternative explanation for these contrasting results could be that fetal hematopoiesis is more dependent on α4 integrin than adult hematopoiesis.
To better understand the role of α4β1 and α4β7 integrin in adult hematopoiesis, we generated and analyzed mice with a blood cell–restricted knockout of β1 and a constitutive knockout of β7 integrin. As a consequence α4β7, α4β1 and also other β1 integrins expressed on blood cells are lost. In contrast to the α4-null somatic chimeras13,14 or the α4 conditional knockout mice17 used previously, we can exclude any effects due to deletion of α4 on nonhematopoietic cells, which might influence hematopoiesis through altered production of cytokines and growths factors or different cell-cell interactions. This model was used to study HSC maintenance, HPC distribution and differentiation, and the migration of differentiated cells in the absence of β1 and β7 integrins in adult mice. We demonstrate now that even in the absence of both α4β1 and α4β7 integrins, hematopoiesis is normal.
Materials and methods
Generation of mice with a deletion of the β1 and the β7 integrin genes in the hematopoietic system
Mice carrying a β1 integrin gene flanked by loxP sites (fl/fl)16 were mated with mice with a neomycin-disrupted β1 integrin gene (+/–),18 mice lacking a functional β7 integrin gene (β7–/–),13 and with mice carrying the Mx transgene (+Mx).19 β1β7 mutant BM chimeras were generated by BM transplantation as described previously.20 Recipient Ly-5.1+ mice were lethally irradiated and received BM cells from Ly-5.2+ β1fl/+β7–/–+Mx or β1fl/flβ7–/– mice (β7 mutant BM chimeras) or from β1fl/flβ7–/–+Mx mice (β1β7 mutant BM chimeras). Four weeks after the transfer, deletion of the β1 gene was induced by polyIC injections as described previously.20
Animal treatment
Mice were maintained and bred under pathogen-free conditions. All animal experiments were approved by the local ethics committee. Blood samples were obtained from the retro-orbital plexus under anesthesia. Acute hemolysis was assessed after phenylhydrazine (PHZ; Sigma, Steinheim, Germany) treatment as described.16
Türk staining
Whole blood of control and β1β7 mutant BM chimeras was isolated, diluted 1:10 with Türk stain (0.01% gentian violet, 1.0% acetic acid), and differentially counted for polymorphonuclear and mononuclear cells in a hemocytometer.
Flow cytometry
Single-cell suspensions were prepared and analyzed as described.16 Erythrocytes in blood samples were lysed by incubation in ACK-lysis buffer for 5 minutes at room temperature prior to staining.21
Deletion of the β1 integrin gene on BM stroma cells was assessed by measuring the activity of the β-galactosidase reporter.16 Five days after a single injection of 250 μg polyIC, BM cells were plated on tissue-culture plates as described.22 After 24 hours, nonadherent cells were removed and adherent cells detached by trypsin/EDTA. Nonhematopoietic BM stroma cells were characterized as Ly-5.2–Ter119–adherent cells, which consist of mesenchymal stem cells, fibroblasts, endothelial progenitor cells, and endothelial cells.23 Hematopoietic cells, on the other hand, were identified as Ly-5.2+ or Ter119+ nonadherent cells. Cells were stained for β-galactosidase activity as described,24 with minor changes. Briefly, 4 × 106 cells were suspended in 20 μL phosphate-buffered saline (PBS) added to 20 μL of 2 mM fluorescein-di-(beta-D-galactopyranoside) (FDG; Sigma). Cells were incubated at 37°C for 75 seconds and subsequently 200 μL ice-cold PBS was added. Cells were incubated for 3 hours on ice and analyzed by flow cytometry as described.16
For the analysis of platelets, 5 μL antibody solution containing FITC-conjugated anti–β1 integrin (Ha2/5; 1:10 diluted; BD Pharmingen, San Diego, CA) and PE-conjugated anti–GPIb-IX (p0p1) (kindly donated by Dr B. Nieswandt, University of Würzburg, Germany; 1:10 diluted) was added to 1 μL whole blood. After a 15-minute incubation at room temperature in the dark, 100 μL PBS was added and samples were analyzed by fluorescence-activated cell sorting (FACS).
Colony formation assay
Pre-B and CFU-C colony formation assays were performed as described previously.16 CFUe assays were carried out following the instructions of the manufacturer (Stem Cell Technologies, Vancouver, BC, Canada).
Separation of splenocytes by MACS
Leukocyte subpopulations were isolated from single-cell suspensions of splenocytes by positive selection using FITC-conjugated antibodies against B220 (B cells), CD4 (CD4 T cells), or CD8 (CD8 T cells) and anti-FITC MACS beads according to the manufacturer's instructions (Miltenyi Biotec, Bergisch Gladbach, Germany). The purity of the sort was checked by FACS analysis.
Southern blot analysis
Southern blot analysis was carried out as described.16 Membranes were exposed to x-ray films and the resulting bands quantified using Bio-PROFIL Bio-1D V97.03 software (Vilber Lourmat, Marne-la-Vallée, France).
DNA microarray hybridization and analysis
Total RNA was isolated from FACS-sorted populations of thymocytes (DN, CD4–CD8–; DP, CD4+, CD8+) and splenocytes (CD19+ B cells; CD4+ T cells). For biotin-labeled target synthesis reactions were performed using standard protocols supplied by the manufacturer (Affymetrix, Santa Clara, CA). Briefly, 5 μg total RNA was converted to double stranded (ds) DNA using 100 pmol of a T7T23V primer (Eurogentec, Seraing, Belgium) containing a T7 promotor. The cDNA was then used directly in an in vitro transcription reaction in the presence of biotinylated nucleotides.
The concentration of biotin-labeled cRNA was determined by UV absorbance. In all cases, 12.5 μg of each biotinylated cRNA preparation was fragmented and placed in a hybridization cocktail containing 4 biotinylated hybridization controls (BioB, BioC, BioD, and Cre) as recommended by the manufacturer. Samples were hybridized to an identical lot of Affymetrix MOE430A for 16 hours. After hybridization, the GeneChips were washed, stained with SA-PE, and read using an Affymetrix GeneChip fluidic station and scanner. Gene expression levels were determined by means of Affymetrix's Microarray Suite 5.0 (MAS 5.0).
