Thirty-six patients with AIDS-associated Kaposi sarcoma (KS) requiring chemotherapy were treated for six 3-week cycles of pegylated liposomal doxorubicin (20 mg/m2) plus interleukin-12 (IL-12; 300 ng/kg subcutaneously twice weekly), followed by 500 ng/kg subcutaneous IL-12 twice weekly for up to 3 years. All received highly active antiretroviral therapy (HAART). Twenty-two had poor-prognosis KS (T1S1). Thirty patients had a major response, including 9 with complete response, yielding an 83.3% major response rate (95% confidence interval: 67.2%-93.6%). Median time to first response was 2 cycles. Median progression was not reached at median potential follow-up of 46.9 months. Of 27 patients with residual disease when starting maintenance IL-12, 15 had a new major response compared with this new baseline. The regimen was overall well tolerated; principal toxicities were neutropenia, anemia, transaminitis, and neuropsychiatric toxicity. Patients had increases in serum IL-12, interferon gamma, and inducible protein-10 (IP-10), and these remained increased at weeks 18 and 34. The regimen of IL-12 plus liposomal doxorubicin yielded rapid tumor responses and a high response rate in patients with AIDS-KS receiving HAART, and responses were sustained on IL-12 maintenance therapy. A randomized trial of IL-12 in this setting may be warranted. This study is registered at http://www.clinicaltrials.gov as no. NCT00020449.
Introduction
Kaposi sarcoma (KS) is a multicentric angioproliferative neoplasm caused by Kaposi sarcoma–associated herpes virus (KSHV), also called human herpes virus-8 (HHV-8).1,2 In the early years of the AIDS epidemic, KS was a frequent cause of morbidity and mortality.3,4 The development of highly active antiretroviral therapy (HAART) has resulted in decreased risk of KS in HIV-infected patients.5,–7 However, KS remains a major complication of HIV infection.8
Certain KS patients respond to HAART, especially those with limited tumor burden who are naive to antiretroviral therapy.9,–11 Nevertheless, HAART alone is inadequate for patients with aggressive KS,12,13 and such patients are generally treated with a liposomal anthracycline plus HAART.14,15 While response rates of 70% or so are observed,16 aggressive KS is still associated with high mortality.8,17,18 Moreover, such therapy is not curative, and can be limited by cumulative toxicity. Advances in KS therapy are thus urgently needed.
IL-12 is a heterodimeric cytokine that can promote development of Th1-type helper T cells, induce production of interferon gamma (IFN-γ), facilitate cytotoxic T-cell responses, and enhance natural killer (NK) cell lytic activity.19,,–22 There are several mechanisms by which IL-12 might have activity in KS. IL-12 has antitumor activity in murine models23,24 and can enhance activity against other herpesviruses.25 IL-12 can manifest antiangiogenic activity through induction of IFN-γ and subsequent induction of interferon inducible protein-10 (IP-10) and monokine induced by interferon gamma (MIG).26,27 Finally, IP-10 has been shown to down-regulate a constitutively active G protein–coupled receptor encoded by KSHV ORF74 that is believed important in KS pathogenesis.28,,,–32
In a pilot study of IL-12 in AIDS-KS, we identified doses that were tolerated and induced major responses in 71% of patients.33 However, tumor responses often took several months, which could limit utility in patients needing more immediate relief. Based on models showing enhancement of antitumor activity from combination antiangiogenic and cytotoxic therapy,34,,–37 we hypothesized that a regimen of IL-12 and liposomal anthracyclines might yield rapid KS tumor responses. To test this, we initiated a trial in which patients with advanced KS were treated with a combination of pegylated liposomal doxorubicin and IL-12 for 18 weeks, followed by IL-12 alone.
