Abstract
Primary cutaneous B-cell lymphomas (CBCL) represent approximately 20% to 25% of all primary cutaneous lymphomas. With the advent of the World Health Organization-European Organization for Research and Treatment of Cancer (EORTC) Consensus Classification for Cutaneous Lymphomas in 2005, uniform terminology and classification for this rare group of neoplasms were introduced. However, staging procedures and treatment strategies still vary between different cutaneous lymphoma centers, which may be because consensus recommendations for the management of CBCL have never been published. Based on an extensive literature search and discussions within the EORTC Cutaneous Lymphoma Group and the International Society for Cutaneous Lymphomas, the present report aims to provide uniform recommendations for the management of the 3 main groups of CBCL. Because no systematic reviews or (randomized) controlled trials were available, these recommendations are mainly based on retrospective studies and small cohort studies. Despite these limitations, there was consensus among the members of the multidisciplinary expert panel that these recommendations reflect the state-of-the-art management as currently practiced in major cutaneous lymphoma centers. They may therefore contribute to uniform staging and treatment and form the basis for future clinical trials in patients with a CBCL.
Introduction
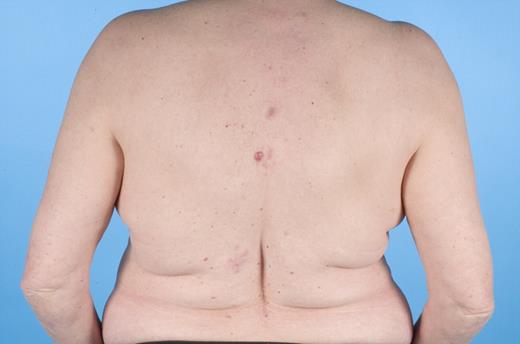
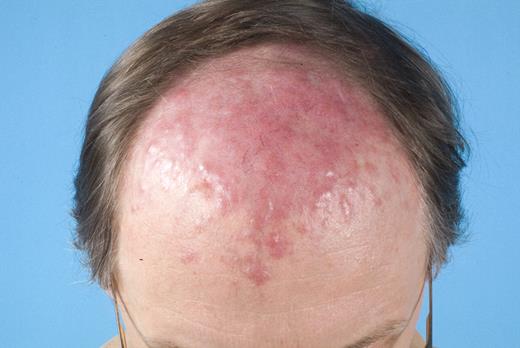
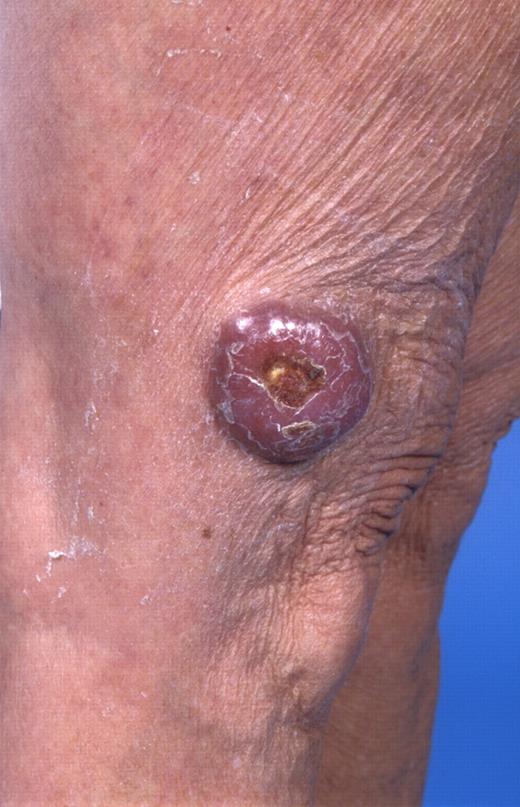
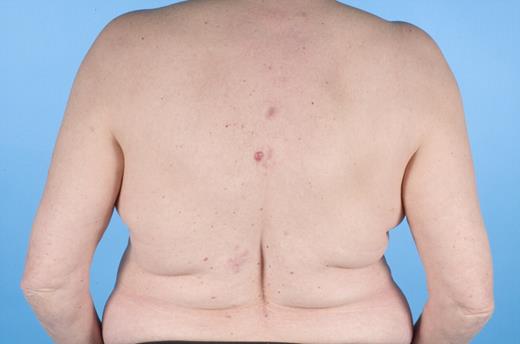
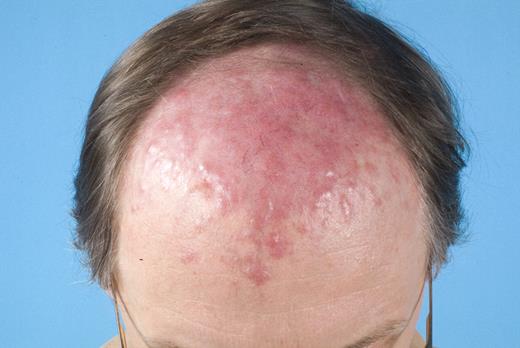
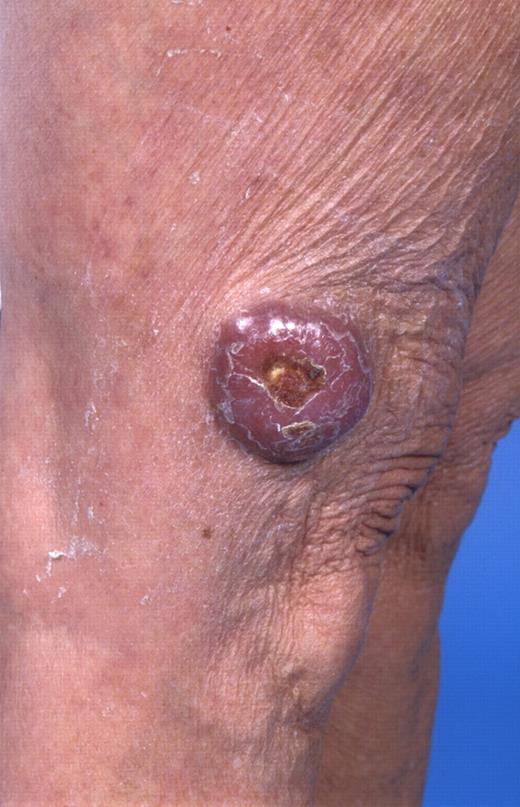