Results
β1 and β7 integrins are coexpressed in many hematopoietic cells, including HSCs
In order to replace each other functionally, α4β1 and α4β7 must be expressed in the same cells. While β1 integrin is expressed on all hematopoietic cells besides erythrocytes,12,16 the expression of β7 integrin is more restricted (Figure S1, available on the Blood website; see the Supplemental Figure link at the top of the online article). In BM, β7 integrin was found on lin–c-kit+Sca1 high cells (ie, bona fide stem cells), most mature B cells (B220 high), on subpopulations of mature and immature granulocytes (Gr-1 high; Gr-1 medium), and on few erythroid cells (Ter119+) and immature B cells (B220 low). β7 integrin was furthermore found on subsets of DN, CD4SP, and CD8SP thymocytes, whereas it was virtually absent on DP thymocytes. In spleen and lymph nodes, β7 integrin was present on most B cells (B220+), T cells (CD4+, CD8+), and granulocytes (Gr-1+). In lymph nodes, about 50% of the erythroid cells (Ter119+) expressed β7 integrin, whereas only a few percent of the erythroid cells in the spleen had β7 on their surface.
Normal maintenance of HSCs in the combined absence of β1 and β7 integrins
To directly assess possible redundant functions of α4β1 and α4β7, mice were generated lacking both receptors in the hematopoietic system. Mice carrying a conditional knockout for β1 integrin, a β1-null allele, and a cre recombinase transgene under the control of the polyIC-inducible Mx-promotor were intercrossed with mice lacking a functional β7 integrin gene.15 Thus, mice that were deficient for β7 integrin and carried an inducible β1-null gene (β1fl/–β7–/–Mx-cre+ or β1fl/flβ7–/–Mx-cre+) were obtained. Mice lacking β7, but constitutively expressing β1 (β1fl/+β7–/–Mx-cre+ or β1fl/flβ7–/–) were used as controls. To restrict the deletion to the hematopoietic system, BM from these mice was transplanted into lethally irradiated recipient mice (Figure 1A). The ablation of the conditional β1 gene was induced by 3 intraperitoneal injections of polyIC after reconstitution of the hematopoietic system (4 weeks after irradiation). Mice were analyzed 2, 6, and 10 to 12 months after the polyIC treatment.
Efficient deletion of β1 integrin in the hematopoietic system does not affect cellularity of lymphoid organs. (A) Bone marrow of mice with a conditional knockout of β1 integrin, an Mx-cre transgene, and a constitutive knockout of β7 integrin (β1fl/–β7–/–Mx cre) was transplanted into lethally irradiated wild-type host mice. In bone marrow chimeras, donor and host cells could be distinguished by expression of Ly-5.1/Ly-5.2 surface marker. After repopulation of the hematopoietic system (4 weeks), polyIC was injected to induce expression of the cre recombinase in the donor cells and deletion of the β1 integrin gene. (B) Percentages of β1 integrin–negative platelets isolated from control and β1β7 mutant BM chimeric mice are shown at indicated time points after the first of 3 polyIC injections (day 0). Error bars show the standard deviation (n [control BM chimera]/[β1β7 mutant BM chimera]: 9/6). (C) DNA was isolated from single-cell suspensions from BM, thymus, and spleen from β1β7 mutant BM chimeras 10 months after polyIC treatment. Southern blot analysis detecting the conditional and the null allele was performed (a representative result is shown on the right panel). Band intensities were quantified and visualized in a bar graph. Error bars show the standard deviation. (n [control BM chimera]/[β1β7 mutant BM chimera]: 3/3). (D) Left panel: Single-cell suspensions were made from BM (2 femurs), thymus, spleen, and lymph nodes (2 inguinal, 2 axial, 1 para-aortic) of control and β1β7 mutant BM chimeric mice 2 months after the gene deletion. Cells were counted using a hemacytometer. The bar graph shows the absolute cell number in the respective tissues. Error bars show the standard deviation (n [control BM chimera]/[β1β7 mutant BM chimera]: 4/4). Right panel: Whole blood was collected retro-orbitally from control and β1β7 mutant BM chimeric mice 6 months after the gene deletion. The blood was diluted 1:10 with Türk stain and differentially counted in a hemacytometer. The bar graph shows the concentration of mononuclear (M) and polymorphonuclear (PMN) cells in the blood. Error bars show the standard deviation (n [control BM chimera]/[β1β7 mutant BM chimera]: 4/4).
Efficient deletion of β1 integrin in the hematopoietic system does not affect cellularity of lymphoid organs. (A) Bone marrow of mice with a conditional knockout of β1 integrin, an Mx-cre transgene, and a constitutive knockout of β7 integrin (β1fl/–β7–/–Mx cre) was transplanted into lethally irradiated wild-type host mice. In bone marrow chimeras, donor and host cells could be distinguished by expression of Ly-5.1/Ly-5.2 surface marker. After repopulation of the hematopoietic system (4 weeks), polyIC was injected to induce expression of the cre recombinase in the donor cells and deletion of the β1 integrin gene. (B) Percentages of β1 integrin–negative platelets isolated from control and β1β7 mutant BM chimeric mice are shown at indicated time points after the first of 3 polyIC injections (day 0). Error bars show the standard deviation (n [control BM chimera]/[β1β7 mutant BM chimera]: 9/6). (C) DNA was isolated from single-cell suspensions from BM, thymus, and spleen from β1β7 mutant BM chimeras 10 months after polyIC treatment. Southern blot analysis detecting the conditional and the null allele was performed (a representative result is shown on the right panel). Band intensities were quantified and visualized in a bar graph. Error bars show the standard deviation. (n [control BM chimera]/[β1β7 mutant BM chimera]: 3/3). (D) Left panel: Single-cell suspensions were made from BM (2 femurs), thymus, spleen, and lymph nodes (2 inguinal, 2 axial, 1 para-aortic) of control and β1β7 mutant BM chimeric mice 2 months after the gene deletion. Cells were counted using a hemacytometer. The bar graph shows the absolute cell number in the respective tissues. Error bars show the standard deviation (n [control BM chimera]/[β1β7 mutant BM chimera]: 4/4). Right panel: Whole blood was collected retro-orbitally from control and β1β7 mutant BM chimeric mice 6 months after the gene deletion. The blood was diluted 1:10 with Türk stain and differentially counted in a hemacytometer. The bar graph shows the concentration of mononuclear (M) and polymorphonuclear (PMN) cells in the blood. Error bars show the standard deviation (n [control BM chimera]/[β1β7 mutant BM chimera]: 4/4).
