Abstract
Mutation status of FLT3, NPM1, CEBPA, and WT1 genes and gene expression levels of ERG, MN1, BAALC, FLT3, and WT1 have been identified as possible prognostic markers in acute myeloid leukemia (AML). We have performed a thorough prognostic evaluation of these genetic markers in patients with pediatric AML enrolled in the Nordic Society of Pediatric Hematology and Oncology (NOPHO) 1993 or NOPHO 2004 protocols. Mutation status and expression levels were analyzed in 185 and 149 patients, respectively. Presence of FLT3-internal tandem duplication (ITD) was associated with significantly inferior event-free survival (EFS), whereas presence of an NPM1 mutation in the absence of FLT3-ITD correlated with significantly improved EFS. Furthermore, high levels of ERG and BAALC transcripts were associated with inferior EFS. No significant correlation with survival was seen for mutations in CEBPA and WT1 or with gene expression levels of MN1, FLT3, and WT1. In multivariate analysis, the presence of FLT3-ITD and high BAALC expression were identified as independent prognostic markers of inferior EFS. We conclude that analysis of the mutational status of FLT3 and NPM1 at diagnosis is important for prognostic stratification of patients with pediatric AML and that determination of the BAALC gene expression level can add valuable information.
Introduction
Acute myeloid leukemia (AML) is a clinically and biologically heterogeneous disease that constitutes 15%-20% of childhood leukemia and has an overall survival (OS) of ∼ 70%. The prognosis has improved over the past couple of decades through optimization of treatment protocols, in which identification of disease subgroups that are based on genetic markers has turned out to be essential.
One of the first genes identified as being commonly mutated in AML was that of the Fms-related tyrosine kinase 3 (FLT3, chromosome localization 13q12).1,2 FLT3 is a class III receptor tyrosine kinase with multiple downstream targets involved in proliferation, apoptosis, and differentiation of hematopoietic progenitors. Mutations in FLT3 include both internal tandem duplication (FLT3-ITD) in the juxtamembrane part and mutations in the tyrosine kinase domain (FLT3-TKD), and they result in constitutive activation of the FLT3 receptor. FLT3-ITD is associated with a higher risk of disease progression and inferior survival in adult AML with normal karyotype (CN-AML),3,4 and several studies on pediatric AML have also shown an association with poor prognosis.5-7 The prognostic relevance of FLT3-TKD mutation is less well established.4 The prognostic role of increased level of wild-type (wt) FLT3 in AML is also unclear, but overexpression of wt FLT3 has been reported in a large proportion of patients with AML without FLT3 mutations. Furthermore, high expression of wt FLT3 has been suggested to be associated with both distinct AML subtypes and an unfavorable prognosis in both adult and pediatric AML.8-10
Mutations in the nucleophosmin gene (NPM1, 5q35) and in the CCAAT/enhancer binding protein, α gene (CEBPA, 19q13) have been identified in patients with AML.11,12 AML with mutated NPM1 or mutated CEBPA is associated with a favorable prognosis,13-16 and these have been defined as provisionally distinct entities in the 2008 World Health Organization classification of AML. NPM1 is a multifunctional nuclear chaperone and histone binding protein involved in ribosome biogenesis, chromatin remodeling, DNA replication, and DNA repair. The mutations in NPM1 are frameshift mutations that alter the C-terminus and cause delocalization from the nucleus to the cytoplasm.11 CEBPA is a transcription factor essential for granulocyte differentiation. Two types of mutations are found in the CEBPA gene, either frameshift mutations in the N-terminal part that lead to expression of a truncated protein or missense mutations near the C-terminus that cause impaired DNA binding.12 A few studies have investigated the role of NPM1 and CEBPA mutations in pediatric AML and found a correlation with favorable prognosis.17-20
Wilms tumor 1 (encoded by WT1, 11p13) is a transcription factor for genes regulating cell growth, metabolism, and growth factors, and disruptive frameshift mutations have been identified in patients with AML.21 In adult AML, predominantly in patients with CN-AML, the presence of WT1 mutation has been associated with poor prognosis.22-24 High WT1 expression has been associated with poor prognosis in some studies, but others have failed to detect a correlation with survival at diagnosis in adult AML.25,26 In pediatric AML, the prognostic relevance of both WT1 mutation and high WT1 expression level is unclear, but recent results have suggested that the presence of WT1 mutation has no independent prognostic significance in predicting outcome in pediatric AML.27
