In this issue of Blood, Sindrilaru and colleagues' elegant study of a Vav3 mouse model demonstrates that Vav3 is critical for phagocytic synapse formation between apoptotic neutrophils and macrophages, thereby providing a switch within the wound microenvironment that regulates TGF-β production and tissue regeneration.
It has been recognized for over 2 decades that apoptotic cells in developing or injured tissues can be cleared in the absence of inflamed, fibrotic, or loss of functional tissue. Myeloid-derived phagocytes, upon interacting with apoptotic determinants, stimulate the production of anti-inflammatory cytokines, such as TGF-β and IL-10, and down-regulate proinflammatory cytokines, such as TNF-α, IL-12, and IL-6.1 In this issue of Blood, Sindrilaru et al provide an elegant demonstration showing that apoptotic cell clearance is not merely a passive event to suppress inflammation, but an active and necessary component in tissue regeneration.2
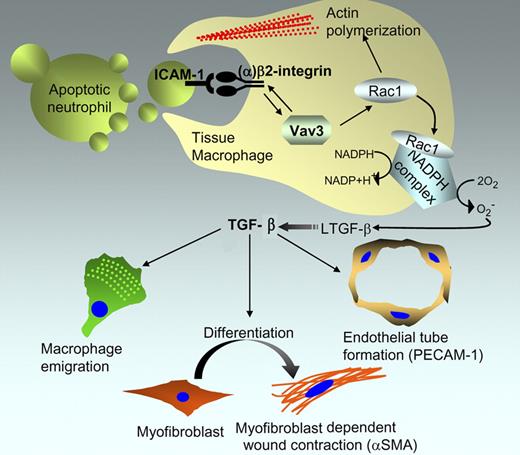
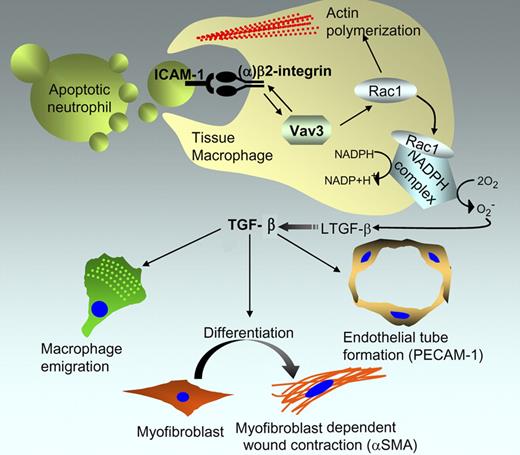
Proposed role of Vav3 in formation of the macrophage phagocytic synapse and its implications in wound regeneration. Based on the study by Sindrilaru et al, Vav3 stimulates a bidirectional signal to control (1) the avidity of CD18/β2 integrin toward ICAM on the apoptotic cell, and (2) the β2-integrin activation of Rac1 to stimulate phagocytosis and activate the NADPH complex that generates reactive oxygen species to activate latent TGF-β. Active TGF-β, in turn, coordinates a response within the microenvironment by acting on multiple cells, including macrophages, myofibroblasts, and endothelial cells that control wound healing.
Proposed role of Vav3 in formation of the macrophage phagocytic synapse and its implications in wound regeneration. Based on the study by Sindrilaru et al, Vav3 stimulates a bidirectional signal to control (1) the avidity of CD18/β2 integrin toward ICAM on the apoptotic cell, and (2) the β2-integrin activation of Rac1 to stimulate phagocytosis and activate the NADPH complex that generates reactive oxygen species to activate latent TGF-β. Active TGF-β, in turn, coordinates a response within the microenvironment by acting on multiple cells, including macrophages, myofibroblasts, and endothelial cells that control wound healing.
Vav proteins (Vav1, Vav2, and Vav3) are a family of guanine nucleotide exchange factors (GEFs) for Rho family GTPases that have important functions in cytoskeletal reorganization and hematopoiesis, and deregulated expression is often linked to tumorigenicity. Using a subcutaneous skin lesion model, Sindrilaru et al show that Vav3−/− mice, but not Vav1−/− or Vav2−/− mice, exhibit a delayed wound repair program, characterized by a deficit in macrophage emigration to the wound, myofibroblast-dependent wound contraction, and neoangiogenesis (see figure).