At all time points analyzed, no expression of β7 integrin was detectable in any tissue by FACS (data not shown). To analyze the time course of the β1 integrin gene ablation, we monitored the loss of β1 integrin expression on short-lived platelets. Two days after the first polyIC injection, β1-deficient platelets were already detectable in the blood of β1β7 mutant BM chimeras (Figure 1B). The relative amount of β1-deficient platelets increased continuously to reach 93% after 14 days and 97% after 21 days and later. In control BM chimeras, on the other hand, virtually all platelets expressed β1 integrin at all time points analyzed. These data show that the deletion of the β1 integrin gene can be induced within a few days in a β7 mutant background. Furthermore, they confirm that the development of megakaryocytes and platelets is not crucially dependent on β1 and β7 integrins. Southern blot analysis of BM, spleen, and thymus of 2- and 10-month-old β1β7 mutant and control BM chimeras confirmed the efficient β1 gene deletion in all these tissues (Figure 1C and data not shown). Since only HSCs can sustain hematopoiesis for more than 3 months, these data indicate that β1β7-deficient HSCs are maintained in vivo.
To investigate the development of different hematopoietic lineages that derive from HSCs, we first checked the cellularity of different lymphoid organs. At 2 months (Figure 1D, left) and 10 to 12 months (data not shown) after induction of the gene deletion there were no differences observed in the cellularity of BM, thymus, or spleen of control and β1β7 mutant BM chimeras, providing no evidence for defective hematopoiesis in the absence of β1 and β7 integrins. Differential blood counts revealed similar numbers of mononuclear and polymorphonuclear cells in the peripheral blood (PB) of control and β1β7 mutant BM chimeras 6 months after polyIC treatment (Figure 1D, right).
Normal B-cell development in the absence of β1 and β7 integrins
Since previous studies suggested that normal B-cell development was dependent on α4 integrin,13,14,17 but not on α4β116 or α4β715 alone, we investigated whether β1 and β7 integrins have a redundant function in B-cell development. In pre-B colony assays, control and β1β7 mutant BM gave rise to colonies that were derived each from a single pre-B-cell pecursor. FACS analysis of randomly picked colonies confirmed that 36 of 39 colonies (92.3%) of β1β7 mutant BM did not express β1 integrins, whereas all tested colonies derived from control BM expressed β1 integrins. No host-derived colonies expressing Ly-5.1 were detected. To further monitor B-cell development, single-cell suspensions from BM, spleen, and lymph nodes (LNs) were analyzed using B-cell–specific markers: B220 (pre-proB and later), CD19 (proB and later), IgM (immature B), and IgD (all mature B). The relative amount of cells positive for the respective markers was unaltered in β1β7 mutant BM chimeras compared with control BM chimeras 2 and 12 months after the knockout induction (Table 1 and data not shown).
FACS analysis of immature B cells (B220 medium) proved the loss of β1 integrin (Figure 2A). Mature B cells (B220 high) express only low amounts of β1 integrin, which makes it difficult to distinguish normal from β1-deficient mature B cells by FACS (Figure 2A). Therefore, the knockout efficiency in B220+ B cells purified from spleen was determined by Southern blot analysis (Figure 2B). B220+ B cells were enriched by MACS beads to a purity of more than 95% (Figure 2B, left). Southern blot of genomic DNA isolated from these cells revealed a deletion efficiency of the β1 integrin gene of 93.5% ± 8.3% (n = 5). These data indicate that in the absence of β1 and β7 integrins, B cells can fully mature. Furthermore, since spleen, LN, and BM contained normal numbers of B cells, migration of immature B cells to spleen and of mature B cells to LN and BM is apparently not impaired by the combined loss of β1 and β7 integrins.
Normal B-cell population sizes in the absence of β1 and β7 integrins. (A) Representative histogram overlays show the β1 integrin expression on immature (B220med) and mature (B220hi) B cells of BM and spleen, respectively, of control (filled) and β1β7 mutant BM chimeras (line). (B) Single-cell suspensions from spleen of control and β1β7 mutant BM chimeras 6 months after polyIC treatment were prepared, stained with B220-FITC antibody, and subsequently sorted using anti-FITC MACS beads. Left panel: FACS analysis of the B220+-enriched fraction indicated higher than 95% purity (representative histogram is shown). Right panel: DNA was prepared from MACS-enriched B220+ splenocytes, analyzed by Southern blot, and densitometrically evaluated. The bar graph shows the relative amount of B220+ cells deficient for a functional β1 integrin gene. Error bar shows the standard deviation (n [control BM chimera]/[β1β7 mutant BM chimera]: 5/5).
Normal B-cell population sizes in the absence of β1 and β7 integrins. (A) Representative histogram overlays show the β1 integrin expression on immature (B220med) and mature (B220hi) B cells of BM and spleen, respectively, of control (filled) and β1β7 mutant BM chimeras (line). (B) Single-cell suspensions from spleen of control and β1β7 mutant BM chimeras 6 months after polyIC treatment were prepared, stained with B220-FITC antibody, and subsequently sorted using anti-FITC MACS beads. Left panel: FACS analysis of the B220+-enriched fraction indicated higher than 95% purity (representative histogram is shown). Right panel: DNA was prepared from MACS-enriched B220+ splenocytes, analyzed by Southern blot, and densitometrically evaluated. The bar graph shows the relative amount of B220+ cells deficient for a functional β1 integrin gene. Error bar shows the standard deviation (n [control BM chimera]/[β1β7 mutant BM chimera]: 5/5).
T-cell development in the absence of β1 and β7 integrins
Since in α4-null somatic chimeric mice α4-null T-cell precursors were described to be unable to migrate to the thymus for further differentiation, thymocyte development was analyzed in β1β7 mutant BM chimeric mice using the T-cell markers CD4 and CD8. No significant difference was found in the population sizes of CD4–CD8– (DN) thymocytes, which contain the early thymic immigrants indicating that thymic colonisation was not altered in β1β7 mutant BM chimeric mice which lack both α4β1 and α4β7 integrins. Furthermore, the relative amounts of CD4+CD8+ (DP), CD4+ (CD4SP), and CD8+ (CD8SP) cells in the thymus were normal in β1β7 mutant BM chimeric mice (Figure 3A and Table 2). Staining of thymocytes for β1 integrin and subsequent FACS analysis proved the absence of β1 integrin from DP T cells (Figure 3B). Normal numbers of CD4 and CD8 T cells in spleen, LN, and BM of β1β7 mutant BM chimeras 2 and 12 months after induction of the β1 gene deletion suggested normal migration of these cells to secondary lymphoid organs and to the BM (data not shown). Since mature CD4+ and CD8+ T cells express only low levels of β1 integrin, the deletion efficiency in these populations was tested on the genomic level. Southern blot from CD4+ and CD8+ T cells, enriched from the spleen, revealed that 78.5% ± 5% (n = 3) of CD4+ T cells and 83.4% ± 10.8% (n = 4) of CD8+ T cells lacked a functional β1 integrin gene (Figure 3C). These data show that β1 and β7 integrins are neither essential for the migration of T-cell precursors to the thymus nor for T-cell maturation within the thymus.