In the last 2 decades, it has become clear that some subtypes of B-cell non-Hodgkin lymphoma can exclusively present in the skin. These primary cutaneous B-cell lymphomas (CBCLs) are much less common than primary cutaneous T-cell lymphomas (CTCLs) and represent approximately 20% to 25% of all primary cutaneous lymphomas.1 For many years, there has been confusion regarding the terminology and classification of CBCL (Table 1), which may have come to an end with the publication of the new World Health Organization-European Organisation for Research and Treatment of Cancer (WHO-EORTC) classification for cutaneous lymphomas.1 This classification distinguishes 3 main types of CBCL: primary cutaneous marginal zone B-cell lymphoma (PCMZL), primary cutaneous follicle center lymphoma (PCFCL), and primary cutaneous diffuse large B-cell lymphoma, leg type (PCLBCL, LT). The characteristic clinical, histologic, and immunophenotypic features of these 3 subgroups are presented in Table 1 and characteristic clinical presentations in Figures 1, 2, and 3. In the forthcoming update of the WHO classification (to be published in the WHO Blue Book Series in June 2008), PCFCL and PCLBCL, LT will be included as separate entities, whereas PCMZL will be included in the broader category of extranodal marginal zone B-cell lymphomas.
It is generally recognized that PCMZL and PCFCL are indolent types of CBCL, which should not be treated primarily with systemic chemotherapy. Nonetheless, many patients with a PCFCL, in particular patients with a diffuse infiltration of large cleaved cells that were previously classified as diffuse large B-cell lymphoma (DLBCL) in the WHO 2001 classification,2 are often still treated unnecessarily with systemic chemotherapy. Moreover, staging procedures and treatment strategies vary between different cutaneous lymphoma centers, which may be because, in contrast to CTCL,3,4 consensus guidelines for the management of CBCL have never been published.