In addition to gene mutations, aberrant expression of the Ets-related gene (ERG, 21q22), the Meningioma 1 gene (MN1, 22q12), and the gene for brain and acute leukemia, cytoplasmic (BAALC, 8q22) have been identified as prognostic markers. ERG is a transcription factor and a potent oncogene involved in fusion genes found in prostate cancer, Ewing sarcoma, and AML. Deregulation of ERG is common both in acute lymphoblastic leukemia and in AML.28,29 MN1 is a hematopoietic oncogene and transcription factor. In AML, MN1 has been reported to be involved in an uncommon balanced translocation that results in expression of the MN1-TEL fusion gene, and high MN1 expression is common, especially in association with inv(16)(p13q22) (CBFB-MYH11) rearrangement.30,31 BAALC is expressed in hematopoietic progenitor cells and in neuroectodermal tissues and was discovered in a gene expression screen of patients with AML with sole trisomy 8.32 The function of BAALC, both in normal BM and in leukemogenesis, remains unknown. The prognostic significance of ERG, MN1, and BAALC expression levels has mainly been studied in adult AML, and high expression has been linked to poor response to chemotherapy and to poor prognosis, especially in CN-AML.29,30,33-35
In this study, we have evaluated the prognostic relevance of mutations in the FLT3, NPM1, CEBPA, and WT1 genes and of levels of expression of ERG, MN1, BAALC, FLT3, and WT1 both as single markers but also in relation to each other because there are a limited number of studies on pediatric AML that include all these factors.
Methods
Patient samples and treatment protocols
Leukemic cells from 185 children with newly diagnosed AML were collected at the centers for pediatric oncology in Denmark, Finland, Iceland, Norway, and Sweden between 1997 and 2007. The age range was 0-18 years with a median age of 6 years. Patients with Down syndrome or acute promyelocytic leukemia were not included. The patients were treated according to 2 consecutive treatment protocols, NOHPO-AML 93 (1997-2003)36 and Nordic Society of Pediatric Hematology, and Oncology (NOPHO)–AML 2004 (2004-2007).37 Patient characteristics, treatment, and follow-up data have been collected continuously in the NOPHO AML database with the last day of follow-up on June 1, 2010. Genomic DNA was obtained from all 185 patients, whereas RNA was available from 149 of these patients. Cytogenetic data were available for 183 of the patients. The study was performed in accordance to the Declaration of Helsinki, and parents and/or patients gave informed consent. The local research ethics committee (EPN) at Uppsala University approved the study, Dnr 2007/023.
Cell fractionation and DNA and RNA extractions
Peripheral blood or BM samples were collected in heparinized glass tubes, and mononuclear cells were isolated by Ficoll gradient centrifugation. DNA and RNA were extracted with the use of the All Prep DNA/RNA mini kit (QIAGEN) either from pelleted cells frozen at −70°C or from cryopreserved cells stored in liquid nitrogen. The quality and concentration of DNA and RNA were analyzed with a Nanodrop 1000 spectrophotometer (Thermo Scientific) and a Bioanalyzer (Agilent Technologies).
Mutation analysis
FLT3-ITD mutations were analyzed by PCR amplification of genomic DNA and subsequent fragment analysis as previously described38 with the use of an ABI 3130XL Genetic Analyzer (Applied Biosystems) and Peak Scanner Version 1.0 software (also Applied Biosystems). The presence of any PCR fragment larger than that derived from the wt allele (328 bp) was considered evidence of positivity for FLT3-ITD. To screen for FLT3-TKD mutations (D835 or I836), genomic DNA was PCR-amplified and analyzed as described previously.2 The presence of mutations in exon 12 of the NPM1 gene were detected by PCR amplification of genomic DNA with the use of primers NPM1_F_FAM (5′-TTAACTCTCTGGTGGTAGAATGAA-3′) and NPM1_R (5′-ACCATTTCCATGTCTGAGCACC-3′). PCR products were analyzed by fragment analysis with the use of an ABI 3130XL Genetic Analyzer (Applied Biosystems) and Gene Mapper Version 4.0 software (also Applied Biosystems).