β1 and β7 integrins are not essential for normal T-cell development. Single-cell suspensions from thymus of control and β1β7 mutant BM chimeras 2 months after polyIC injection were prepared, stained with antibodies against CD4, CD8, and β1 integrin, and analyzed by FACS. (A) The dot blots show a representative staining of DN, DP, CD4SP, and CD8SP thymocytes for both control and β1β7 mutant BM chimeras. (B) A representative histogram displays β1 integrin expression on DP T cells of control (filled) and β1β7 mutant (line) mice. (C) Single-cell suspensions from spleen of control and β1β7 mutant BM chimeras 6 months after polyIC treatment were prepared, stained with CD4-FITC or CD8-FITC antibody, and subsequently sorted using anti-FITC MACS beads. FACS analysis of the CD4+- or CD8+-enriched fraction indicated higher than 95% purity (representative histogram is shown). DNA was prepared from MACS-enriched CD4+ or CD8+ splenocytes and analyzed by Southern blot and densitometrically evaluated. The bar graphs show the relative amount of CD4+ or CD8+ cells deficient for a functional β1 integrin gene. Error bar shows the standard deviation (n [control BM chimera]/[β1β7 mutant BM chimera]: CD4+, 3/3; CD8+, 4/4).
β1 and β7 integrins are not essential for normal T-cell development. Single-cell suspensions from thymus of control and β1β7 mutant BM chimeras 2 months after polyIC injection were prepared, stained with antibodies against CD4, CD8, and β1 integrin, and analyzed by FACS. (A) The dot blots show a representative staining of DN, DP, CD4SP, and CD8SP thymocytes for both control and β1β7 mutant BM chimeras. (B) A representative histogram displays β1 integrin expression on DP T cells of control (filled) and β1β7 mutant (line) mice. (C) Single-cell suspensions from spleen of control and β1β7 mutant BM chimeras 6 months after polyIC treatment were prepared, stained with CD4-FITC or CD8-FITC antibody, and subsequently sorted using anti-FITC MACS beads. FACS analysis of the CD4+- or CD8+-enriched fraction indicated higher than 95% purity (representative histogram is shown). DNA was prepared from MACS-enriched CD4+ or CD8+ splenocytes and analyzed by Southern blot and densitometrically evaluated. The bar graphs show the relative amount of CD4+ or CD8+ cells deficient for a functional β1 integrin gene. Error bar shows the standard deviation (n [control BM chimera]/[β1β7 mutant BM chimera]: CD4+, 3/3; CD8+, 4/4).
Myeloid and erythroid development in the absence of β1 and β7 integrins
To analyze myeloid development, we first studied the capacity of myeloid progenitors in control and β1β7 mutant BM chimeras lacking α4β1 and α4β7 integrins to form colonies in vitro (CFU-C). All CFU-C colonies analyzed from BM (n = 37), spleen (n = 38), and PB (n = 36) from control mice were positive for β1 integrin. From β1β7 mutant BM chimeras only 2 of 36 colonies from the BM, 1 of 37 of the spleen, and 3 of 37 colonies derived from PB were positive for β1 integrin. These results show first, that in the absence of β1 and β7 integrin granulocyte/monocyte precursors have the potential to form colonies in vitro and second, that the efficiency of the β1 integrin gene deletion is very high in the myeloid lineage. Both control and mutant BM cells also formed erythroid colonies (CFUe's) in vitro. Of 42 colonies tested from mutant BM, none showed a functional β1 integrin gene as tested by genomic PCR, whereas in 24 of 24 colonies from control BM a functional β1 gene was detected.
Monitoring the development of monocytes, granulocytes, and erythroblasts in β1β7 mutant BM chimeric mice 2 and 12 months after the β1 integrin gene deletion in vivo revealed no significant differences in the numbers of granulocytes, monocytes, and erythroblasts, indicating no developmental defects in the absence of both α4β1 and α4β7 integrins (Table 3 and data not shown). The β1 gene deletion on these cells was confirmed by staining for β1 integrin and subsequent FACS analysis (Figure 4). These data strongly suggest that HSCs and HPCs continuously provide myeloid and erythroid cells in the absence of α4β1 and α4β7 integrins.
Emigration of granulocyte/monocyte progenitors from the BM to the blood and spleen
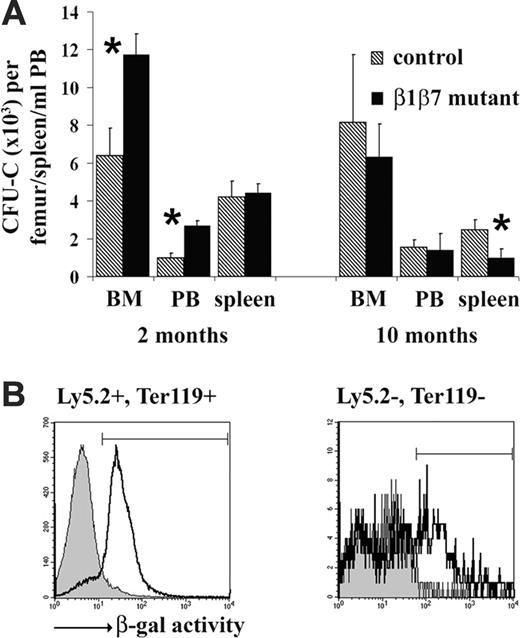
Induced deletion of α4 integrin in hematopoietic and many nonhematopoietic cells resulted in a slow increase of CFU-Cs in the BM, an overproportional release into the PB, and an accumulation of CFU-Cs in spleen, suggesting a role for α4 in the retention of progenitor cells in the BM.17 To test this in β1β7 mutant BM chimeras, we determined the frequency of CFU-C progenitors in BM, PB, and spleen 2 and 10 months after the β1 integrin gene deletion (Figure 5A). At 2 months, the number of precursor cells was significantly elevated in the BM of β1β7 mutant mice as compared with controls. We also observed an increase of progenitors in PB roughly proportional to the progenitor increase in the BM, but significantly less than reported for α4 conditional knockout mice 8 weeks after induced gene deletion, thus not indicating a severe defect in progenitor retention in the BM. Furthermore, these alterations were transient, since they were observed 2 months but not 10 months after knockout induction, when β1β7 mutant and control mice had similar CFU-Cs, both in BM and PB (Figure 5A). Unlike the conditional α4 integrin knockout mice 2 weeks and 6 months after gene deletion,17 β1β7 mutant BM chimeras did not accumulate precursor cells over time in the spleen, as tested 2 and 10 months after the knockout induction (Figure 5A). To the contrary, CFU-Cs were significantly decreased in 10-month-old mutant chimera.
