Abstract
Abstract 1462
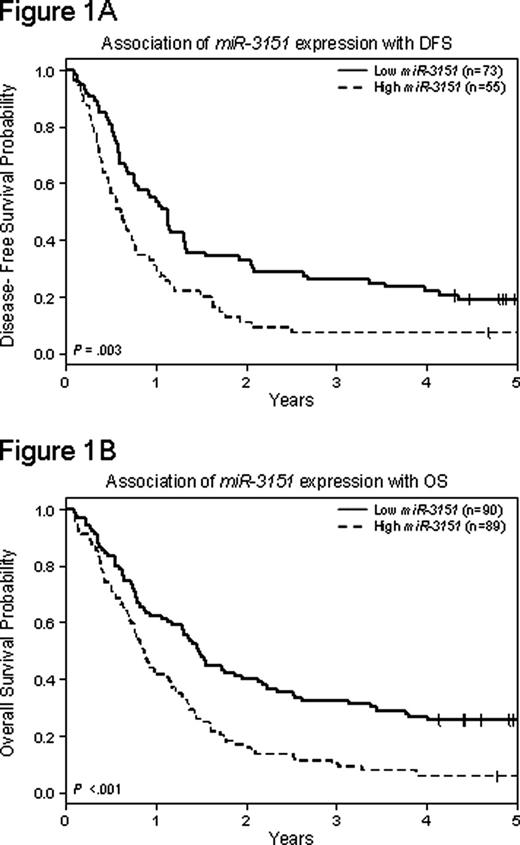
Despite progress made in understanding the biology and risk-adapted treatment of AML, prognosis of older pts [age ≥60 years (y)] remains poor. We have shown that high expression of the BAALC gene bestows worse outcome in older CN-AML pts. Yet, the mechanism(s) by which BAALC affects response to therapy remain unknown. MicroRNAs (miRs) are small noncoding RNAs that hybridize to target mRNAs and inhibit their translation or promote degradation, resulting in downregulation of the corresponding proteins. Recently, miR-3151 was discovered embedded in intron 1 of BAALC. We thus hypothesized that miR-3151 might be co-expressed with BAALC and contributes alone or in concert with its host to poor prognostic impact in older CN-AML. MiR-3151 and BAALC expression were measured by quantitative real time (RT) PCR in pretreatment blood of 179 older CN-AML pts enrolled on Cancer and Leukemia Group B (CALGB) cytarabine/daunorubicin-based protocols. The expression values of miR-3151 and BAALC were normalized to the internal controls SNORD44 and RN18S1, respectively. MiR-3151 expression correlated only weakly with the expression of BAALC (Spearman correlation r= 0.3). For clinical and prognostic analyses pts were dichotomized into high and low miR-3151 and BAALC expressers at the median miR and gene transcript expression values and analyzed for other molecular prognosticators (FLT3 -ITD, FLT3 -TKD, CEBPA, IDH1, IDH2, NPM1, TET2, WT1 mutations and ERG expression). Gene (GEP) and microRNA (MEP) expression profiles associated with miR-3151 expression were derived, using Affymetrix U133 plus 2.0 & OSU CCC v4.0 arrays, respectively. At diagnosis, higher miR-3151 expression was associated with lower % blood blasts (P =.02), FAB types M4 and M5 (P =.05), and wild-type NPM1 (P<. 001). High miR-3151 expressers had a lower complete remission (CR) rate (62% v 81%, P=. 005), and, with a median follow-up of 5.1 y for pts alive, shorter disease-free (DFS; hazard ratio [HR]=1.76, P=. 003; 3y rates, 7% v 26%; Figure 1A) and overall survival (OS; HR=1.86, P <.001; 3y rates, 10% v 32%; Figure 1B) than low expressers. In multivariable analyses (MVA), higher miR-3151 expression no longer remained in the model for CR after adjusting for BAALC expression and white blood count (WBC), but higher miR-3151 expression remained associated with shorter DFS (HR=2.32, P <.001), after adjusting for FLT3 -TKD, ERG expression status and WBC, and shorter OS (HR=1.77, P=. 003), after adjusting for ERG and BAALC expression status. To gain biological insights of miR-3151 -associated AML, we derived a GEP comprising 377 annotated genes and a MEP comprising 14 miRs. High miR-3151 expressers exhibited upregulation of genes associated with immature differentiation stage and adverse outcome (eg, MN1, ID1), downregulation of genes involved in hematopoietic differentiation (eg, MEIS1, PDCD4, PBX3) and transcriptional regulators (ZNF s, E2F3), including predicted targets of miR-3151 (eg, MEIS1). In the MEP, high miR-3151 expressers showed downregulation of let-7a/b/c that play crucial roles in cell cycle and proliferation regulation. While miR-3151- associated GEP/MEP shared genes with BAALC expression-associated GEP/MEP (eg, upregulated MN1 and DNTT and downregulated MEIS1, let-7b, miR-10a/b), other genes were unique to miR-3151- associated GEP/MEP (eg, upregulated ID1 and downregulated ZNFs, miR-206 and let-7a/c), suggesting biologic mechanisms distinct from those directly connected with BAALC upregulation operating in miR-3151- associated leukemia. In conclusion, high miR-3151 expression is independently associated with worse survival in older CN-AML pts. The weak correlation between expression of miR-3151 and its host gene BAALC, the independent impact of each on outcome endpoints (BAALC was a strong predictor of CR but did not predict DFS while miR-3151 was not independently associated with CR, but remained in the DFS MVA) and differences in GEP/MEP suggest that miR-3151 and BAALC are deregulated and contribute to disease outcome differently. We surmise that determining the expression levels of miR-3151 at diagnosis might help to improve the risk-stratification of older CN-AML. Development of therapies targeting miR-3151 upregulation with synthetic inhibitors may provide novel, effective strategies for personalized treatment of older CN-AML pts.
Disclosures:
No relevant conflicts of interest to declare.
Author notes
*
Asterisk with author names denotes non-ASH members.
© 2011 by The American Society of Hematology
2011