Abstract
Abstract 3743
CC-Chemokine receptor 8 (CCR8) is a chemokine receptor expressed on the surfaces of activated effector T cells (Teffs) and resting regulatory T cells (Tregs). CCR8 has previously been shown to play a role in the trafficking of memory T cells into cutaneous and possibly pulmonary sites, and to contribute to the ability of Tregs to properly interact with antigen presenting cells within secondary lymphoid tissue. Since the lungs and in particular the skin are frequent sites of tissue injury in those developing graft-versus-host disease (GVHD) after hematopoietic stem cell transplantation (HSCT), we set out to study the contribution of CCR8 to GVHD pathogenesis using a haploidentical murine HSCT model. Given the emerging utilization of therapeutic donor Treg infusions as a means for preventing GVHD, we also explored the role that CCR8 plays in the protective immunosuppressive function of Tregs in the HSCT setting.
For our studies we used a haplotype-matched murine transplant model. C57BL/6 mice (B6; H-2b) functioned as donors, and B6 × DBA2 (B6D2; H-2bxd) mice served as recipients. For Teff studies, B6D2 mice were lethally irradiated to 950 rads on transplant day -1, and then administered 3×106 T cell depleted (TCD) B6 bone marrow (BM) cells +/− 4×106 CD25− Teffs isolated from the spleens of wild-type (WT) or CCR8−/− B6 mice on day 0. For studies involving regulatory T cells, B6D2 animals were lethally irradiated on day -1 and administered WT B6 TCD BM cells +/− 1–1.25×106 CD4+CD25+ Tregs isolated from WT or CCR8−/− B6 mice on day 0. 4×106 WT B6 Teffs were then administered on transplant day +2. For some transplants, WT B6 and CCR8−/− animals transgenic for enhanced green-fluorescent protein (eGFP+) were used as Teff and/or Treg donors in order to study T cell trafficking following HSCT. Both stereo-fluorescence microscopy and anti-eGFP ELISA approaches were utilized for these in vivo trafficking experiments. The in vitro suppressive potential of WT and CCR8−/− Tregs was evaluated by examining their ability to inhibit Teff proliferation in a one way mixed lymphocyte reaction (MLR).
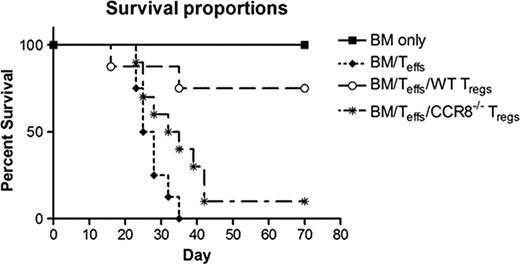
CCR8 was not required for the induction of lethal GVHD, with recipients of CCR8−/− Teffs demonstrating a 100% mortality rate by transplant day +32. However, CCR8 was important for Treg function. In vivo, CCR8−/− Tregs were significantly less effective at preventing GVHD lethality than WT Tregs (see figure; P=0.013 for day +70 survival proportion comparison using Fisher's exact test). When we evaluated donor Treg trafficking on transplant days 6–7 using eGFP+ cells, no significant differences were noted between those animals receiving WT versus CCR8−/− Tregs. By transplant days 9–10, however, a generally higher eGFP signal was observed by stereo-fluorescence microscopy within the lymph nodes of those animals receiving WT eGFP+ Tregs compared to those given CCR8−/− eGFP+ Tregs. In addition, significantly more eGFP was detected by ELISA in the spleens and colons of WT eGFP+ Treg recipients (P=0.0015 and 0.021 respectively for mean total eGFP comparison between WT and CCR8−/− Treg groups by student's t test), and a strong trend for greater WT Treg accumulation was noted within host livers and lungs (P=0.059 and 0.066 respectively). Finally, CCR8−/− Tregs were relatively impaired in their ability to suppress T cell proliferation in vitro compared to WT cells, particularly at higher dilutions.
CCR8 is not required for donor Teffs to induce lethal GVHD in our HSCT model. However, CCR8 expression does appear to be important for the function of Tregsin vitro and in vivo, particularly at limiting Treg dilutions. CCR8 is not required for the accumulation of donor Tregs within any one recipient site. Rather, CCR8 appears to globally potentiate the accumulation of donor Tregs within most recipient tissues, with the extent of this effect increasing over time. Future experiments will focus on elucidating a mechanism for this finding.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.