FACS analysis of BM cells of nonchimeric (β1fl/flMx-cre+) mice 3 days after a single polyIC injection revealed that the β1 integrin gene is not only deleted on most hematopoietic cells (Figure 5B, Ly-5.2+, Ter119+), but also on many nonhematopoietic BM stroma cells, defined as (Ly-5.2–, Ter119–) plastic adherent cells (Figure 5B). To assess whether loss of β1 and β7 integrin on nonhematopoietic cells might contribute to the progenitor release, the frequency of progenitor cells was determined in the PB of nonchimeric β1β7 mutant mice 4 weeks after the knockout induction. We found that the progenitor content in PB increased approximately 8-fold in β1β7 mutant mice (data not shown), comparable to the more than 10-fold increase of the CFU-Cs in α4 conditional knockout mice 4 weeks after gene deletion,17 indicating that loss of α4 integrin on nonhematopoietic cells might contribute to the release of CFU-C progenitors from BM to PB.
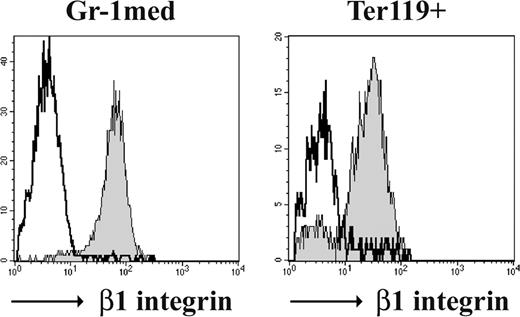
β1 and β7 integrins are not essential for myeloid and erythroid development. The β1 integrin expression of immature granulocytes (Gr-1med) and erythroblasts (Ter119+) of control (filled) and β1β7 mutant BM chimeras (line) is shown in representative histogram overlays.
β1 and β7 integrins are not essential for myeloid and erythroid development. The β1 integrin expression of immature granulocytes (Gr-1med) and erythroblasts (Ter119+) of control (filled) and β1β7 mutant BM chimeras (line) is shown in representative histogram overlays.
No overproportional release of CFU-Cs from the BM into the PB. (A) Single-cell suspensions were made from BM and spleen, and erythrocyte-depleted blood cells (PB) from control and β1β7 mutant BM chimeras were prepared at indicated times after the gene deletion. BM cells (180 000), splenocytes (3 600 000), and PB (250 μL) were seeded into MethoCult GF M3534 medium and counted 7 days later. Total numbers of colonies per femur, spleen, and mL PB are shown. Error bars show the standard deviation, star indicates significant difference (P < .05) (n (control BM chimera)/(β1β7 mutant BM chimera): 2 months 5/5, 10 months 3/3). (B) Single-cell suspensions from BM of nonchimeric control (β1fl/flMx-cre–) and β1fl/flMx-cre+ mutant mice were plated on tissue-culture plastic dishes. After 24 hours, nonadherent cells were removed and adherent cells detached. Adherent and nonadherent cells were then stained for Ly-5.2 and Ter119 and tested for β-galactosidase activity by an FDG assay as described in “Materials and methods.” Since loss of β1 integrin results in expression of the β-galactosidase reporter, 12 high β-galactosidase activity indicates deletion of the β1 gene. Representative histogram overlays show the β-galactosidase activity on hematopoietic (Ly-5.2+ or Ter119+) and nonhematopoietic (Ly-5.2–, Ter119–) cells of control (filled line) and mutant mice (line). The marked region on the overlay indicates cells with high β-galactosidase activity. PolyIC injection induced an efficient deletion of the β1 integrin gene on hematopoietic cells; about 86% of the (Ly-5.2+ or Ter119+) cells of the mutant mice showed high green fluorescence, compared with only 6% of the corresponding cells of the control mice. Also, among the nonhematopoietic BM cells (Ly-5.2–, Ter119–) the percentage of cells with high β-galactosidase activity increased from less than 5% in control to more than 42% in mutant, clearly indicating the presence of β1 integrin–deficient nonhematopoietic cells in the BM of mutant mice.
No overproportional release of CFU-Cs from the BM into the PB. (A) Single-cell suspensions were made from BM and spleen, and erythrocyte-depleted blood cells (PB) from control and β1β7 mutant BM chimeras were prepared at indicated times after the gene deletion. BM cells (180 000), splenocytes (3 600 000), and PB (250 μL) were seeded into MethoCult GF M3534 medium and counted 7 days later. Total numbers of colonies per femur, spleen, and mL PB are shown. Error bars show the standard deviation, star indicates significant difference (P < .05) (n (control BM chimera)/(β1β7 mutant BM chimera): 2 months 5/5, 10 months 3/3). (B) Single-cell suspensions from BM of nonchimeric control (β1fl/flMx-cre–) and β1fl/flMx-cre+ mutant mice were plated on tissue-culture plastic dishes. After 24 hours, nonadherent cells were removed and adherent cells detached. Adherent and nonadherent cells were then stained for Ly-5.2 and Ter119 and tested for β-galactosidase activity by an FDG assay as described in “Materials and methods.” Since loss of β1 integrin results in expression of the β-galactosidase reporter, 12 high β-galactosidase activity indicates deletion of the β1 gene. Representative histogram overlays show the β-galactosidase activity on hematopoietic (Ly-5.2+ or Ter119+) and nonhematopoietic (Ly-5.2–, Ter119–) cells of control (filled line) and mutant mice (line). The marked region on the overlay indicates cells with high β-galactosidase activity. PolyIC injection induced an efficient deletion of the β1 integrin gene on hematopoietic cells; about 86% of the (Ly-5.2+ or Ter119+) cells of the mutant mice showed high green fluorescence, compared with only 6% of the corresponding cells of the control mice. Also, among the nonhematopoietic BM cells (Ly-5.2–, Ter119–) the percentage of cells with high β-galactosidase activity increased from less than 5% in control to more than 42% in mutant, clearly indicating the presence of β1 integrin–deficient nonhematopoietic cells in the BM of mutant mice.