Based on an extensive literature search and discussions at meetings of the EORTC Cutaneous Lymphoma Group (EORTC-CLG) and the International Society for Cutaneous Lymphoma (ISCL), the present report aims to provide uniform recommendations for the staging and treatment of the 3 main types of CBCL. Rare types of CBCL that occasionally present in the skin without detectable extracutaneous disease (eg, intravascular large B-cell lymphoma and plasmablastic or lymphoblastic lymphoma) will not be discussed in this review.
Development process of recommendations
At a meeting of the ISCL Group in March 2006, representatives of the Dutch Cutaneous Lymphoma Group were asked to formulate a proposal for recommendations for the management of CBCL. Based on the results of an extensive literature search, concept recommendations were made, which were discussed and agreed on with minor modifications at an ISCL meeting in Buenos Aires in September 2007. The final text was agreed on by an expert group of dermatologists, hematologists/oncologists, and radiation oncologists, selected from the membership of the EORTC-CLG and ISCL group for their expertise in research and clinical practice of CBCL.
Review of the literature
A search of PubMed, EMBASE, and Web of Science was conducted to identify relevant studies (final update, December 7, 2007). Relevant articles were identified by combining terms and phrases related to cutaneous or skin, B-cell lymphoma(s), and treatment or therapy. Relevant articles were selected and reviewed by 2 reviewers (N.J.S., R.W.).
Articles were selected for inclusion if they described treatment results in patients with CBCL or if they contained a table with clinical and follow-up data of CBCL patients, containing at least information on initial treatment and treatment results. Because of the low incidence of CBCL and the fact that (randomized) controlled trials and systematic reviews were not available, case reports and small retrospective cohort studies were also included. Papers describing other entities besides the 3 main groups of primary cutaneous B-cell lymphoma (PCMZL, PCFCL, and PCLBCL, LT), papers without relevance to treatment, reviews merely summarizing existing literature, meeting abstracts, and most non-English literature were excluded. Many other papers were of limited value because the type of CBCL was not specified or treatment results were not reported separately for different diagnostic subgroups. If the same cohort was described in several subsequent papers, only the last publication was used in our analysis. From the original 318 retrieved references, 92 were selected for further analysis. These included 32 case reports (35%) and 60 case series or retrospective cohort studies, including 2 to 5 patients (20 papers; 22%), 6 to 10 patients (9 papers; 10%), 11 to 50 patients (23 papers; 25%), or more than 50 patients (8 studies; 9%).
Papers were scored according to the levels of evidence as described by the Oxford Center for Evidence-Based Medicine (available from: http://www.cebm.net/levels_of_evidence.asp. Accessed 3 December 2007). However, because only case reports, case series, and retrospective cohorts were available and, as a result, all evidence was scored as level 4 or 5 and grades of recommendation were mostly of the D category, levels of evidence are not further discussed.
Diagnosis and staging of cutaneous B-cell lymphomas
Diagnosis
When a cutaneous B-cell lymphoma is clinically suspected, adequate histologic and immunohistochemical studies are required to confirm or discard the diagnosis. A definite diagnosis can only be made on a representative biopsy of adequate length and diameter (preferably an excisional biopsy; but in case of punch biopsies, the diameter should be at least 4 mm whenever possible). Besides considering the morphology of the neoplastic B-cell population and the growth pattern of the malignant infiltrate, immunohistochemical studies are required to make a definite diagnosis. Because a detailed description of markers used in the differential diagnosis of cutaneous B-cell infiltrates is beyond the scope of this review, only a short list of relevant immunostainings is provided:
CD3, CD20, and/or CD79a for confirmation of B-cell lineage and assessment of the amount of admixed reactive T cells;
Surface and cytoplasmic immunoglobulins (sIg; cIg) on frozen and paraffin sections, respectively, to confirm B-cell lineage and to detect monotypic Ig expression;
CD35 or CD21 to visualize reactive follicles or (remnants of) dendritic networks;
Ki-67 to determine the proliferative fraction and to aid in the differentiation between neoplastic and reactive follicles5 ;
Bcl-2, Bcl-6, CD10, MUM-1, and FOXP1 may aid in the distinction between different types of CBCL, pseudo B-cell lymphoma, and secondary cutaneous B-cell lymphoma (Table 1).6-9 Strong expression of Bcl-2, Bcl-6, and CD10 in follicular structures should always raise suspicion of a systemic follicular lymphoma with secondary skin involvement, and the presence of the interchromosomal translocation t(14;18) should be examined.
