Abstract
A high incidence of autologous graft-versus-host-disease (auto-GVHD) was observed after an alemtuzumab-containing conditioning regimen and autologous stem cell transplantation (auto-SCT) for chronic lymphocytic leukemia (CLL). Skin rash developed in almost all surviving patients (87%). In 7 patients (58%), a diagnosis of auto-GVHD was made (compared with 0% after TBI/Cy; P = .01). All patients with auto-GVHD required immunosuppression, and 3 of 7 were hospitalized because of GVHD. The median duration of GVHD was 517 days (range, 60-867 days). Auto-GVHD was associated with an abnormally high CD4/CD8 ratio because of severe depletion of CD8+ T cells, pointing to a potential pathomechanism. High non–relapse-related mortality led to the discontinuation of the trial. Current results do not support the use of high-dose alemtuzumab combined with total body irradiation (TBI) and autologous stem cell transplantation (auto-SCT). However, the addition of alemtuzumab led to improved disease control at the molecular level. Longer follow-up will show whether the GVHD-like syndrome may contribute to prolonged minimal residual disease (MRD) negativity.
Introduction
Autologous graft-versus-host disease (auto-GVHD) is an autoimmune syndrome initiated by auto-effector T cells that recognize self–major histocompatibility complex (MHC) class II antigens.1-3 Although auto-GVHD is rarely observed after conventional autologous stem cell transplantation (auto-SCT), the syndrome is commonly associated with the use of short-course cyclosporine (CSA), and trials have been conducted to exploit a potential antitumor effect.1,2 Autologous SCT in patients with chronic lymphocytic leukemia (CLL) may prolong the treatment-free interval but is unlikely to lead to cure.4-7 This observation and the known effect of alemtuzumab on minimal residual disease (MRD) clearance led to the integration of alemtuzumab into the conditioning regimen before auto-SCT within a trial of the German CLL Study Group (GCLLSG).8-10 A high incidence of initially unexplained skin rashes among patients treated in this trial led to the current analysis.
Study design
Between July 2000 and June 2004, we treated 27 patients with CLL (CLL3 and CLL3C trials) with auto-SCT at the University of Ulm. Patients received cytoreduction with fludarabine plus cyclophosphamide and stem cell mobilization with dexamethasone BCNU, etoposide, ara-C, melphalan (Dexa-BEAM). In the CLL3 trial (n = 11), auto-SCT was performed after conditioning with 12 Gy total body irradiation and cyclophosphamide (TBI/Cy; 120 mg/kg). Patients in the CLL3C trial (n = 16) received additional alemtuzumab before SCT (day –10, 3 mg; day –9, 10 mg; day –8, 30 mg; day –6, 30 mg; day –4, 30 mg; day –2, 30 mg [n = 1])/(actual doses given: 43 mg, n = 1; 103 mg, n = 14; 133 mg, n = 1). The standard TBI/Cy group received a CD34-selected graft, but only 1 patient in the alemtuzumab/TBI/Cy (Alem/TBI/Cy) group received a CD34-selected graft (mean CD34 count, 6.0 × 106/kg vs 7.6 × 106/kg). The distributions of age (53 vs 52 years), male sex (91% vs 75%), and Binet B stage (82% vs 87%) were similar between the 2 groups.
GVHD was diagnosed by the appearance of the typical skin rash (graded according to the Glucksberg criteria), other organ involvement, and biopsy findings.11 Acute skin reactions after alemtuzumab and infections with skin involvement were not included as skin rashes. Because the study was not designed to test whether alemtuzumab can trigger GVHD, skin biopsy specimens and extensive information on the rash were not prospectively collected and not available for all patients. Approval for these studies was obtained from the University of Ulm institutional review board. Informed consent was provided according to the Declaration of Helsinki.
Results and discussion
Two patients in the TBI/Cy control group died, compared with 5 patients in the group receiving Alem/TBI/Cy (P = .10) (Figure S1A, available on the Blood website; see the Supplemental Figures link at the top of the online article). No deaths were caused by nonrelapse mortality (NRM) in the standard TBI/Cy group, compared with 4 in the Alem/TBI/Cy group, which led to the discontinuation of the CLL3C trial (Figure S1B) (P = .07).
No patients in the standard TBI/Cy group had unexplained skin rashes or auto-GVHD. In contrast, unexplained skin rashes developed in 12 of 16 patients receiving Alem/TBI/Cy (maculopapular rash, n = 5; erythrodermia, n = 3; eczema, n = 3) between 43 and 601 days after SCT (Table 112,13 ; Figure S2A-B). Eleven of these patients were treated with steroids.