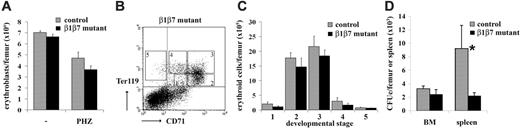
Reduced number of splenic CFUe's after hemolytic stress. (A) Single-cell suspensions from BM of control and β1β7 mutant mice (no BM transplantation) untreated and treated at day 1 and 2 with PHZ were prepared at day 4, stained with Ter119 antibody, and subsequently analyzed by FACS. The total amount of Ter119+ erythroblasts per femur is shown. Error bars show the standard deviation (n (controls)/(β1β7 mutants): 4/4). (B) BM single-cell suspensions from PHZ treated β1β7 mutant BM chimeras were prepared, stained with antibodies against α4, β1 integrin, CD71, and Ter119 and subsequently analyzed by FACS. (Ter119-CD71 staining distinguishes 5 different developmental stages.) (C) The amount of erythroid BM cells of PHZ-treated β1β7 mutant mice for each of the 5 developmental stages as distinguished by Ter119 and CD71 is shown. Error bars show the standard error (n (controls)/(β1β7 mutants): 4/4). (D) Single-cell suspensions from BM of control and β1β7 mutant mice (no BM transplantation) treated at day 1 and 2 with PHZ were prepared at day 4 and tested for CFUe's. Total numbers of colonies per femur and spleen are shown. Error bar shows the standard error (n (controls)/(β1β7 mutants): 3/3).
Reduced number of splenic CFUe's after hemolytic stress. (A) Single-cell suspensions from BM of control and β1β7 mutant mice (no BM transplantation) untreated and treated at day 1 and 2 with PHZ were prepared at day 4, stained with Ter119 antibody, and subsequently analyzed by FACS. The total amount of Ter119+ erythroblasts per femur is shown. Error bars show the standard deviation (n (controls)/(β1β7 mutants): 4/4). (B) BM single-cell suspensions from PHZ treated β1β7 mutant BM chimeras were prepared, stained with antibodies against α4, β1 integrin, CD71, and Ter119 and subsequently analyzed by FACS. (Ter119-CD71 staining distinguishes 5 different developmental stages.) (C) The amount of erythroid BM cells of PHZ-treated β1β7 mutant mice for each of the 5 developmental stages as distinguished by Ter119 and CD71 is shown. Error bars show the standard error (n (controls)/(β1β7 mutants): 4/4). (D) Single-cell suspensions from BM of control and β1β7 mutant mice (no BM transplantation) treated at day 1 and 2 with PHZ were prepared at day 4 and tested for CFUe's. Total numbers of colonies per femur and spleen are shown. Error bar shows the standard error (n (controls)/(β1β7 mutants): 3/3).
Expansion of erythrocyte precursors after hemolytic anemia
After phenylhydrazine (PHZ) induced lysis of erythrocytes in vivo, erythroid precursor cells expand in order to compensate for the loss of erythrocytes. In addition, hemolytic anemia promotes extramedullary erythropoiesis leading to proliferation of progenitors in the spleen.25 Since in α4 conditional knockout mice the ability of erythroblasts to expand in response to a PHZ-induced hemolytic anemia was reduced,17 we investigated whether combined loss of β1 and β7 integrins shows a similar effect. For better comparison with the nonchimeric α4 conditional knockout mice we used nonchimeric β1β7 mutant mice.
Two days after PHZ treatment the amount of erythrocytes dropped in both control and β1β7 mutant mice by more than 55% in BM (n = 3) and was not significantly different between both groups. Similarly, also the number of erythroblasts in the BM as assessed by Ter119 staining was reduced after the PHZ treatment but comparable between β1β7 mutant mice and controls (Figure 6A). Since α4 conditional knockout mice were reported to have fewer erythroid progenitor cells in the BM after hemolytic stress, we tested at the same time point (ie, 2 days after PHZ treatment) the relative amounts of cells of different erythroid developmental stages by Ter119-CD71 staining and subsequent FACS analysis separating different maturation stages of BM erythroblasts18 (Figure 6B). Neither in BM nor in spleen was a significant difference detected between β1β7 mutant and control mice at any of these stages, providing no evidence for an impaired recovery from hemolytic anemia in the absence of β1 and β7 integrin (Figure 6C and data not shown). Analysis of CFUe's confirmed a normal frequency of erythroid progenitors in BM, but surprisingly revealed a significant reduction of CFUe's in the spleen of β1β7 mutant mice compared with controls. Since the spleen is the most prominent place for hematopoiesis after PHZ treatment these data support a role for α4β1 and α4β7 integrin in the recovery of the erythropoietic system after hemolytic anaemia.
No apparent compensatory change in gene expression in the combined absence of β1 and β7 integrin in different leukocyte subsets
To investigate whether hematopoietic subpopulations of β1β7 mutant mice show major alterations in gene expression, we tested mRNA levels of different hematopoietic subsets (DN, DP, B cells, CD4+ T cells) by array analysis. RNA was prepared from DP and DN cells from the thymus, and CD19+ (B cells) and CD4+ cells from the spleen, obtained from 5 pooled mutant and control mice, respectively, and tested on affymetrix chips. All mutant mice had an efficient knockout of β1 integrin indicated by a loss of surface β1 integrin on more than 97% of the platelets.
We then analyzed the data by searching for genes that are up- or down-regulated in mutant mice in all 4 different populations investigated, which would suggest a crucial compensatory response. However, only 3 genes encoding heat shock proteins (heat shock protein 1α, heat shock protein 1β, heat shock protein 105) were found with increased expression in the absence of β1 and β7 integrin. No genes were found with reduced expression in all subpopulations derived from mutant mice.
We then screened the genes up- or down-regulated in the individual hematopoietic subpopulations (thymus: DN, DP; spleen: B cells, CD4+ T cells) for integrins (β3-β6, α2-α10, αX, αD, αM, αL, αE), selectins (P-, L-, E-), CD44, and for the α4β1 and α4β7 integrin ligands VCAM-1 and MAdCAM-1. All these genes showed normal expression in β1β7 mutant cells compared with control cells.