In selected cases, use of CD5 and cyclin D1 is useful to differentiate PCMZL (CD5−, cyclin D1−) from mantle cell lymphomas (CD5+, cyclin D1+) and skin localizations of B-cell lymphocytic leukemia (CD5+, cyclin D1−).
Although successful application of flow cytometry in the diagnosis of CBCL has been reported,10 it is not widely used and cannot be considered as a substitute for immunohistochemistry. Disadvantages of this approach are the difficulties to obtain sufficient viable single-cell suspensions because of the vulnerability of the cutaneous B cells, the lack of architectural information, and the need of additional fresh tissue material. Demonstration of clonal IgH gene rearrangements, using the standard BIOMED-2 primers and protocol,11-14 may be a useful aid in the diagnosis of CBCL, but the results should always be considered in conjunction with clinical, histologic, and immunohistochemical data.
Staging procedures
ISCL/EORTC recommendations for staging in cutaneous lymphomas other than mycosis fungoides/Sezary syndrome have recently been published.15 Proper clinical staging evaluation should begin with a complete history and review of systems (eg, B symptoms, organ-specific signs) and a thorough physical examination. Laboratory studies include a complete blood cell count with differential and a comprehensive blood chemistry measurement, including lactate dehydrogenase, and in selected cases, serum electrophoresis to exclude a monoclonal gammopathy and/or flow cytometry on peripheral blood. Because an association between Borrelia burgdorferi infection has been reported in a significant minority of European cases of PCMZL, but not in Asian cases or cases from the United States,16-19 in European areas with endemic B burgdorferi infection, the presence of B burgdorferi should be investigated by serology and polymerase chain reaction techniques on skin biopsy specimens. Adequate imaging studies (contrast-enhanced CT scan with or without positron emission tomography, or whole-body integrated positron emission tomography/CT15 ) should be performed of at least chest, abdomen, and pelvis, and in cases of skin lesions in the head and neck area, of the neck as well. In the recently published ISCL/EORTC guidelines, bone marrow biopsy and aspirate are required in PCLBCL, LT, but considered optional in indolent CBCL (PCMZL and PCFCL).15
However, a recent study demonstrated bone marrow involvement in 22 of 193 (11%) patients with a follicle center lymphoma (FCL) first presenting in the skin, compared with 2 of 82 patients with a marginal zone lymphoma.20 In 9 of these 22 FCL patients, this was the only evidence of extracutaneous disease. The 5-year overall and disease-specific survivals of these 9 patients were 44% and 63%, respectively, compared with 84% and 95%, respectively, in 157 patients without extracutaneous disease. These results indicate that bone marrow examination should be considered an essential part of staging procedures in patients with a FCL first presenting in the skin and that, from a clinical point of view, bone marrow examination appears to have limited value in patients with a marginal zone lymphoma presenting in the skin. Because currently there is no uniform consensus on whether bone marrow biopsy is required in FCLs presenting in the skin, the clinician is advised to follow the standard of care of his or her regional practice.
Recommendations for the treatment of primary cutaneous B-cell lymphomas
The different treatment options emerging from our literature search are summarized in Table 2. Therapy options most often reported included radiotherapy, systemic chemotherapy, rituximab, and excision. For the other treatment modalities, relative few studies were available for review. Table 2 also includes therapies that are still under investigation as well as some therapies, for which only anecdotal reports are available. Most of these will not further be discussed. Cumulative complete remission (CR) and relapse rates for the different treatment modalities used in patients with PCMZL, PCFCL, and PCLBCL, LT were synthesized from the reviewed literature and are provided in Table 3. Data on relapse-free survival or progression-free survival are hardly available and will therefore not be discussed. In the following paragraphs, the results of these different treatment options are described in more detail, and recommendations for the management of these CBCL are formulated.
Primary cutaneous marginal zone B-cell lymphoma
Radiotherapy
In the literature, a total of 132 PCMZL patients are described who were treated with radiotherapy.21-30 In nearly all cases (130 of 132; 99%), a CR after initial therapy was reached. Sixty patients (46%) showed one or more relapses, which were mostly confined to the skin. Extracutaneous progression was reported in only 3 of 132 patients, one of whom died of lymphoma.26 Reported cumulative doses per irradiation field were mostly between 30 and 45 Gy. However, doses as low as 10 Gy and as high as 50 Gy have been reported.22,29 Most studies included a margin of clinically normal skin in the radiation field, ranging from 1 to more than 5 cm, partly depending on the affected body site.