Patients and methods
Study population
Eligible patients were HIV-seropositive individuals 18 years or older with biopsy-proven KS for whom cytotoxic chemotherapy was considered as indicated treatment. The criteria used to consider cytotoxic chemotherapy as indicated included the presence of symptomatic visceral or pulmonary disease, painful tumor-associated edema, ulcerating KS, decreased mobility due to tumor-associated pain or restricted range of joint motion, KS that was unresponsive to prior HAART and/or chemotherapy, and social withdrawal due to KS. In general, patients had to have KS that was worsening in the 3 weeks prior to screening, despite the patient taking stable regimen of HAART for a minimum of 4 weeks; however, this requirement could be waived for patients needing urgent KS therapy. Karnofsky performance score had to be at least 30% and expected survival 2 months. Inclusion criteria included total bilirubin level of 64.98 μM (3.8 mg/dL) or less, cardiac ejection fraction of 40% or more, hemoglobin level of 90 g/L (9.0 g/dL) or higher, and absolute neutrophil count (ANC) of 750 cells/mm3 (750×109/L) or higher. Exclusion criteria included history of hepatic cirrhosis; AST/GOT more than 2.5 times the upper limit of normal; serum creatinine level higher than 132.6 μM (1.5 mg/dL) and estimated or measured creatinine clearance less than 1.002 mL/s (60 mL/min); and clinically significant autoimmune disease. Patients could not have received chemotherapy within 21 days; cytokines or bone marrow–stimulating factors other than erythropoietin, filgrastim, or sargramostim within 2 weeks; or high-dose steroids within 2 months. The protocol and informed consent were approved by the National Cancer Institute (NCI) Institutional Review Board, and informed consent was obtained from all patients in accordance with the Declaration of Helsinki.
Treatment regimen
Patients were treated as outpatients at the National Institutes of Health (NIH) Clinical Center with six 3-week cycles of pegylated liposomal doxorubicin (Doxil, obtained from commercial sources) at a dose of 20 mg/m2 administered intravenously over 30 minutes every 3 weeks, and with 300 ng/kg twice-weekly subcutaneous interleukin-12. IL-12 was provided by the Cancer Therapy Evaluation Program (CTEP) of the NCI through an agreement with Genetics Institute (Cambridge, MA). IL-12 was administered by the nursing staff or self-administered. After 18 weeks, pegylated liposomal doxorubicin was discontinued and patients received maintenance subcutaneous IL-12 at 500 ng/kg twice weekly for up to 3 years. Filgrastim was used as needed to maintain absolute neutrophil counts above 1000/mm3.
Induction cycle delays and dose adjustments were allowed for drug toxicity. Recurrent drug toxicity after dose reduction for toxicity resulted in removal from study. Other off-study criteria included disease progression that required additional cytotoxic therapy or was life threatening.
Response evaluations
Patients were evaluated at baseline, weekly for 6 weeks, every 3 weeks during combination therapy, and then every 4 weeks. Toxicity was graded using the NCI Common Toxicity Criteria version 2.0. Patients were assigned at entry into good- or poor-prognostic KS groups based on tumor involvement (T), immunologic status (I), and systemic illness (S).4,18 Assessments of visible lesions were made every 3 weeks during combination therapy and every 4 weeks thereafter. Radiologic and photographic documentation was periodically obtained. Response assessments of complete response (CR), partial response (PR), progressive disease (PD), or stable disease (SD) were made using a minor modification of the method developed by the AIDS Clinical Trial Group38 as described.39 If CR was confirmed by biopsy, it was called pathological complete response (PCR), and if not, clinical complete response (CCR). Duration of response was defined as the interval between first achievement of response and the time when a patient met criteria for PD. Certain patients were coenrolled on a study of thermography and laser Doppler imaging as novel techniques to evaluate KS lesions as has been previously reported40 ; only representative lesions were assessed using these experimental techniques, and the results were not used to assess the patient in the overall study as reported here.