Discussion
It has been reported that several classes of integrins play an important role to attach hematopoietic stem cells and progenitor cells as well as their differentiated lineages to the extracellular matrix and to other cells.1 α4 integrins, for example, have been suggested to be crucial for the retention of hematopoietic stem cells in the bone marrow, for the homing of lymphocytes to Peyer patches and for the migration of T cells during inflammation.13,15,26 In addition, development of the hematopoietic system, characterized by the formation of the different blood cell lineages and their distribution within hematopoietic organs, was reported to be α4 integrin–dependent, although the gradual contribution of α4 integrins differed significantly depending on the experimental approach.7,17 To study the role of α4 integrins in hematopoiesis, mouse models were applied in which the α4 integrin gene was deleted on hematopoietic as well as on nonhematopoietic cells. Loss of the α4 integrins on the latter cell population might affect hematopoietic development. To overcome this problem and to assess by an alternative approach the function of α4 integrins in hematopoiesis, we decided to generate and analyze mice, which lack β1 and β7 integrins, and hence both α4β1 and α4β7 integrins, exclusively in the hematopoietic system. Unexpectedly, we could not find an essential function for α4β1 and α4β7 integrins in blood cell development or in progenitor retention in the bone marrow. Detailed analysis of lymphoid and myeloid development by testing the size of different blood cell subsets in lymphoid organs at different time points and investigating the formation of pre-B and CFU-C colonies did not reveal any impairment in the hematopoiesis of β1β7 mutant mice.
The only abnormality we observed in untreated mutant BM chimeras was an increase in the amount of CFU-C precursors in the bone marrow coupled with a proportional increase in the number of CFU-C progenitor cells in the peripheral blood. This impairment indicates a role of α4 integrins in the maintenance of HPCs. Whether the increase in BM CFU-Cs is due to elevated proliferation, decreased cell death, or changed migratory behavior of progenitor cells is currently unclear. The increase, however, was only transient, since it was detected 2 months after knockout induction, but not 8 months later, pointing to compensatory mechanisms that kick off in the β1β7 mutant mice leading to a reduction of progenitor numbers in BM and PB back to normal levels. Alternatively, it is possible that transplantation-dependent effects affect β1β7 mutant and control mice with different efficiencies, thus contributing to the reduction of CFU-Cs. While in α4 integrin conditional knockout mice HPCs accumulate in spleen,17 this was not the case in β1β7 mutant BM chimeras. To the contrary, relative to PB the number of HPCs in spleen was decreased in young and old β1β7 mutant BM chimeras, which might indicate a migration defect of β1β7 mutant HPCs to the spleen. Interestingly, no increased CFU-Cs were observed in β1-null BM chimera.
Induction of hemolytic anemia revealed a reduced number of splenic CFUe's in β1β7 BM chimera, indicating a role for α4β1 and α4β7 integrin in erythroid recovery after stress. Also, α4 mutant mice showed an impaired recovery after PHZ-induced hemolysis, although in that case the frequency of CFUe's was reduced in BM and not in spleen.17
Different explanations might be possible to reconcile the published data with the findings of our investigations. First, in α4-null somatic chimera α4 integrin is lost already before the development of hematopoietic stem cells, whereas in our system α4β1 and α4β7 are lost in adult animals 1 month after bone marrow transplantation. It is possible, therefore, that the absence of these integrins during the entire embryogenesis impairs development of the hematopoietic system and causes defects that persist to adulthood, whereas the loss of α4 in adult animals has no severe consequences for hematopoiesis. However, recent data by Gribi et al27 showed that transplantation of fetal α4-null hematopoietic stem cells derived from the aorta-gonad-mesonephros (AGM) region of α4-deficient embryos into adult microenvironment results in long-term generation of mature B and T lymphocytes and myeloid cells. These data indicate that neither fetal nor adult HSCs require α4 integrins for hematopoietic development.
Second, the defects observed in α4 integrin conditional knockout mice might be related to the fact that in this model α4β1 and α4β7 integrin are lost also on nonhematopoietic cells, whereas deletion of the β1 and β7 integrin in our BM chimeras was restricted to the hematopoietic system. Interference with the functions of α4 integrins in nonhematopoietic tissues such as BM stroma cells or endothelial cells might result in the production of cytokines, which inhibit lymphopoiesis or favor the mobilization of HPCs. We tested this possibility and found that nonchimeric β1β7 mutant mice, which lack α4β1 and α4β7 integrin with a similar tissue distribution as the α4 conditional knockout mice, do show an overproportional release of HPCs into the peripheral blood. Furthermore, we could demonstrate that induction of Cre in these mice disrupts the β1 integrin gene in BM stromal cells. Altogether, these data underline the importance of a strict tissue-restricted gene deletion in order to avoid unwanted cross-talk between different tissues.
In summary, we demonstrate that α4β1 and α4β7 integrins are not crucial for the differentiation of lymphoid and myeloid cells in adult mice. In fact, even the additional loss of all other β1 integrin receptors besides α4β1, as occurring in our β1β7 mutant mice, did not prevent hematopoietic development. β1 and β7 integrins affect maintenance and distribution of CFU-C progenitors, though differently than previously suggested for α4 integrins.17 Our results do not exclude effects of α4β1 and α4β7 integrins on fetal hematopoiesis, during immune response or in inflammation.
Prepublished online as Blood First Edition Paper, May 30, 2006; DOI 10.1182/blood-2005-10-007658.
Supported by the Max Planck Society and the German Research Council (DFG BR 2201/1; DFG FOR471/2 DFG SFB571).
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Dr Bernhard Nieswandt for generously providing antibodies, Dr Robert Geffers for generating array data, Dr Lothar Gröbe for flow cytometry assisstance, and Dr Alexander Medvinsky for discussion and sharing unpublished data.