Excision
In daily practice, surgical excision is a first choice of treatment in patients presenting with one or few small skin lesions. In the literature, 75 patients treated with surgical excision have been reported.21,25,29-32 A CR was reached in all but one case.30 However, 32 patients (43%) developed new skin lesions. Information whether these represent local recurrences or true relapses (new skin lesions at other skin sites) is not provided. In addition, no information was given regarding the margins of resection or whether dissemination to extracutaneous sites had occurred.
Interferon-alpha intralesional
Only one paper describes the use of interferon (IFN)–α in PCMZL.33 In contrast to its systemic use in CTCL, in this study 8 patients received intralesional injections of 3 million IU, 3 times per week. All patients reached a CR after a median of 8 weeks (range, 3-20 weeks). Two patients experienced a local recurrence, which went again in CR after treatment with IFN-α. No extracutaneous relapses were reported and side effects were generally mild.
Rituximab intralesional or intravenous
Five reports describe the use of rituximab in PCMZL.34-38 In 3 patients, it was administered systemically (375 mg/m2 intravenously once weekly, for 4 or 8 weeks; 2 of these 3 patients had multifocal disease),35,36,38 and in 9 patients intralesionally (5-30 mg once or 3 times per week; 8 of 9 patients had ≤ 4 skin lesions).34,36,37 Two of 3 (67%) systemically treated patients and 8 of 9 (89%) intralesionally treated patients reached a CR, with the remaining patients reaching a partial remission (total response rates, 100%). Relapse rates were 50% (1 of 2) and 62% (5 of 8), respectively, and no extracutaneous relapses were reported.
Single-agent and combination chemotherapy
With respect to single-agent chemotherapy, 4 reports describe the results of chlorambucil, a nitrogen mustard derivative, which has been used for many years in the treatment of low-grade systemic non-Hodgkin lymphomas and leukemias. There was a total number of 14 patients with a PCMZL in these studies, all with multifocal skin lesions.23,25,31,39 The response rate was 100% with a CR in 9 patients (64%) and a partial remission in the other 5 patients. Of the 9 patients with a CR, 3 patients showed relapses (33%), one of whom also with extracutaneous localizations. Reports on other agents used in indolent B-cell non-Hodgkin lymphoma, such as fludarabine and 2-chlorodeoxyadenosine, are lacking.
Antibiotics
Analogous to the treatment of gastric mucosa-associated lymphoid tissue lymphomas with antibiotic therapy to eradicate Helicobacter pylori,41-43 recent reviews suggest that PCMZL associated with B burgdorferi infection should first be treated with antibiotics before more aggressive therapies are used.44 However, the efficacy of antibiotic treatment in B burgdorferi–associated PCMZL is poorly documented.29,31,45-48 Six of 14 (43%) reported patients achieved CR after various antibiotic regimens. Data on 8 patients for whom antibiotic treatment was specified suggest that systemic treatment with cephalosporins is superior to oral treatment with high-dose tetracyclins.
Recommendations
Patients presenting with solitary or few scattered skin lesions can best be treated with local radiotherapy (20-36 Gy) or excision (Table 4). For patients presenting with disseminated skin lesions, several management strategies can be considered. A strategy not supported by data from literature, but generally practiced by the expert panel members as the management of choice, is a wait-and-see strategy, similar to what is often used for indolent noncutaneous B-cell lymphomas and leukemias. This strategy implies that patients are carefully followed and only symptomatic lesions are treated (“treat as needed” concept). Individual, symptomatic skin lesions can be treated with surgery, topical or intralesional steroids, or low-dose radiotherapy. Intralesional administration of IFN-α or rituximab may be interesting alternatives, but further studies are required.
In patients with very extensive skin lesions, chlorambucil is still often used in European centers, in particular in older patients and for a limited period of time (maximum, 3 months), but it is uncommonly used in the United States. Other single-agent or combination chemotherapy regimens appropriate for systemic low-grade B-cell lymphomas may be considered, but published results are not available. Alternatively, systemic rituximab can be considered. Multiagent chemotherapy is rarely indicated in this type of CBCL.