Analysis of virologic and immunologic parameters
HIV-1 viral loads were measured by quantitative RNA polymerase chain reaction (Roche Amplicor HIV-1 Monitor Kits; Roche Diagnostic Systems, Branchburg, NJ) with lower limit of detection of fewer than 50 virions/mL. Lymphocyte subsets were assessed by fluorescent activated cell sorting.41 Pharmacokinetic studies were performed on the initial 9 patients in whom this was feasible after the first dose of IL-12 (before receiving pegylated liposomal doxorubicin), after induction cycle 6, and at 4 months of maintenance. If a patient came off study before pharmacokinetics were completed, pharmacokinetics were obtained on a subsequent patient until 9 were assessed at 3 time points. Blood was obtained prior to IL-12 injection and 24 hours after injection. In addition, blood was obtained at 2, 4, 6, 8, 12, and 18 hours after the first dose of IL-12. Serum was assayed for IL-12, IFN-γ, and IP-10 using enzyme-linked immunosorbent assays (R&D Systems, Minneapolis, MN).
Statistical considerations
The trial used a 2-stage MinMax design to rule out a 55% response rate (PR + CR) and targeting 75% responses, using alpha = 0.05 and beta = 0.20, with total accrual of 36 patients if 16 or more responses were noted in the first 24 patients.42 Changes in CD4 counts, log10 viral load, and serum cytokine levels were evaluated by subtracting the entry value from subsequent time points, and tested for being different from zero using a Wilcoxon signed rank test. A value of 50 copies/mL was used for HIV viral loads below the level of detection. Differences in log10 values of cytokines were used for pharmacokinetic analysis. The Jonckheere-Terpstra test for trend was used to assess the significance of a difference in CD4 cell counts according to 3 ordered categories of clinical response.43 A Cochran-Armitage trend test was used to determine whether CD4 increments above baseline in 3 ordered categories affected tumor response.9,44 Probability of progression-free survival was determined by the Kaplan-Meier method. All P values are 2-sided.
Results
Patient characteristics
Between January 2001 and October 2003, 36 patients were enrolled (Table 1). Median age was 36.5 (range: 21-54) years. Median CD4 cell count and log10 HIV-1 viral loads were 161.5 (range: 10-1060) cells/mm3 and 2.385 (range: 1.69-5.72) copies HIV-1 mRNA/mL plasma. By revised TS criteria,18 22 (61%) had poor-prognosis KS (T1S1). Twenty-four patients had more than 50 cutaneous lesions. Thirteen patients had pulmonary involvement, and 12 patients had biopsy-documented visceral and/or nodal involvement. All patients had KS that was progressing within the past 3 weeks. Three patients requiring urgent KS therapy had had HAART initiated within a month of entry; all the rest had been on HAART for at least a month and up to 7 years. Twenty-three patients had received prior KS therapy, including 13 who had received cytotoxic chemotherapy.
Treatment and outcome
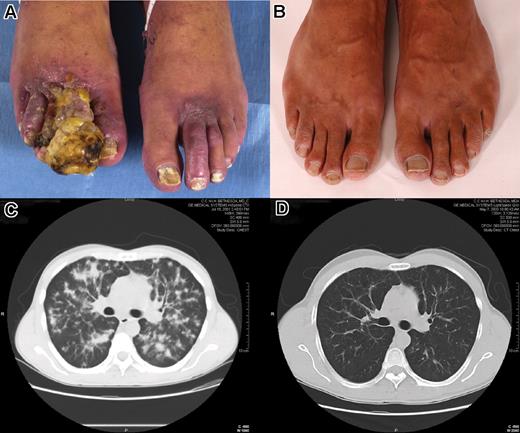
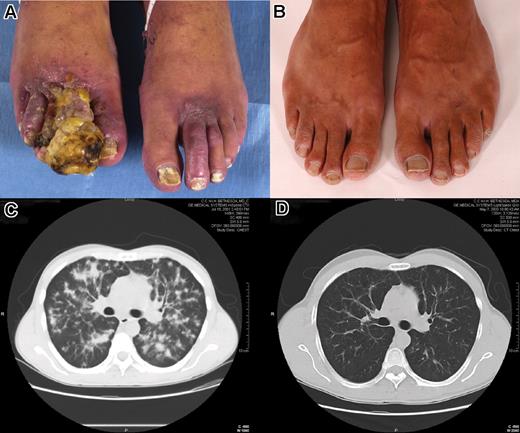
All 36 patients were assessed for response. Thirty patients (83.3%) received the 6 cycles of induction therapy. Major response (PR or CR) was documented in 30 patients (83.3%; 95% exact confidence interval: 67.2% to 93.6%) (Table 2). Median time to PR was 2 cycles. Nine patients (25%) had CR and 5 of these were PCR by biopsy. Two of the CR were achieved during combination treatment, and the remaining 7 occurred during IL-12 maintenance. Twenty-one patients (58%) had PR. Two patients had SD, and 4 patients had PD. Thirteen patients had received prior cytotoxic chemotherapy to which none had had a major response. Ten of these had a major response while being treated with pegylated liposomal doxorubicin and IL-12, including 3 who were documented to have a complete response. Two of these 10 patients who responded subsequently had disease progression, one at 6.7 and the other at 10 months. Figure 1 shows the initial tumors and outcome in a patient with CR.
