Abstract
Abstract 3025
Poster Board II-1001
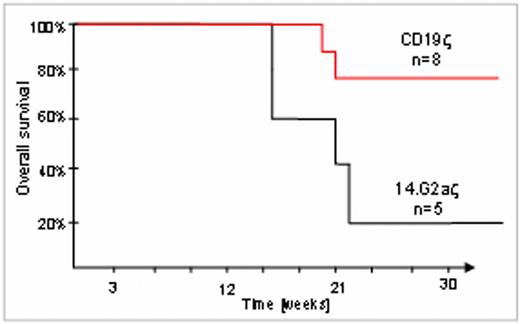
Due to its restriction to the B-cell lineage and high surface expression in B-cell malignancies, CD19 is an attractive target antigen for immunological strategies in B-cell precursor acute lymphoblastic leukemia (BCP-ALL). While preclinical in vivo studies of CD19-specific cellular immunotherapy have generally used xenografts from human CD19+ leukemia cell lines, primary leukemia cells are likely to more closely mimic the disease in humans and allow to differentiate between standard and high risk situations. Therefore, we investigated the in vivo sensitivity of human leukemic bone marrow to adoptive immunotherapy with gene-modified CD19-specific T cells. Among 15 primary leukemias obtained from the bone marrow of pediatric patients at diagnosis, 10 were successfully engrafted in NOD/scid mice by intrafemoral injection within 6 to 20 weeks. For therapeutic experiments, we focused on one standard risk leukemia, characterized by a rapid and sustained response to multiagent chemotherapy, and on a leukemia bearing the high-risk feature of an MLL rearrangement, which was refractory to standard treatment. Titration experiments demonstrated reliable engraftment of 1×104 leukemic cells per mouse. For CD19-directed T-cell therapy, cytotoxic T cells (CTLs) with native specificity for Epstein-Barr virus antigens were expanded from 4 healthy donors and transduced to express either a codon-optimized CD19-specific chimeric antigen receptor (CAR) containing the intracellular signaling domain of the TCRz chain (CD19-z), or a control CAR directed against the neuroectodermal antigen GD2 (14.G2a-z). Costimulatory domains now commonly used to ensure sustained T-cell activation via CARs were not included, since previous studies have shown that CAR activity in virus-specific CTLs does not benefit from additional signaling elements. CTLs had a uniform CD8+ effector memory T-cell phenotype (CD45RO+, CCR7-), and CAR surface expression was 73±21%, range 32-93% (CD19-z, n=9) and 18±13%, range 6-35% (14.G2a-z, n=5). In vitro cytotoxicity experiments confirmed specific lysis of the CD19+ leukemia cell lines REH (51Cr release 59.7±7.2% at an effector target ratio of 20:1) and SupB15 (66.7±8.6) as well as primary CD19+ leukemic cells from 5 pediatric patients (47.2±13.2%), in the absence of background lysis by 14.G2a-z-transduced control CTLs. 1×104 leukemic cells per mouse from primary engrafted mice were transferred into further cohorts of NOD/scid mice by secondary intrafemoral transplantation, followed by adoptive transfer of 4 doses of 5×106 CTLs via tail vein injection on days 1, 4, 8, and 11. IL-2 (500 IU/mouse) was administered twice-weekly, and sequential murine bone marrow aspirates were analyzed for human leukemia engraftment by flow cytometry using human CD45 and CD19-specific antibodies starting 3 weeks after transplantation. CD19z CTLs prevented engraftment of the standard risk leukemia in 3 of 4 mice, while 3 of 4 control mice developed the leukemia (p = 0.158, Log Rank/Mantel-Cox Test). Moreover, while the MLL-rearranged human leukemia became detectable in the bone marrow of 4 of 5 control mice, followed by overt and fatal leukemia, 5 of 8 mice receiving transfusions of CD19-z transduced CTLs remained disease-free (p = 0.067), and 6 of 8 remained alive, one of them with detectable leukemia cells (p = 0.054) (see Figure). Thus, adoptive transfer of CD19-redirected CTLs efficiently delayed or prevented engraftment of both standard and high risk ALLs in mice and therefore provides a promising treatment option for patients with BCP-ALL refractory to standard treatment.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.