Cutaneous relapses do not signify a worse prognosis and can be treated in the same way as the initial skin lesions.
Theoretically, it is attractive to suggest that PCMZL associated with a B burgdorferi infection should first be treated with antibiotics before other therapies are used. However, although some CRs have been reported, in particular in patients treated with systemic cephalosporins, additional studies are required to establish which patients may benefit from antibiotic treatment and to assess which type, dose, and duration of antibiotic treatment should be recommended.
Primary cutaneous FCL
Radiotherapy
Already in 1951, Crosti described 7 cases of “reticulohistiocytoma of the dorsum” that were markedly sensitive to radiation therapy.49 This entity was later reclassified as PCFCL.50,51 Most recent reports on treatment of PCFCL describe local radiotherapy as the first choice of treatment because it is highly effective and has few side effects. In the literature, we found a total number of 460 PCFCL patients initially treated with radiation therapy.5,22,23,26,27,29,30,45,50,52-61 These were mostly patients with solitary or localized skin lesions, but some reports also include patients with multifocal skin disease, necessitating the use of multiple radiation fields.22,26,27,58 In one study specifically addressing such patients, it was found that radiotherapy of all visible skin lesions was equally effective as treatment with multiagent chemotherapy.40 A CR was reached in nearly all cases, irrespective of the extent of the disease (457 of 460; 99%). Three major studies on radiation therapy in PCFCL (combined including 147 patients; median follow-up, 52, 62, and 41 months, respectively) report a relapse rate of approximately 30%,22,26,27 which contrasts with the 76% in an Italian study (104 patients; median follow-up, 62 months).58 This latter group also reported in-field and marginal recurrences, which were not seen in the other studies. These differences might be because of the variance in techniques used and, most important, the margins of healthy-looking skin included in the radiation field (range, 0.5- > 5 cm). The cumulative dose per irradiation field was generally more than 30 Gy (range, 20-54 Gy).
Excision
A total of 93 patients were found in the literature that were initially treated with complete surgical excision.5,28-30,53-55,60 Most patients reached CR (91 of 93; 98%) and less than half of them (36 of 91; 40%) developed a skin relapse. No details are provided concerning excision margins or specific sites of local recurrence or relapse at distant skin sites.
Interferon alpha (IFN-α) intralesional
Intralesional treatment of PCFCL patients with IFN-α has been reported in only 7 cases, with dosages varying from 1 million IU, 3 times per week to 6 million IU, 3 times per week for larger lesions.46,62-64 All 7 patients reached a CR. Two patients experiencing a clinical relapse were again successfully treated with a second course of IFN-α.62 However, because the follow-up periods were rather short, no firm conclusions regarding relapse rate or long-term efficacy can be made.
Rituximab intralesional and intravenous
Both intralesional and systemic rituximab have been used in PCFCL. Ten of 12 patients treated intralesionally reached CR, and the other 2 patients had a partial remission.34,36,65-67 Dosages and treatment regimens varied between 10 and 30 mg per lesion per application for 2 or 3 times a week. Duration of treatment was more variable, ranging from one or 2 injections in total, to treatment for up to 6 months. Four of 10 patients with CR developed new skin lesions: 2 in the originally treated site and 2 at distant skin sites. No extracutaneous relapses were seen.
The 28 patients systemically treated with rituximab all received doses of 375 mg intravenously/m2 body surface area once weekly.34-36,68-77 Duration of treatment was mostly 4 weeks but ranged from 1 to 8 weeks. Twenty-one patients reached CR, 6 patients showed partial remission, and 1 patient76 had progressive disease but went into CR after 8 CHOP courses. In 4 of the 21 patients with CR, relapses were reported, and these were all confined to the skin.