Photographs of the feet and computed tomographs of the chest of a patient treated with IL-12 and liposomal doxorubicin. Images in panels A and C were taken at baseline; those in panels B and D were taken at 22 months of treatment, the time of complete response. This patient was docu-mented to have a partial response at 4 weeks. Panels A and B were taken by NIH Patient Photographic Services using Nikon Digital SLR cameras (D1 and D1X respectively). Computed tomography scans were acquired from the NIH Clinical Center Diagnostic Radiology Department using the Kodak Carestream PACS web-based program. Images were imported into Adobe Photoshop v7 (Adobe Systems, San Jose, CA) for cropping and labelling. The images were then converted into a single Microsoft JPEG using MS Word 2003 (Microsoft, Seattle, WA) for importing into the manuscript as a single picture.
Photographs of the feet and computed tomographs of the chest of a patient treated with IL-12 and liposomal doxorubicin. Images in panels A and C were taken at baseline; those in panels B and D were taken at 22 months of treatment, the time of complete response. This patient was docu-mented to have a partial response at 4 weeks. Panels A and B were taken by NIH Patient Photographic Services using Nikon Digital SLR cameras (D1 and D1X respectively). Computed tomography scans were acquired from the NIH Clinical Center Diagnostic Radiology Department using the Kodak Carestream PACS web-based program. Images were imported into Adobe Photoshop v7 (Adobe Systems, San Jose, CA) for cropping and labelling. The images were then converted into a single Microsoft JPEG using MS Word 2003 (Microsoft, Seattle, WA) for importing into the manuscript as a single picture.
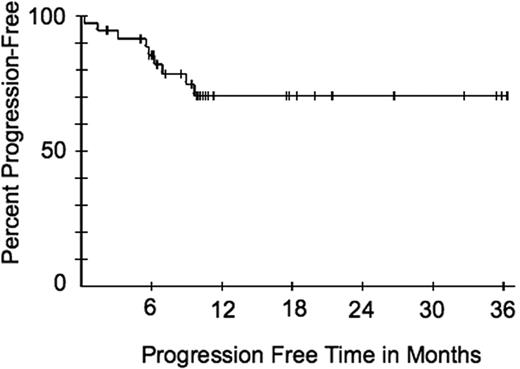
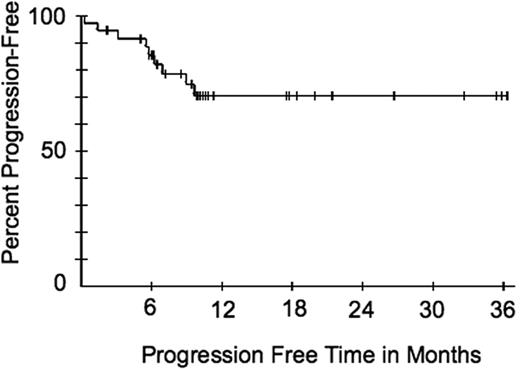
Median potential follow-up time was 46.9 months and median progression was not yet reached; the progression-free survival curve plateaus at 70.4% after 9.7 months (Figure 2). Five patients had disease progression after achieving PR. One additional patient did not have tumor progression, but was removed from study for psychologic distress caused by increasing KS lesions; the data were censored at this time point. No patient with CR had recurrent KS.