Mutations in the CEBPA gene were detected by PCR amplification of the entire coding region with the use of primer pairs PP1 and PP2 previously described12 and sequenced with BigDye Terminator Version 1.1 Cycle Sequencing Kit (Applied Biosystems). Patients with identified mutations were in addition analyzed by fragment analysis and sequenced as described elsewhere.39 SeqScape Version 2.5.0 software (Applied Biosystems) with GenBank NM_0004364.3 as reference sequence was used to determine sequence variants, with the following being regarded as polymorphisms: c.690G > T (rs34529039), c.574_578dup, and c.573G > C.40,41
WT1 mutations were detected by PCR amplification of genomic DNA and sequencing of exon 7 and exon 9 where the majority of reported WT1 mutations have been found.24 Primers used for PCR and sequencing were WT1ex7-F (5′-ATGGGGATCTGGAGTGTGAAT-3′), WT1ex7-R (5′-ACAGCGGGCACACTTACC-3′), WT1ex9PCR-F (5′-GGACTGGGGAAATCTAAG-3′), WT1ex9PCR-R (5′-TTCTGCTGCTTTTCTGTA-3′), WT1ex9Seq-F (5′-GGACTGGGGAAATCTAAG-3′), and WT1ex9Seq-R (5′-AAAGATAGCCACGCACTA-3′). Sequences were analyzed on the ABI 3130 Avant (Applied Biosystems) with the use of the BigDye Terminator Version 1.1 Cycle Sequencing Kit (Applied Biosystems).
Expression analysis
cDNA was synthesized from 100-500 ng of total RNA with the use of SuperScript II reverse transcriptase (Invitrogen) and random hexamer primers. cDNA was diluted 1:100 in QuantiTect SYBR Green PCR Master Mix (QIAGEN) containing gene-specific primers. Primer sequences for ERG, BAALC, and MN1 have been described previously.28,30,34 For WT1, FLT3, and ABL1 the following primers were used: WT1-s (5′-GATAACCACACAACGCCCATC-3′), WT1-as (5′-CACACGTCGCACATCCTGAAT-3′), FLT3-s (5′-GCATCCCAGTCAATCAGCTT-3′), FLT3-as (5′-GATTTTCGAGAGAAGGTCCAC-3′), ENF1003(ABL1-s) (5′-TGGAGATAACACTCTAAGCATAACTAAAGG-3′), and ENR1063(ABL1-as) (5′-GATGTAGTTGCTTGGGACCCA-3′). Reactions were analyzed on a Rotorgene 6000 instrument (QIAGEN), and relative expression was calculated according to the 2−ΔΔC(t) method with a calibrator sample included in all separate runs as reference, and with ABL1 value as endogenous control. High and low gene expressions were defined as above and below the median value, respectively.
Statistical methods
To compare the distribution of categorical or dichotomized variables, the chi-square test was used, and Fisher exact test (2-tailed) was used when the expected count in ≥ 1 cell of the table was < 5. Mann-Whitney U test (comparing 2 groups) and Kruskal Wallis test (comparing > 2 groups) were used to test for differences in continuous variables. OS was defined as the time from diagnosis to last follow-up or death from any cause. Event-free survival (EFS) was defined as the time from diagnosis until relapse, resistant disease (defined as event on day 0), or death from any cause. The probabilities of EFS and OS were estimated by the Kaplan-Meier method, and differences between 2 survival distributions were compared with the log-rank test. Multivariate Cox proportional hazard regression analysis was used to determine independent prognostic factors for OS and EFS. P values of < .05 were considered statistically significant (2-tailed testing), and, when subgroup analyses were performed, Bonferroni correction for multiple testing was applied.