Subcutaneous lesions incurred in the Vav3−/− mice were not deficient in the infiltration of polymorphonuclear neutrophils (PMNs) during the early inflammatory phases of the wound repair, and infiltrating neutrophils underwent typical apoptotic cell death following their activation in the injury repair process. On the other hand, macrophages from Vav3−/− mice were unable to form a functional phagocytic synapse with the apoptotic PMNs in the wound margin, thereby suppressing phagocytosis and the ensuing production of TGF-β. Remarkably, the wound healing defect in the Vav3−/− mice could be corrected by a single injection of recombinant TGF-β at the wound margin at day 2 after injury.
To differentiate between a Vav3 deficit at the level of macrophage emigration or phagocytosis, wild-type or Vav3−/− macrophages were injected directly into the subcutaneous wound margin. Injection of wild-type Vav3 macrophages into Vav3−/− mice increased TGF-β production and wound closure while injection of Vav3−/− macrophages into wild-type mice failed to enhance wound healing, arguing convincingly for a role of Vav3 in phagocytosis.
The defect in wound healing in Vav3−/− mice recapitulated a similar phenotype associated with leukocyte adhesion deficiency-1 (LAD1), a condition characterized by recurrent bacterial infections stemming from leukocytes that are deficient in a wide range of adhesion-dependent functions.3 LAD1 deficiency in both humans and mice arises from a deficiency in CD18 (β2 integrin), and in mice, CD18−/− KO exhibited impaired myofibroblast differentiation and wound contraction, suggesting a relationship between β2 integrin and Vav3. Indeed, several lines of biochemical and cell biological evidence argue that there is Vav3 and β2 integrin crosstalk whereby Vav3 induces inside to outside integrin signaling and increases the strength of the β2 synapse for ICAM-1, a ligand expressed on apoptotic neutrophils. Further evidence for such functional crosstalk comes from studies showing that CD18−/− macrophages, like Vav3−/− macrophages, failed to rescue a normal wound healing phenotype, and biochemically, Vav3−/− macrophages show reduced adhesion to soluble ICAM or ICAM-1 surfaces.
The results of Sindrilaru et al contribute to a general debate on the mechanisms by which phagocytes recognize and internalize apoptotic cells and blebs. Several important questions arise from this study, not the least being why only Vav3, but not Vav1 or Vav2, mediates this wound repair phenotype. It is also not yet clear how Vav3 couples TGF-β production with actin-mediated phagocytosis. Rac1 appears to act on these pathways in distinct ways; the former involved in the formation of the NADPH complex to activate reactive oxygen species (ROS), and the latter for the cytoskeletal assemblages required for F-actin–mediated phagocytosis. The ability of Vav3 to activate Rac1 downstream of CD18/β2 integrin would clearly imply that distinct GEFs, besides CED12/ELMO and CED5/DOCK,4 have functional roles in physiologic cell clearance. It also remains to be seen whether Vav3 mediates inside to outside signaling toward other integrins that tether apoptotic cells. Interestingly, the Vav3−/− KO macrophages also showed decreased binding to αvβ3 integrin, this latter integrin having been clearly shown to recognize MFG-E8 on the surface of apoptotic cells and promote phagocytic clearance.5 Recent studies suggest that αv integrins also stimulate TGF-β production during physiological clearance.6
Finally, these studies raise the provocative possibility that Vav3 may be a selective target to alter macrophage responses in pathologic conditions. Although in the current study the authors showed the importance of phagocyte-derived TGF-β for controlling the wound healing microenvironment, TGF-β produced in the microenvironment of tumors, possible via the same clearance mechanisms described within, promotes tumor progression and extravasation of tumor cells from blood vessels, an essential stage for metastasis.7 In broader terms, strategies to target Vav3 may open up an important avenue of therapeutic medicine that harnesses phagocytic responsivity to apoptotic cells.
Conflict-of-interest disclosure: The authors declare no competing financial interests. ■
REFERENCES
National Institutes of Health