Progression-free survival of all patients. Time includes both time that they were being treated and a period of observation after treatment.
Progression-free survival of all patients. Time includes both time that they were being treated and a period of observation after treatment.
Six patients died: 3 from progressive KS that was also unresponsive to other therapy after removal from study; 2 of infection related to KS; and 1 for unknown reasons apparently unrelated to HIV disease or therapy. Several days after being assessed as having minimal residual KS, normal laboratory values, high CD4 cells, and undetectable HIV viral load, he presented to an emergency room and died suddenly of unknown causes. Six patients were removed from the study prior to completion of induction therapy: 3 for disease progression, 1 for IL-12–related hepatic toxicity, and 2 for infectious complications thought related to advanced AIDS-KS.
Although the study was not designed to segregate responses between induction and maintenance phases, we examined the data for evidence of IL-12 activity during maintenance. KS tumor burden was assessed at the end of the combination regimen, and this was considered a new baseline. Twenty-nine patients received at least one injection of IL-12 maintenance therapy. 2 of these had CR at the time they entered maintenance therapy and maintained that CR. Of the remaining 27 patients, 15 patients (56%) met the criteria for PR or CR during maintenance as assessed against the new baseline, including one who did not respond during induction but then had a PCR after 104 weeks of IL-12 alone. Eight patients (29.6%) had PD compared with the new baseline. Six subsequently developed PD compared with protocol entry.
HIV and T-cell dynamics
There were no clear associations between baseline CD4 cell count and response. The response rate (CR + PR vs SD + PD) was not statistically different when patients were stratified by 50 or less, 50 to 150, or more than 150 CD4 cells/mm3 (P2 = 0.17). In addition, there were no apparent major differences in T-cell dynamics among patients when comparisons were made on the basis of response (Table 2). There was no association between tumor response (PD + SD vs PR vs CR) and actual change in CD4 count from entry to first response (P = .08). HIV viral loads were relatively stable. Those with