In 7 patients, a clinical diagnosis of auto-GVHD was made (Table 1). The cumulative incidence of auto-GVHD among the patients alive at 1 year was 58% for those treated with Alem/TBI/Cy, compared with 0% for those treated with TBI/Cy (competing risk analysis, P = .01). Typically, concurrent symptoms included conjunctivitis (n = 4), sicca syndrome (n = 5), and cholestasis (n = 4). Histologic findings were compatible with GVHD grades 1 and 2 in all 5 patients with clinical auto-GVHD in whom skin biopsy was performed. Histologic examination revealed interface dermatitis with perivascular lymphocytic infiltrates, epithelial tropism of lymphocytes, focal vacuolization of epidermal basal cells, and some intraepithelial apoptotic figures (Figure S2C).14 Pathology specimens were reviewed independently by several expert pathologists.
Characteristics of auto-GVHD are onset very early after SCT, mild and self-limiting course, and confinement primarily to the skin.1,2 In contrast, patients treated with Alem/TBI/Cy acquired GVHD between days 43 and 324, and the symptoms led to severe impairment (n = 4) and hospitalization (n = 3). All patients were treated with steroids, and 2 patients needed further immunosuppression with CSA, mycophenolate mofetil, or FK506. Median duration of GVHD was 517 days (range, 60-867 days), and the reduction of immunosuppression led to a flare of the skin rash in 5 of 7 patients. Signs and symptoms resolved in 3 patients. Conditions improved in another 3 patients, though they continued to have skin rashes and symptoms. One patient developed diarrhea—a severe wasting syndrome requiring home total parenteral nutrition (TPN)—and eventually died of pneumonia while receiving immunosuppression therapy. Three patients developed additional autoimmune diseases (Guillain-Barré syndrome, autoimmune thyroiditis, Hashimoto thyroiditis). A high incidence of thyroid autoimmune disease has been reported for patients with multiple sclerosis (MS) receiving alemtuzumab.15 A GVHD-like syndrome should be included in the differential diagnosis of unclear skin rashes in heavily pretreated (immunosuppressed) patients receiving alemtuzumab.
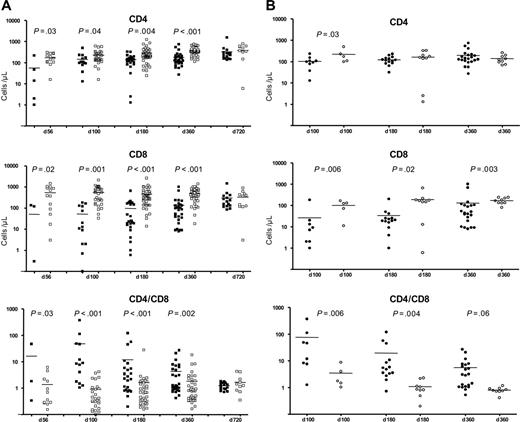
CD4/CD8 reconstitution after Alem/TBI/Cy, compared with that after TBI/Cy and auto-SCT. (A) CD4 and CD8 counts (per microliter) and CD4/CD8 ratios at different time points after SCT for patients receiving Alem/TBI/Cy (n = 16) (▪) and patients receiving TBI/Cy (University of Ulm and Kiel) (n = 14) (□). P values (t test) at different time points are shown if they are lower than .05. Patients in the Alem/TBI/Cy group experienced severe depletion of CD4 and CD8 cells with a highly abnormal CD4/CD8 ratio in the first year after SCT caused by severe CD8 lymphopenia. As shown in panel B, the difference was caused by the severe depletion of CD8 cells (hence the high CD4/CD8 ratio) in patients with auto-GVHD. (B) CD4 and CD8 counts (per microliter) and CD4/CD8 ratios at different time points after SCT for patients receiving Alem/TBI/Cy who acquired GVHD (n = 7) (•) and patients receiving Alem/TBI/Cy who did not acquire GVHD (n = 9) (○). P values (t test) at different time points are shown if they are lower than .05. Although the differences in CD4 cell counts were not significant, differences in CD8 counts and CD4/CD8 ratios were highly significant at different time points in the first year after SCT. Bars represent mean value.