Results
Presence of FLT3-ITD and NPM1 mutations but not FLT3-TKD mutations are prognostic markers in pediatric AML
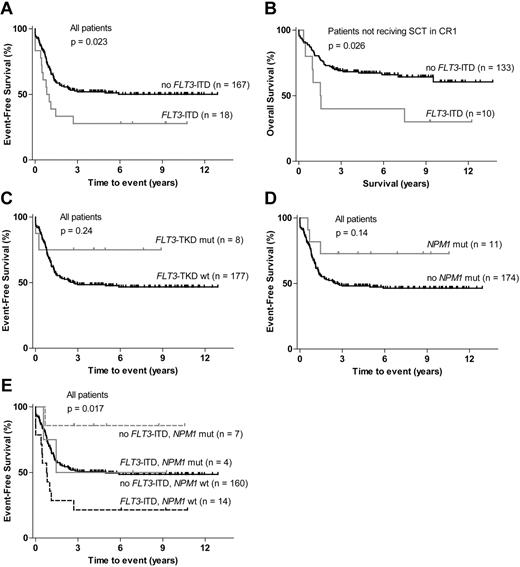
FLT3-ITD was detected in 18 of the 185 patients (10%) included in the study. FLT3-ITD–positive patients had an inferior EFS compared with patients without FLT3-ITD (P = .023; Figure 1A). No significant difference was observed in OS between the FLT3-ITD–positive and the FLT3-ITD–negative groups when all 185 patients were included in the analysis (P = .36; supplemental Figure 1A, available on the Blood Web site; see the Supplemental Materials link at the top of the online article). However, the percentage of patients who underwent SCT in CR1 was significantly higher in the FLT3-ITD group (44%) than in the FLT3-ITD–negative group (20%; Table 1). For the 142 patients who did not receive SCT in CR1, OS was inferior in the FLT3-ITD group (P = .026; Figure 1B). Patients with FLT3-ITD had significantly higher WBC count and were older than patients without FLT3-ITD (Table 1). FLT3-ITD was significantly more common in the normal cytogenetic subgroup (Table 2) and commonly coexisted with NPM1 and WT1 mutations (P = .014 and P = .005; supplemental Figure 2).
Survival according to FLT3 and NPM1 mutation status. (A) EFS for patients with FLT3-ITD compared with FLT3-ITD–negative patients. (B) OS for patients not treated with SCT (stem cell transplantation) in first complete remission (CR1): patients with FLT3-ITD compared with FLT3-ITD–negative patients. (C) EFS for patients with FLT3-TKD mutations compared with wt FLT3-TKD patients. (D) EFS for patients with NPM1 mutations compared with wt NPM1 patients. (E) EFS for patients stratified on FLT3-ITD and NPM1 mutation status.
Survival according to FLT3 and NPM1 mutation status. (A) EFS for patients with FLT3-ITD compared with FLT3-ITD–negative patients. (B) OS for patients not treated with SCT (stem cell transplantation) in first complete remission (CR1): patients with FLT3-ITD compared with FLT3-ITD–negative patients. (C) EFS for patients with FLT3-TKD mutations compared with wt FLT3-TKD patients. (D) EFS for patients with NPM1 mutations compared with wt NPM1 patients. (E) EFS for patients stratified on FLT3-ITD and NPM1 mutation status.
FLT3-TKD mutations (D835 or I836) were found in 8 patients (4%). No significant difference was observed in EFS or OS between FLT3-TKD–mutated and FLT3-TKD wt patients (Figure 1C; supplemental Figure 1B). No patients with both FLT3-TKD and FLT3-ITD mutation were found.
NPM1 mutations were detected in 11 cases (6%), and these patients had a trend for favorable EFS and OS although not statistically significant (Figure 1D; supplemental Figure 1C). Because FLT3-ITD and NPM1 mutations commonly coexisted, the different FLT3-ITD/NPM1 subgroups were analyzed, and this showed that patients with NPM1 mutation without FLT3-ITD had significantly superior EFS (Figure 1E).
High FLT3 gene expression associates with high WBC count but not with survival in pediatric AML
Relative levels of FLT3 transcripts were analyzed in 149 patients in whom RNA was available. There was a significant association between high FLT3 expression and high WBC count (Table 1). High FLT3 expression also correlated with the presence of FLT3-TKD and WT1 mutations (P = .028 and P = .036; supplemental Figure 2). The expression level of FLT3 did not correlate with EFS or OS (supplemental Figure 3A; data not shown). There was no significant difference in FLT3 expression level between different cytogenetic subgroups (Table 2).