progressive disease had a median change of 0.07 (range: −.360 to 1.31) log10, while those who responded had a median change of 0 (range: −3.26 to 1.47) log10. Response was not clearly associated with TS stage, although all with disease progression without first having a PR had a poor TS status (Table 2). Finally, recent HAART initiation did not appear to favor tumor response; among the 30 patients who had a major response, time on HAART prior to entry was fewer than 6 months in 30%, between 6 and 12 months in 10%, and greater than 12 months in 60%. The median time on HAART for all patients enrolled was 18.7 (range: 0-100) months.
Serum levels of IL-12, IFN-γ, and IP-10
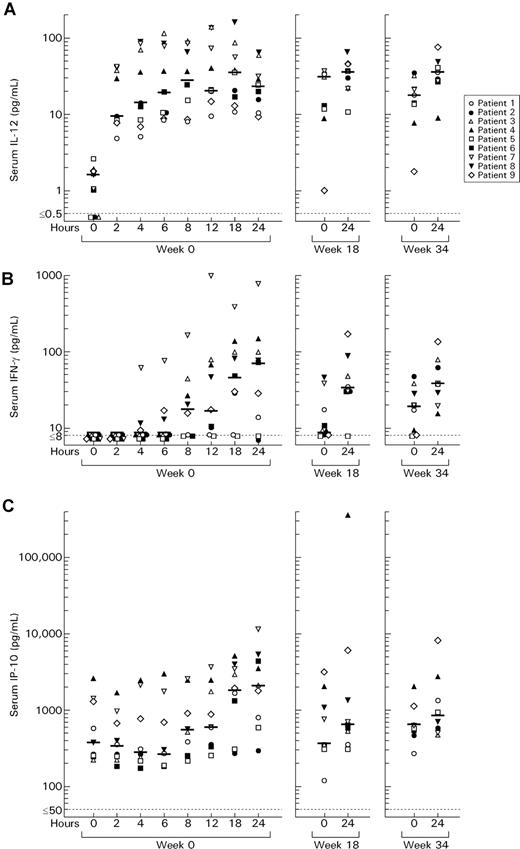
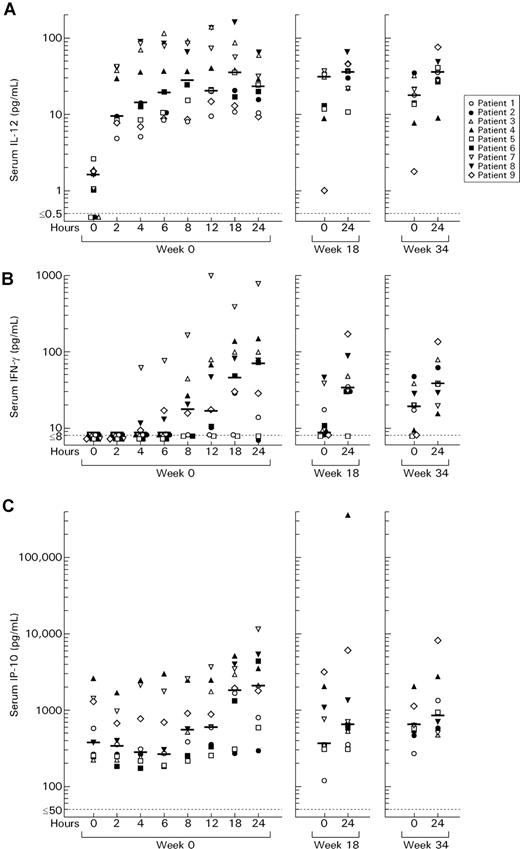
In the 9 patients studied, serum levels of IL-12 increased starting 2 hours after administration of the first subcutaneous 300-ng/kg dose of IL-12 and remained elevated for at least 24 hours (Figure 3). Subsequent increases in IFN-γ and IP-10 were seen beginning at 8 to 12 hours. Levels of IL-12 (P2 = 0.0039), IFN-γ (P2 = 0.016), and IP-10 (P2 = 0.0039) were still elevated compared with baseline at 24 hours. These cytokines were again measured just before (0 hour) and 24 hours after a dose of IL-12 at the completion of 18 weeks of combination therapy and then after 16 weeks of IL-12 alone (protocol week 34) (Figure 3). The 0-hour levels of IL-12 were substantially higher at weeks 18 and 34 than at week 0. In addition, at weeks 18 and 34, increases above the original baseline were seen for IFN-γ (week 18: P2 = 0.016; week 34: P2 = 0.0078) and IP-10 (week 18: P2 = 0.016; week 34: P2 = 0.055) 24 hours following IL-12 administration, suggesting ongoing cytokine responses.
Serum interleukin-12 (IL-12), interferon-gamma (IFN-γ), and inducible protein-10 (IP-10) before and after the first dose of subcutaneous 300 ng/kg IL-12, a dose at week 18, and a dose at week 34. Patients 1 to 3 at week 18 and patients 7 and 8 at week 34 received 300 ng/kg IL-12; the others received 500 ng/kg.
Serum interleukin-12 (IL-12), interferon-gamma (IFN-γ), and inducible protein-10 (IP-10) before and after the first dose of subcutaneous 300 ng/kg IL-12, a dose at week 18, and a dose at week 34. Patients 1 to 3 at week 18 and patients 7 and 8 at week 34 received 300 ng/kg IL-12; the others received 500 ng/kg.