![Figure 1. Efficient deletion of β1 integrin in the hematopoietic system does not affect cellularity of lymphoid organs. (A) Bone marrow of mice with a conditional knockout of β1 integrin, an Mx-cre transgene, and a constitutive knockout of β7 integrin (β1fl/–β7–/–Mx cre) was transplanted into lethally irradiated wild-type host mice. In bone marrow chimeras, donor and host cells could be distinguished by expression of Ly-5.1/Ly-5.2 surface marker. After repopulation of the hematopoietic system (4 weeks), polyIC was injected to induce expression of the cre recombinase in the donor cells and deletion of the β1 integrin gene. (B) Percentages of β1 integrin–negative platelets isolated from control and β1β7 mutant BM chimeric mice are shown at indicated time points after the first of 3 polyIC injections (day 0). Error bars show the standard deviation (n [control BM chimera]/[β1β7 mutant BM chimera]: 9/6). (C) DNA was isolated from single-cell suspensions from BM, thymus, and spleen from β1β7 mutant BM chimeras 10 months after polyIC treatment. Southern blot analysis detecting the conditional and the null allele was performed (a representative result is shown on the right panel). Band intensities were quantified and visualized in a bar graph. Error bars show the standard deviation. (n [control BM chimera]/[β1β7 mutant BM chimera]: 3/3). (D) Left panel: Single-cell suspensions were made from BM (2 femurs), thymus, spleen, and lymph nodes (2 inguinal, 2 axial, 1 para-aortic) of control and β1β7 mutant BM chimeric mice 2 months after the gene deletion. Cells were counted using a hemacytometer. The bar graph shows the absolute cell number in the respective tissues. Error bars show the standard deviation (n [control BM chimera]/[β1β7 mutant BM chimera]: 4/4). Right panel: Whole blood was collected retro-orbitally from control and β1β7 mutant BM chimeric mice 6 months after the gene deletion. The blood was diluted 1:10 with Türk stain and differentially counted in a hemacytometer. The bar graph shows the concentration of mononuclear (M) and polymorphonuclear (PMN) cells in the blood. Error bars show the standard deviation (n [control BM chimera]/[β1β7 mutant BM chimera]: 4/4).](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/108/6/10.1182_blood-2005-10-007658/4/m_zh80180601020001.jpeg?Expires=1765368709&Signature=O3O2dGdoCKkWhh4858-eyRqJZ276mq8C~hF5FtdDaGf28xR9FNyTKC6Q3hH25Hj-ji1DsSiZkKSCh3MdO3mwgm3Wx499KtUk1c3Io1Ft5AecwH2zK~2Du7wvjjY5E6oeRUrl7vgSwsAsLLtY1~Eyv9AptS9K1GgNf1HdPdOzrUrLyuleOF1AbK22ZRXyzLXSoZotA8SwuCxWCOM7w3YUfiIYyr5JQ3hl2Z5vx~QneWWfy74axKGvMHmGltcNa7O038Ri5~lZcuZ51H0GBj~dGOGijqe8qoIEGb2SwLR7OaKyHfqmtUhgAdwTXmXDfWw8q737slND8GF-p9BrkYeo8w__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Figure 2. Normal B-cell population sizes in the absence of β1 and β7 integrins. (A) Representative histogram overlays show the β1 integrin expression on immature (B220med) and mature (B220hi) B cells of BM and spleen, respectively, of control (filled) and β1β7 mutant BM chimeras (line). (B) Single-cell suspensions from spleen of control and β1β7 mutant BM chimeras 6 months after polyIC treatment were prepared, stained with B220-FITC antibody, and subsequently sorted using anti-FITC MACS beads. Left panel: FACS analysis of the B220+-enriched fraction indicated higher than 95% purity (representative histogram is shown). Right panel: DNA was prepared from MACS-enriched B220+ splenocytes, analyzed by Southern blot, and densitometrically evaluated. The bar graph shows the relative amount of B220+ cells deficient for a functional β1 integrin gene. Error bar shows the standard deviation (n [control BM chimera]/[β1β7 mutant BM chimera]: 5/5).](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/108/6/10.1182_blood-2005-10-007658/4/m_zh80180601020002.jpeg?Expires=1765368709&Signature=Q-pNLuPWtzlztxKGf92kmGgT8D6tAEXVUCmUE6QUjDwL~IFRrqjxHhG9hw7ZNE149AIG-O0MhEBw0UCv42872-pHt~Kju2nz9S27Moa5KGj9oneAMQhIyNKuYn8yAZVavtv1Mg59E6D0hv6qVdwdMJNDoTHMLFVnEnUNwRIhxN4PdG2Lku1IvFS4-3l5PAu-REWDv1d9UlsHdOTPrPNZ5ttjGUr1Fow~87BV8UqJ3yZlUC9kbR1ywmiX7gppCQPs0xHQ-AVkrjIbNODsW5gdN34pBENyji3NiZcKiV6Bf5Fvucmo0kbMfa2m2JkMetceazD9mbvhQCBnqzSXO-86IQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Figure 3. β1 and β7 integrins are not essential for normal T-cell development. Single-cell suspensions from thymus of control and β1β7 mutant BM chimeras 2 months after polyIC injection were prepared, stained with antibodies against CD4, CD8, and β1 integrin, and analyzed by FACS. (A) The dot blots show a representative staining of DN, DP, CD4SP, and CD8SP thymocytes for both control and β1β7 mutant BM chimeras. (B) A representative histogram displays β1 integrin expression on DP T cells of control (filled) and β1β7 mutant (line) mice. (C) Single-cell suspensions from spleen of control and β1β7 mutant BM chimeras 6 months after polyIC treatment were prepared, stained with CD4-FITC or CD8-FITC antibody, and subsequently sorted using anti-FITC MACS beads. FACS analysis of the CD4+- or CD8+-enriched fraction indicated higher than 95% purity (representative histogram is shown). DNA was prepared from MACS-enriched CD4+ or CD8+ splenocytes and analyzed by Southern blot and densitometrically evaluated. The bar graphs show the relative amount of CD4+ or CD8+ cells deficient for a functional β1 integrin gene. Error bar shows the standard deviation (n [control BM chimera]/[β1β7 mutant BM chimera]: CD4+, 3/3; CD8+, 4/4).](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/108/6/10.1182_blood-2005-10-007658/4/m_zh80180601020003.jpeg?Expires=1765368709&Signature=Uz1~GA5aHYadXpOnyDcIf9GbVGeEk6beFhl9y136QsH14jeOocCI9fS2prfa-EWwub7XVPPfkXFUeh8-fo3DZ12Nl2VMX3KEN~VPfJ1QDETQuHWOX03ovxNFkOSkT9vWdCBqYERHT47~CgDjJKFZj0Gc3f7j-bm4ojRzBRZB4hfSxDiIEMvWgzcQwSqYIFpDF0k1Ma9TzpLP4apqDQCX20jZBE6g0M8M1Xh~zRkbNOHufzkkXQKNas5AiI-PWvJazHrIKV8F3YN2EsCjb8o2wq-NTDGGDaxx1ZFZ9DpCsHR1NHrKmJe2-90XDoUdMwhEzPJgaxmwoBQuGT7fRSSQGg__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)