CEBPA and WT1 mutations are associated with normal karyotype but do not correlate with survival in pediatric AML
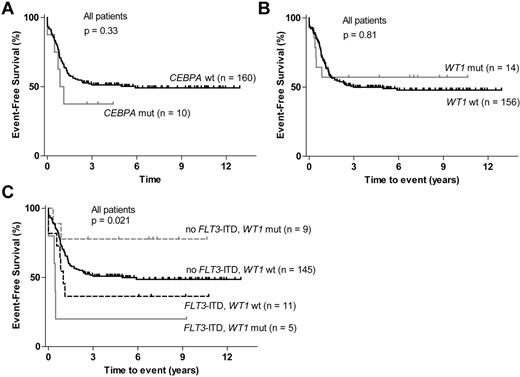
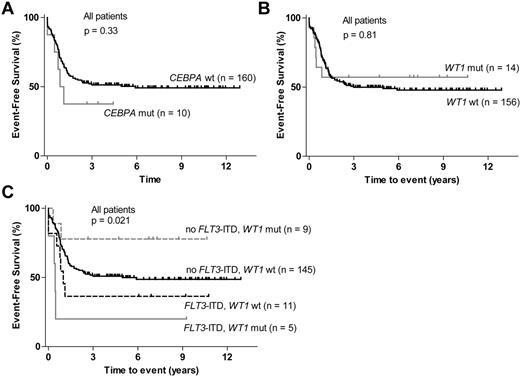
CEBPA and WT1 mutations could be analyzed in 170 of the 185 patients. CEBPA mutations were detected in 10 of these patients (6%). No significant association between mutated CEBPA (double, single, or any) and EFS or OS could be detected (Figure 2A; supplemental Figure 1E). Three patients with CEBPA mutation also had a WT1 mutation (P = .038; supplemental Figure 2), and CEBPA mutation was significantly associated with a normal karyotype (Table 2).
Survival according to CEBPA and WT1 mutation status. (A) EFS for patients with CEBPA mutations and for patients with wt CEBPA. (B) EFS for patients with WT1 mutations compared with wt WT1 patients. (C) EFS stratified according to FLT3-ITD and WT1 mutation status.
Survival according to CEBPA and WT1 mutation status. (A) EFS for patients with CEBPA mutations and for patients with wt CEBPA. (B) EFS for patients with WT1 mutations compared with wt WT1 patients. (C) EFS stratified according to FLT3-ITD and WT1 mutation status.
WT1 mutations were found in 14 of the 170 patients (8%), and most mutated patients were in the cytogenetically normal subgroup (Table 2). No correlation between mutated WT1 and EFS or OS could be detected (Figure 2B; data not shown). WT1 mutation has been reported to be an adverse prognostic factor in other studies, although not always as an independent factor, partly because of a high frequency of coexisting FLT3-ITD.27,42 In accordance with this, 5 of the 14 patients with WT1 mutation also had FLT3-ITD (P = .005; supplemental Figure 2). Because of this association we also analyzed the different WT1/FLT3-ITD subgroups. Patients with a WT1 mutation and no FLT3-ITD actually had superior EFS compared with patients with wt WT1 with or without concurrent FLT3-ITD (Figure 2C).
High WT1 gene expression correlates with the presence of WT1 mutation and FLT3-ITD but not with survival in pediatric AML
WT1 expression levels were analyzed in 149 patients in whom RNA was available. High levels of WT1 transcripts correlated with the presence of WT1 mutation and with FLT3-ITD (P = .005 and P = .009; supplemental Figure 2). There was no significant difference in EFS or OS between patients with high and low WT1 expression (supplemental Figure 3B; data not shown). High WT1 expression was seen in AML with core binding factor (CBF) rearrangements [inv(16)(p13.1q22), t(16;16)(p13.1;q22), or t(8;21)(q22;q22)] and in CN-AML, whereas low expression levels were observed in AML with MLL rearrangement and monocytic or monoblastic AML (FAB M5; Table 2; supplemental Table 1).
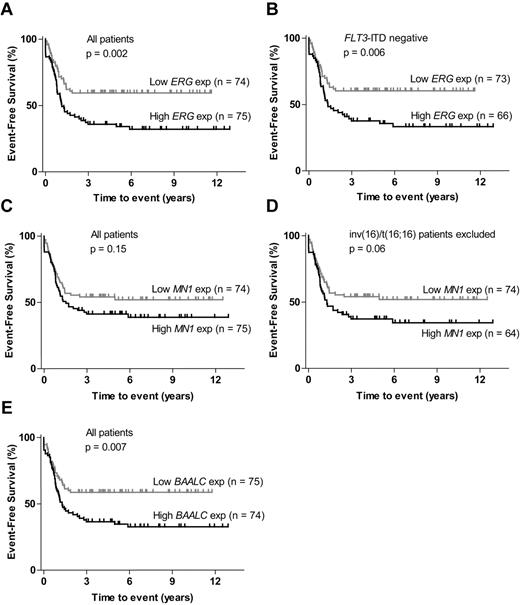
High ERG and high BAALC expression are associated with inferior EFS in pediatric AML
Relative levels of ERG, MN1, and BAALC expression were analyzed in 149 patients. For all 3 genes, high expression level was associated with higher age and AML without or with differentiation (FAB M1 and M2), whereas there was strong correlation between low expression of these genes and monocytic or monoblastic AML (FAB M5; Table 1; supplemental Table 1). ERG, MN1, and BAALC were commonly coexpressed, 44 of the 149 patients (30%) had high levels of all 3 genes (supplemental Figure 2).