Toxicity
Patients received therapy for up to 36 months (median: 8.4 months). During induction, there were no dose reductions, but 9% of cycles had treatment delays. Ten patients had dose reductions to 250 ng/kg or 300 ng/kg on maintenance. Principal laboratory toxicities are listed in Table 3. Thirteen patients, all of whom had a major response, met criteria for study removal due to adverse events: 4 for transaminitis; 2 each for hyperamylasemia or infection; 3 for neurologic or neuropsychiatric toxicity; and 1 each for hyperlipidemia or pain. Neuropsychiatric toxicity included 1 case of grade 4 depression, 1 cerebrovascular accident, and 1 transient ischemic attack. Both vascular events occurred in the maintenance period; one had a previous cerebrovascular attack, and the other had predisposing factors including dyslipidemia and diabetes. Neither suffered long-term consequences. Six patients with CR, PR, or SD were electively removed after a number of months on IL-12 for grade 1 to 2 chronic toxicities such as arthralgias or leukopenia because of concerns that the toxicity was not justified in a situation where further benefit was unclear.
Discussion
In this trial of pegylated liposomal doxorubicin and IL-12 administered to patients with KS requiring chemotherapy, we showed that the therapy was generally well tolerated and that high response rates were obtained. It was particularly noteworthy that 25% of the patients had a CR and that 56% of the patients with evaluable KS when they went on maintenance had a major response (PR or CR) when measured against the new baseline.
Our previous study of IL-12 provided evidence for activity in AIDS-KS.33 However, patients often responded slowly, limiting utility in advanced disease. To address this issue, we explored a regimen in which IL-12 was combined with cytotoxic chemotherapy for several months and then administered alone. Evidence from a number of drug combinations and models suggests that combining antiangiogenic and cytotoxic agents can enhance the activity of either agent.34,35,37,45 The potential synergy is not definitively addressed by this study design, but the favorable outcome supports further study.
KS can respond to HAART, especially when it is initiated,9,–11 and HAART was envisioned as part of the treatment regimen. Moreover, patients could be entered with KS requiring urgent therapy even if KS had not increased on 4 weeks of HAART, and 3 such patients were entered. It is thus worth considering whether the results may have simply reflected HAART. In this regard, the patients here are very dissimilar from those who typically respond to HAART alone,9,13 and the majority had increasing KS on long-term HAART. In addition, one mechanism by which HAART is believed to affect KS is through CD4 count increases,11,46 but as seen in Table 2, there was no obvious relationship between CD4 cell or HIV viral load changes and KS responses. Moreover, HAART duration did not predict response. Thus, it is very unlikely that HAART alone was responsible for KS responses.
A related question is the contribution of IL-12. KS response rates approaching the 83% seen in this study have been observed in studies of HAART plus cytotoxic chemotherapy.12,16 It is noteworthy that the median time to response here was 2 cycles of combined therapy. By contrast, more cycles appeared to be required to achieve sustained responses in previous studies of HAART plus cytotoxic chemotherapy.12,16 In addition, unlike the present study, those reports did not specifically target advanced KS. Additional support for the role of IL-12 comes from the observation that a substantial number of responses occurred in the maintenance phase, when all patients were off pegylated liposomal doxorubicin and had been on HAART for at least 18 weeks. Nonetheless, definitive assessment of the effect of IL-12 in this setting will require randomized trials.
Previous studies have reported that IFN-γ induction by IL-12 is blunted after repeated doses47,48 and that HIV patients treated with IL-12 for up to 4 weeks had no increases in phytohemagglutinin-inducible IFN-γ.49 The current study revealed that nadir IL-12 levels were increased at weeks 18 and 34. In addition, IL-12–induced increases in IFN-γ levels at weeks 18 and 34 were only slightly less than that observed after the first dose. This provides pharmacokinetic plausibility for sustained IL-12 activity throughout the trial. By contrast, the increases in IP-10 were substantially fewer at weeks 18 and 34. The significance of this observation is unclear. It is possible that some serum IP-10 is produced by KS spindle cells, and that the decrease reflects the reduction in tumor burden.
The current study used initial doses of 300 ng/kg IL-12, and then 500 ng/kg. Although generally well tolerated, occasional serious toxicities were observed. In particular, 2 patients had reversible cerebrovascular events. Although both had predisposing features, IL-12 cannot be ruled out as a contributing factor. In addition, consistent with our initial study of IL-12,33 some patients appeared to manifest neuropsychiatric toxicity related to IL-12 that manifests as depression, confusion, and possibly psychosis. While the studied doses used seemed reasonable, it may be worthwhile considering the 300-ng/kg dose for maintenance in future studies.