CD4/CD8 reconstitution after Alem/TBI/Cy, compared with that after TBI/Cy and auto-SCT. (A) CD4 and CD8 counts (per microliter) and CD4/CD8 ratios at different time points after SCT for patients receiving Alem/TBI/Cy (n = 16) (▪) and patients receiving TBI/Cy (University of Ulm and Kiel) (n = 14) (□). P values (t test) at different time points are shown if they are lower than .05. Patients in the Alem/TBI/Cy group experienced severe depletion of CD4 and CD8 cells with a highly abnormal CD4/CD8 ratio in the first year after SCT caused by severe CD8 lymphopenia. As shown in panel B, the difference was caused by the severe depletion of CD8 cells (hence the high CD4/CD8 ratio) in patients with auto-GVHD. (B) CD4 and CD8 counts (per microliter) and CD4/CD8 ratios at different time points after SCT for patients receiving Alem/TBI/Cy who acquired GVHD (n = 7) (•) and patients receiving Alem/TBI/Cy who did not acquire GVHD (n = 9) (○). P values (t test) at different time points are shown if they are lower than .05. Although the differences in CD4 cell counts were not significant, differences in CD8 counts and CD4/CD8 ratios were highly significant at different time points in the first year after SCT. Bars represent mean value.
Because of the documented allogeneic graft-versus-leukemia effect in CLL, we analyzed the effect of GVHD on progression-free survival (PFS) and MRD.16-18 No difference in PFS was observed (Figure S1C). The addition of alemtuzumab led to continuous MRD negativity in 10 of 14 (71%) patients receiving Alem/TBI/Cy, compared with 0 of 7 patients receiving TBI/Cy (Fisher exact test: P < .001). Within the Alem/TBI/Cy group, continuous MRD negativity was observed in 6 of 6 patients with auto-GVHD, compared with 4 of 8 patients without auto-GVHD (Fisher exact test: P = .08) (Figure S3).
Because immune reconstitution data were available in only 5 patients receiving TBI/Cy, we pooled the data with a control group of CLL patients (n = 9) undergoing auto-SCT within the same protocol at the University of Kiel. The use of Alem/TBI/Cy before auto-SCT led to a delayed reconstitution of white blood cells, lymphocytes, NK cells, and particularly T lymphocytes. The CD4/CD8 ratio was abnormally high in the Alem/TBI/Cy group (Figure 1A). This increased ratio was mainly caused by the extreme CD4/CD8 ratio imbalance among patients with auto-GVHD in contrast to patients without GVHD (Figure 1B). Distinct subgroups of CD4+ cells, CD8+ cells, and NK cells have been shown to exhibit regulatory functions.19,20 It is tempting to speculate that the addition of alemtuzumab to a TBI-containing conditioning regimen resulted in a depletion of regulatory T cells and the development of auto-GVHD. Future trials to exploit the activity of alemtuzumab may have to combine alemtuzumab with a less immunosuppressive conditioning regimen (eg, BEAM). Although the conclusions of the current work should be interpreted with caution because of its retrospective nature, its low overall numbers, and some differences in graft composition, our report shows profound clinical differences in course and presentation of auto-GVHD after alemtuzumab compared with previous reports on auto-GVHD.1,2 Although the lack of fluorescence-activated cell sorter (FACS) analyses with the appropriate markers (eg, CD25) did not allow further investigation, we were able to show a particular immune “signature“in patients with auto-GVHD. The use of alemtuzumab before allogeneic SCT results in effective prevention of GVHD when combined with further immunosuppression (eg, CSA).21,22
In summary, the current study demonstrates a remarkable incidence of a severe GVHD-like syndrome attributable to the addition of alemtuzumab to the TBI/Cy conditioning regimen before auto-SCT in patients with CLL. Although the concept of in vivo purging appeared attractive, the current results do not support the use of high-dose alemtuzumab combined with TBI and auto-SCT. Adding alemtuzumab to the conditioning regimen led to improved disease control at the molecular level, which can be attributed to the direct effect of alemtuzumab. Improved MRD control may be enhanced by a graft-versus-leukemia effect; however, any impact of GVHD on MRD was difficult to assess in this study because alemtuzumab itself was also capable of inducing MRD negativity.
Prepublished online as Blood First Edition Paper, May 25, 2006; DOI 10.1182/blood-2006-04-007898.
Supported by Deutsche Krebshilfe (grant 106116), Wilhelm-Sander-Stiftung (grant 2001.0042), Carreras Stiftung (grant DJCLS R02/18), and Medac Schering Onkologie.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank the GCLLSG office for its support. We thank Prof Dr Scharfetter-Kochanek for sharing skin biopsy samples, Kathrin Ulmer, Silja Groner, and Claire Schleich for data management, and Prof Dr Andreas Mackensen for discussions.
The CLL3 and CLL3C protocols are trials of the German CLL Study Group (GCLLSG; http://dcllsg.de; chairman, Prof Dr Michael Hallek).