EFS for patients with high ERG expression was significantly inferior to patients with low ERG (P = .002; Figure 3A). Similar ERG expression levels were found in all cytogenetic subgroups, except for slightly lower levels in patients with MLL rearrangements (Table 2), and FLT3-ITD was common in patients with high ERG expression (P = .018; supplemental Figure 2). Exclusion of patients with FLT3-ITD mutation had no influence on ERG correlation with EFS (P = .006; Figure 3B). There was no significant difference in OS between patients with AML with high and low ERG expression (supplemental Figure 3C). No significant difference in EFS was detected between patients with high and low MN1 expression; however, a trend for inferior EFS in patients with high MN1 expression could be seen (P = .15; Figure 3C). High MN1 expression levels were detected in almost all patients with CBF leukemia (Table 2). Because it also has been reported that patients with inv(16) or t(16;16) rearrangement are associated with high MN1 expression,31 we excluded these patients. The association between high MN1 expression and inferior EFS was strengthened but still not significant (P = .06; Figure 3D). No significant difference in OS between patients with high and low MN1 expression could be detected (supplemental Figure 3D). EFS for patients with high BAALC expression was significantly inferior than for patients with low BAALC expression (P = .007; Figure 3E). Expression levels of BAALC varied between the different cytogenetic subgroups, with lower BAALC levels being detected in MLL-rearranged AML and high BAALC expression levels in CBF AML (Table 2). No significant difference in OS could be detected between patients with high and low BAALC expression (supplemental Figure 3E).
ERG, MN1, and BAALC expression. (A) EFS for patients with high versus low ERG expression. (B) EFS in FLT3-ITD–negative patients: high versus low ERG expression. (C) EFS for patients with high versus low MN1 expression. (D) EFS for non-inv(16)/t(16,16) patients: high versus low MN1 expression. (E) EFS for patients with high versus low BAALC expression.
ERG, MN1, and BAALC expression. (A) EFS for patients with high versus low ERG expression. (B) EFS in FLT3-ITD–negative patients: high versus low ERG expression. (C) EFS for patients with high versus low MN1 expression. (D) EFS for non-inv(16)/t(16,16) patients: high versus low MN1 expression. (E) EFS for patients with high versus low BAALC expression.
FLT3-ITD and high BAALC expression are independent prognostic factors in pediatric AML
The genetic markers that were significantly associated with inferior or improved EFS in univariate analysis (FLT3-ITD, mutated NPM1, high ERG, and high BAALC expression) were evaluated as independent prognostic markers with the use of a Cox regression model. Other variables in the multivariate analysis were WBC count higher than 100 × 109/L, age, cytogenetic risk group, response to induction therapy, and treatment with SCT in CR1 as time-dependent variable (Table 3). One hundred forty-four patients could be included from whom all this information was available. The analysis showed that FLT3-ITD, high BAALC expression, good response to induction therapy, and SCT in CR1 were independent prognostic factors for EFS. High-risk cytogenetics was the only independent factor for OS.
Discussion
The relevance and prognostic value of mutations in FLT3, NPM1, CEBPA, and WT1 have been studied mainly in adult AML and to a lesser extent in patients with pediatric AML. The significance of high ERG, MN1, BAALC, FLT3, and WT1 expression has not been thoroughly evaluated in patients with pediatric AML. In addition, studies on the prognostic relevance of these genetic markers in AML have not been studied in relation to each other and have mainly been focused on CN-AML.