There are several mechanisms by which IL-12 may have activity in KS, including the enhancement of a specific immune response against KSHV or tumor antigens; induction of an antiangiogenic effect, which is mediated through IP-10 and MIG; and suppression of signaling by the KSHV-encoded GPCR which is mediated through IP-10. The latter 2 mechanisms are mediated through induction of IFN-γ by IL-12, and they would also be expected to be induced by administration of IFN-γ. While there are reports that IFN-γ has little or no activity in KS,50,51 these studies performed in the era before anti-HIV therapy used relatively crude biologic preparations of IFN-γ, and it may be of interest to re-explore IFN-γ for possible activity in KS. In addition, these data support the concept that a combination of antiangiogenic therapy and pegylated liposomal doxorubicin is well tolerated and highly effective in the treatment of advanced AIDS-related KS and suggest that readily applicable clinical advances could potentially be developed using other antiangiogenic agents.
In summary, this study provided evidence that combination IL-12 and pegylated liposomal doxorubicin, followed by maintenance IL-12 alone in patients with AIDS-KS receiving HAART, was well tolerated and associated with a high response rate, rapid responses, and sustained anti-KS activity. While this study, plus the previous trial of IL-12, suggests that this cytokine has an anti-KS effect, a randomized trial will be needed to assess this activity. The present study suggests that it may be worth studying IL-12 activity in the context of HAART and liposomal anthracyclines.
The publisher or recipient acknowledges the right of the US government to retain a nonexclusive, royalty-free license in and to any copyright covering this article.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
This work was supported by the Intramural Research Program of the NIH, NCI.
The authors thank the patients who volunteered for this study; Dr James Zwiebel and other members of CTEP; Dr Matthew Sherman of Genetics Institute; Capt Florentino Merced-Galindez, Florence Jarfoi, and Sofia Lukasiak of the HIV and AIDS Malignancy Branch; Randy Stevens and Michael Baseler of Science Applications International Corporation; the medical staff of the Medical Oncology Branch of the NCI, and the nursing, pharmacy, social work, and medical staff of the NIH Clinical Center.
National Institutes of Health
Authorship
Contribution: R.F.L. conceived and designed the study, cared for patients, collected and assembled data, analyzed and interpreted data, and wrote the paper; K.A. and K.M.W. cared for patients, collected and assembled data, and analyzed and interpreted data; P.K. and A.T.C. cared for patients, and collected and assembled data; J.M.P. conceived and designed the study, and analyzed and interpreted data; E.R.-C. collected and assembled data; V.W. collected and assembled data, and analyzed and interpreted data; S.P. analyzed and interpreted data, reviewed histologic material, and confirmed diagnoses; S.M.S. conceived and designed the study, analyzed and interpreted data, and wrote the paper; R.Y. conceived and designed the study, cared for patients, collected and assembled data, analyzed and interpreted data, and wrote the paper. All coauthors approved the paper.
Conflict-of-interest disclosure: Several of the coauthors (R.F.L., J.M.P., K.M.W., and R.Y.) are listed as coinventors on US and/or foreign patents related to the use of IL-12 to treat Kaposi sarcoma, and one of the authors (R.Y.) is listed as a coinventor on one or more US and/or foreign patents involving dideoxycytidine (zalcitabine), dideoxyinosine (didanosine), zidovudine, and/or acyclovir as treatments for HIV infection. These inventions were made as full-time employees of the US government under 45 Code of Federal Regulations Part 7. All rights, title, and interest to these patents have or should by law be assigned to the US government. The government conveys a portion of the royalties it receives to its employee-inventors under the Federal Technology Transfer Act of 1986 (Public law 99-502).
Correspondence: Robert Yarchoan, Bldg 10, Rm 10N106, MSC 1868 NIH, Bethesda, MD 20892-1868; e-mail:yarchoan@helix.nih.gov.