In this issue of Blood, Baessler and colleagues find that the engagement of CD137 on mouse and human NK cells had opposite effects in that CD137 functioned as an inhibitory receptor in human NK cells and as a stimulatory receptor in mice.1 As NK cells emerge as potential therapeutic targets for leukemia treatment, the authors also investigate the expression of CD137L by leukemic cells. A significant level of expression was found for monocytic AML, which could contribute to immune evasion.
CD137, also known as 4-1BB, is a member of the TNFR super family. CD137 is closely related to CD27, OX40, and CD30. It is up-regulated on mouse and human T cells upon activation.2 It has long been recognized as a costimulatory molecule for T cells. CD137 engagement by CD137L on antigen-activated T cells increases proliferation, effector functions, and survival, in both mouse and human. Other members of the TNFR family such as OX40 or CD27 act similarly, and it is currently unknown whether these different receptorsact synergistically, sequentially, or via a more complex crosstalk.2,3 The use of anti-CD137 agonist antibodies in combination with other stimuli has proven to be effective treatment for various cancers in mouse models,2 and clinical trials using humanized agonist anti-CD137 mAb in cancer patients are ongoing.
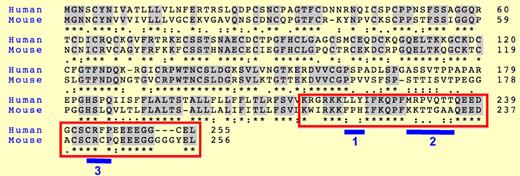
Alignment of human and mouse CD137 protein sequences. The red box corresponds to the intracellular regions. Blue bars highlight divergences between mouse and human. Stars indicate identity, and dots conservation in the type of amino acids between sequences.
Alignment of human and mouse CD137 protein sequences. The red box corresponds to the intracellular regions. Blue bars highlight divergences between mouse and human. Stars indicate identity, and dots conservation in the type of amino acids between sequences.
Many immune cell types including activated natural killer (NK) and NK T cells express CD137. The role of CD137 in these cells is unclear. Early studies showed that stimulation of mouse NK cells with cross-linking anti-CD137 antibodies or with CD137L-expressing cells induced their proliferation and IFN-g secretion but not cytotoxicity.4 Similar results have been reported for mouse CD27.5 However, the impact of CD137 engagement on human NK cells has not been previously reported. Baessler et al measured the expression of CD137 by NK cells from healthy donors and from patients with acute myeloid leukemic (AML), both on NK cells and on leukemic blasts. They found that CD137 is expressed by activated but not resting NK cells in healthy donors. In contrast, NK cells purified from AML patients express low levels of CD137 and CD69, suggesting some degree of activation in vivo. Moreover, they found CD137L expression by some AML blasts, particularly in monocytic leukemia. Next, they studied the consequences of CD137-CD137L interaction between NK cells and AML blasts in an allogenic setting. Surprisingly, they found that this interaction reduces NK-cell activation (IFN-g and cytotoxicity). Because this result clearly contradicts the literature on CD137 in mouse NK cells, they compared the stimulation of human and mouse NK cells via CD137 using CD137L or mock-transfected cell lines, and blocking or agonist anti-CD137 antibodies. They convincingly showed that triggering CD137 mediates an inhibitory signal in human NK cells but an activating one in mouse NK cells.
A battery of receptors, either activating or inhibitory, governs NK-cell activation.6 Although several TNF receptors are expressed by NK cells such as CD27, CD137, or GITR, their contribution to NK-cell activation by target cells is rarely evaluated. This study clearly shows that CD137L on leukemic cells is an important signal recognized by NK cells via CD137. The investigators propose that CD137L expression by AML blasts may contribute to immune evasion. However, as CD137 is costimulatory in CD8 T cells, the overall impact of CD137L expression by AML cells is difficult to evaluate. The findings by Baessler et al may have consequences on the design of therapies based on the manipulation of CD137 activity in patients with cancer. In particular, agonist anti-CD137 antibodies should be evaluated carefully before being used in leukemic patients.
How can the discrepancy in the outcome of CD37 triggering in mouse and human NK cells be explained? This study did not address this question, so, at this point, only speculations are possible. The CD137 intracellular region binds to TRAF1-3.3 Human but not mouse CD137 binds TRAF3, which may have a negative impact on CD137 signaling.7 Moreover, human and mouse CD137 intracellular regions differ by small stretches of amino acids that might bind inhibitory molecules such as phosphatases (see figure). Of note, one of these motifs, present only in human CD137 (blue bar “1” in the figure) resembles an immuno-tyrosine inhibitory motif, although it lacks the canonical terminal amino acid (I, V, or L). Reciprocally, mouse but not human C137 contains the canonical CXCP motif that binds the kinase p56lck.8 Another puzzling point is the opposite outcome of CD137 triggering in NK and CD8 T cells in humans. Again, a differential expression of inhibitory signaling partners between NK and CD8 T cells can be postulated. Moreover, heterodimerization between TNFR members has been observed,9 which may also influence the response of individual receptors in different cell subsets.
This study highlights once again that molecular pathways may vary greatly between mouse and human. Differences between mouse and human immunology have been appreciated10 and genome-wide analyses point to even more differences.11 Therefore, new models that allow in vivo or in vitro testing on a human immune system are necessary before going to the clinic.
Conflict-of-interest disclosure: The authors declare no competing financial interests. ■