Multiagent chemotherapy
Relatively few data exist in the literature concerning treatment of PCFCL patients with multiagent chemotherapy, although one would expect that many of these patients, in particular those with a diffuse growth pattern, classified as DLBCL according to the WHO classification, were treated in this way. Moreover, 2 of the largest studies could not be used for analysis because they describe mixed groups of patients based on older classification schemes.78,79 Cumulative data from 8 papers on 104 patients show a CR rate of 85% (88 of 104 patients) and a relapse rate of 48% (42 of 88 patients).28,30,40,52,53,56,80,81 These were mostly patients with disseminated cutaneous lesions or heavy tumor burden. Most patients had been treated with CHOP or CHOP-like courses, and only 5 patients with COP, which however was reported to be less effective.78,80
In DLBCL presenting with localized disease (Ann Arbor stage I-E), a combination of chemotherapy (3-4 cycles of CHOP) and involved field radiotherapy (IFRT) has been suggested to be superior to one of either modalities alone.79 Reports on this combined treatment in patients with PCFCL are few.28,52,54-56 Cumulative data on 7 patients show a CR in all patients, with a reported relapse in one of them.
Although a combination of rituximab and CHOP (R-CHOP) has become standard treatment for DLBCL for more than 5 years82,83 and many cases of PCFCL would have been classified as DLBCL using the WHO 2001 classification, only 2 patients with PCFCL who were treated with R-CHOP have been published thus far. One patient went into CR and had no relapse in the following 12 months.84 The second patient had only a PR after 6 cycles of R-CHOP. Further progression of the skin lesions was noted 1.5 years later, and single-agent treatment with rituximab resulted in a CR, which was ongoing for more than 19 months.70 A third patient, treated with a combination of cyclophosphamide and rituximab, showed a reduction of less than 30% of skin lesions.85
Recommendations
In patients presenting with solitary or localized skin lesions, radiation therapy with a dose of at least 30 Gy and a margin of clinically uninvolved skin of at least 1 to 1.5 cm, is the preferred mode of treatment (Table 4). Solitary lesions that are small and well-demarcated can be treated with surgical excision.
In patients with an indolent PCFCL presenting with few scattered lesions, both radiotherapy of all visible skin lesions as well as a wait-and-see policy with treatment of only symptomatic skin lesions, similar as recommended for PCMZL, can be considered for initial management. In patients with very extensive skin lesions, systemic rituximab is the first choice of treatment. Combination chemotherapy (R-COP; R-CHOP) should be considered only in exceptional cases, such as patients with progressive disease not responding to rituximab or patients developing extracutaneous disease.
Relapses occur in approximately 30% of patients, are mostly confined to the skin, and do not signify a worse prognosis. For the treatment of cutaneous relapses, a similar approach as for the initial skin lesions is recommended. In addition, in these cases, multiagent chemotherapy is rarely indicated.
Primary cutaneous diffuse large B-cell lymphoma, leg type
Radiotherapy
Radiotherapy is less effective in PCLBCL, LT than in the indolent CBCL. The CR rate in 101 reported cases was 88% (89 of 101 patients) and the relapse rate was 58% (52 of 89 patients), whereas in-field and marginal recurrences have also been reported.22,23,26,27,29,30,56,58,87 Extracutaneous progression was reported in approximately 30% of patients.
Intravenous rituximab
Treatment with intravenous rituximab as single-agent therapy (375 mg intravenously/m2 body surface area once weekly for 4-8 weeks) is reported in 13 patients, in 7 of them as second or third line treatment.35,76,77,88-92 A CR was obtained in only 5 of 13 patients, including 3 of 6 patients, who received rituximab as initial treatment. None of the 4 patients with CR of whom follow-up was reported had a relapse, but the duration of follow-up was short (median, 7 months).
