To the editor:
The fms-related tyrosine kinase-3 internal tandem duplication (Flt3-ITD) confers growth-factor independence on acute myeloid leukemia (AML) cells and is associated with an unfavorable prognosis. Signal transducer and activator of transcription 5 (STAT5) is a downstream mediator of Flt3 signaling in AML; however, flow cytometry for phosphorylated STAT5 (pSTAT5) did not show specific association with Flt3-ITD.1 Since a recent study published in Blood by Harir et al2 reported pSTAT5 in the cytoplasm of leukemic samples, we wanted to determine whether this localization to the cytoplasm might be influenced by Flt3-ITD status and whether nuclear pSTAT5 would be a more informative end point. Using a routine immunohistochemistry technique that we previously reported for detecting activated STAT5 in chronic myeloproliferative disease,3 we have now analyzed pSTAT5 staining in myeloid blasts in bone marrow biopsies from patients with AML.
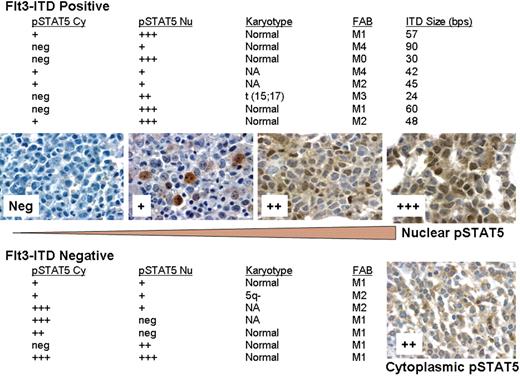
STAT5 tyrosine phosphorylation was detected immunohistochemically with the anti-phosphoSTAT5a/b (Y694/99) mouse monoclonal antibody (AX1; Advantex Bioreagents) as described by others in breast cancer.4 In our study, we analyzed a total of 40 patients with AML for nuclear versus cytoplasmic staining of pSTAT5 using the AX1 antibody. Total pSTAT5 was seen in 15 of 40 patients, while the Flt3-ITD was detected in 8 (20%) patients. Interestingly, as shown in Figure 1, Flt3-ITD+ samples had predominant nuclear pSTAT5, as would be expected from increased transcriptional activation. However, the Flt3-ITD− samples showed some nuclear staining, but had much more prominent cytoplasmic pSTAT5 staining with significant variation between groups (Kruskal-Wallis test; P = .03). A total of 2 patients showed predominant cytoplasmic staining and no nuclear staining. The distribution of cytoplasmic staining was significantly different between Flt3-ITD+ and Flt3-ITD− patients (Mann-Whitney test; P = .04).
Relationship between Flt3-ITD status and distribution of phospho-STAT5 between nucleus and cytoplasm. The relative staining intensity for pSTAT5 was determined by manually scoring slides using a scale of 0 (neg) to 3 (+++) obtained from fixed bone marrow core biopsy of newly diagnosed patients with AML. Those scored as neg did not have detectable pSTAT5, and those scored as 3 had the highest intensity of staining. Shown are examples of patient samples with staining for nuclear pSTAT5 at each level. A patient sample showing predominant cytoplasmic pSTAT5 (++) is shown for comparison. Flt3-ITD status was determined by PCR on marrow clot sections using capillary electrophoresis. pSTAT5 Cy indicates cytoplasmic; pSTAT5 Nu, nuclear; and NA, not available. Slides were viewed with an Olympus BX50 microscope (Olympus, Center Valley, PA) using a 50×/0.90 oil-immersion objective lens and pSTAT5 immunostains. Images were acquired with an Insight 11.2 color mosaic camera (Diagnostic Instruments, Sterling Heights, MI) and processed with Spot Basic 4.1.3 (Diagnostic Instruments) and Adobe Photoshop elements 2.0 (Adobe, San Jose, CA) software.
Relationship between Flt3-ITD status and distribution of phospho-STAT5 between nucleus and cytoplasm. The relative staining intensity for pSTAT5 was determined by manually scoring slides using a scale of 0 (neg) to 3 (+++) obtained from fixed bone marrow core biopsy of newly diagnosed patients with AML. Those scored as neg did not have detectable pSTAT5, and those scored as 3 had the highest intensity of staining. Shown are examples of patient samples with staining for nuclear pSTAT5 at each level. A patient sample showing predominant cytoplasmic pSTAT5 (++) is shown for comparison. Flt3-ITD status was determined by PCR on marrow clot sections using capillary electrophoresis. pSTAT5 Cy indicates cytoplasmic; pSTAT5 Nu, nuclear; and NA, not available. Slides were viewed with an Olympus BX50 microscope (Olympus, Center Valley, PA) using a 50×/0.90 oil-immersion objective lens and pSTAT5 immunostains. Images were acquired with an Insight 11.2 color mosaic camera (Diagnostic Instruments, Sterling Heights, MI) and processed with Spot Basic 4.1.3 (Diagnostic Instruments) and Adobe Photoshop elements 2.0 (Adobe, San Jose, CA) software.
The study by Harir et al2 demonstrated in mouse and human samples that STAT5 may potentially have cytoplasmic functions that promote PI3-kinase activation of Akt. Our data confirm the cytoplasmic localization of pSTAT5 in a larger set of patient samples and further suggests that this localization is modulated by the presence of Flt3-ITD. This is an important finding since it suggests that competing biological mechanisms are responsible for the nuclear versus cytoplasmic distribution of pSTAT5 in AML. Cytoplasmic pSTAT5 may not be directed efficiently to the nucleus in the absence of essential interactions provided by the Flt3-ITD scaffold. Alternatively, the hyperphosphorylation of STAT5 mediated by Flt3-ITD may override other mechanisms that may sequester pSTAT5 in the cytoplasm.5 This finding could be analogous to the requirement for JAK2V617F to interact with type I cytokine receptors to promote STAT5 transcriptional activation and cytokine-independent growth.6 Finally, our data also demonstrate that detection of nuclear, but not total pSTAT5 could be the most meaningful test for rapidly identifying patients who may respond to Flt3 or other tyrosine kinase inhibitors.
Authorship
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Eric D. Hsi, Department of Clinical Pathology and Laboratory Medicine, Taussig Cancer Center, The Cleveland Clinic Foundation, Case Comprehensive Cancer Center, Cleveland, OH 44195; e-mail: hsie@ccf.org.