Multiagent chemotherapy
Recent reviews suggest that PCLBCL, LT should be treated as systemic DLBCL with multiagent chemotherapy with or without rituximab.1,44 However, only 32 patients receiving CHOP(-like) (n = 29) or COP (n = 3) have been reported.29,30,40,45,56,80,81,93 Collectively, 26 of 32 patients (81%) reached a CR and 14 of 26 CR patients (54%) had a relapse. A combination of CHOP and IFRT was used in an additional 6 patients and resulted in a CR in 4 of them.28,45,94,95
One recent study describes 12 patients treated with various combinations of anthracycline-containing chemotherapies and rituximab. All but one patient reached a CR (92%), and only one patient relapsed (9%). The authors suggest that patients with PCLBCL, LT treated with various combinations of age-adapted, anthracycline-containing chemotherapies and rituximab have a better outcome than patients receiving other treatments; however, follow-up of the group treated with chemotherapy and rituximab was insufficient (mean follow-up period, < 2 years) to determine a statistically significant difference.96 In addition, another report described a CR in 1 of 2 patients treated with cyclophosphamide and rituximab.85
Recommendations
Because PCLBCL, LT patients have morphologic, phenotypical, and molecular genetic features as well as a clinical behavior similar to that of systemic DLBCL, they should be treated consistently (Table 4). Nowadays, R-CHOP with or without IFRT is considered as the first line of treatment in these lymphomas, but the efficacy of this approach in patients with PCLBCL, LT is still poorly documented. In case the condition of the patient does not allow such an aggressive treatment, local radiotherapy of all visible skin lesions or perhaps rituximab as single-agent therapy can be considered. Whether radiotherapy should be considered as first choice of treatment in patients presenting with a small solitary tumor is a matter of debate. To gain more knowledge on the most favorable treatment for this rare patient group with a poor prognosis, it is important to treat patients in the setting of controlled clinical trials.
As there are insufficient studies on relapsed PCLBCL, LT, it is recommended that these patients be treated as relapsed DLBCL, using local protocols.
Conclusions
Based on an extensive review of available literature and discussions within the EORTC and ISCL groups, the present report provides consensus recommendations for the management of CBCL. It should be emphasized that, because systematic reviews and (randomized) controlled trials were not available, the treatment recommendations are mainly based on retrospective studies and small cohort series. In addition, in most of these studies, information on relapse-free survival or progression-free survival was not included and in many studies follow-up was too short to draw conclusions on long-term efficacy. Despite these limitations, there was consensus among the members of the multidisciplinary expert panel that these recommendations reflect the state-of-the-art treatment of CBCL, as currently practiced in major cutaneous lymphoma centers. These recommendations may therefore contribute to uniform staging and treatment and may prevent, in particular, overtreatment of subgroups of CBCL patients. These recommendations may also guide the design of future clinical trials in CBCL, which are highly necessary and, in view of the rarity of these conditions, should be multicenter studies, preferably within the frame work of the EORTC Cutaneous Lymphoma Group and/or ISCL. Trials most often suggested by the expert panel members were: rituximab against current best practice (wait-and-see strategy with radiotherapy when needed) in both PCMZL and PCFCL, and radiotherapy versus R-CHOP + IFRT, in patients with PCLBCL, LT presenting with a solitary lesion. In addition, comparison between systemic and intralesional treatment with rituximab in PCFCL could be of interest because also in patients treated intralesionally a complete disappearance of B cells in the peripheral blood has been noted, indicating a systemic effect.34,65 In selected cases, intralesional rituximab might prove to be an equally effective, but much cheaper, alternative for systemic rituximab. Cutaneous lymphoma groups with experience in treating B burgdorferi–associated PCMZL with (systemic) antibiotics are encouraged to publish their treatment results, either positive or negative, to find out whether further studies are required. Controlled multicenter studies are also required to assess the efficacy of new therapies, such as yttrium-90 ibritumomab tiuxetan (Zevalin) or 131I-tositumomab (Bexxar) radioimmunotherapy97 and pegylated liposomal doxorubicin98 (plus rituximab) in PCLBCL, LT and gene therapy with adenovirus-mediated transfer of IFN-γ in PCFCL and PCMZL.99-101 Today, only the last mentioned approach is investigated in a prospective trial. Finally, patients with a CBCL can best be managed in centers where a close collaboration between dermatologist, hematologist/oncologist, and radiation oncologist exists. This is probably the most important recommendation and the best guarantee for optimal management.
Authorship
Contribution: N.J.S. performed the literature review, wrote the manuscript, and checked the final version of the manuscript; E.M.N., Y.H.K., M.B., E.B., L.C., R.D., M.D., R.T.H., N.P., S.T.R., M.H.V., and S.W. participated in consensus meetings, critically reviewed the manuscript, and checked the final version of the manuscript; and R.W. performed the literature review, participated in consensus meetings, wrote the manuscript, and checked the final version of the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Rein Willemze, Department of Dermatology, B1-Q, Leiden University Medical Center, PO Box 9600, 2300 RC Leiden, The Netherlands; e-mail: rein.willemze@planet.nl.