In this study, we have been able to show that the presence of FLT3-ITD was an independent adverse prognostic marker with inferior EFS, especially in the absence of NPM1 mutation. We could not find a significant correlation with OS, but, when patients undergoing SCT in first remission were excluded, OS was also significantly inferior in FLT3-ITD–positive patients. This indicates that SCT in CR1 may be of benefit in FLT3-ITD–positive patients. As a consequence, SCT is now recommended in NOPHO-AML for patients with FLT3-ITD mutation without an NPM1 mutation. The presence of FLT3-ITD was associated with a normal karyotype and was found with similar frequency (28%) to that seen in adult CN-AML. An FLT3-TKD mutation was found in 4% of cases, which is in accordance with previous studies on pediatric AML,6,43 and FLT3-TKD was more frequent in 2 overlapping subgroups, AML with t(9;11)(p22;q23) rearrangement and monocytic or monoblastic AML (FAB M5) but did not show any prognostic relevance (Table 2; supplemental Table 1). In addition, the expression level of FLT3 did not show any prognostic significance, but high FLT3 expression showed significant correlation with WBC count, as has been reported previously.8
NPM1 mutations were predominantly found in CN-AML cases, and we saw a frequency of NPM1 mutations (6%) that was comparable to previous reports on pediatric AML.17,44 The presence of an NPM1 mutation was associated with favorable EFS, but this association only reached statistical significance in patients without FLT3-ITD mutations, in accordance with other reports.16,17
CEBPA is a well-established favorable prognostic marker in adult CN-AML with both superior EFS and OS.45,46 The presence of CEBPA mutation was also recently reported by the Children's Oncology Group as a favorable prognostic marker in pediatric AML with significantly better EFS.18 The frequency of CEBPA mutations (6%) and the association with normal karyotype in our study was also in concordance with that study.18 However, we saw no correlation between CEBPA mutation and EFS or OS, which is why we conclude that CEBPA mutation status did not add any prognostic information in the framework of the NOPHO 1993 and 2004 protocols. Recent reports on adult AML indicate that the presence of double CEBPA mutations, rather than the presence of any CEBPA mutation, are associated with a good prognosis.45,46 Biallelic mutations predominated in our material (7 of 10), but the low number of CEBPA single-mutated patients makes it impossible to draw any conclusions about differences in survival between these 2 subgroups.
The prognostic relevance of WT1 mutation or WT1 expression in pediatric AML has been unclear. In the present study, WT1 mutations were most frequent in patients with normal karyotype and were commonly found together with FLT3-ITD. There was no correlation between WT1 mutations and EFS or OS, and, when the FLT3-ITD–positive cases were excluded, the remaining WT1-mutated cases actually showed a trend for better EFS and OS than cases with wt WT1. There was a positive correlation between low WT1 gene expression and monocytic or monoblastic AML (FAB M5) and with MLL rearrangement. High WT1 gene expression correlated with CN-AML and the presence of WT1 mutations and FLT3-ITD. However, no correlation with EFS or OS was observed. Our data therefore suggest that WT1 mutations and WT1 expression lack independent prognostic significance, which is also in accordance with a recent report from the Children's Oncology Group about WT1 mutations.27
High ERG and BAALC expression showed a significant association with inferior EFS. There was no significant correlation between MN1 expression and survival, even though there was a trend for better EFS for patients with low MN1 expression. Expression levels of ERG, MN1, and BAALC were significantly correlated with each other, and expression of MN1 and BAALC has also been shown to be coexpressed in gene expression profiling of CN-AML in adults.47 There were also some differences in their expression pattern. ERG was evenly expressed in different cytogenetic subgroups, whereas MN1 and BAALC expression showed similar cytogenetically dependent variation, including high expression in patients with CBF leukemia and lower expression in patients with MLL rearrangements. However, further investigation of the underlying events that regulate the expression of these genes is required to fully understand their expression in different subgroups of AML. We did not see any correlation between low ERG expression or low BAALC expression and the presence of NPM1 mutations (data not shown), which has been reported for both pediatric and adult AML.47,48
In multivariate analysis, FLT3-ITD and high BAALC expression remained as prognostic factors for adverse EFS together with good response to therapy and SCT in CR1. High ERG expression was not found as an independent prognostic marker, which might be explained by the strong association with high BAALC expression. Mutated NPM1 was not an independent prognostic marker, which was indicated already in the univariate analysis in which NPM1 mutation was only significantly associated with favorable EFS in the absence of FLT3-ITD. However, mutated NPM1 has previously been found as an independent prognostic marker of EFS in pediatric CN-AML not influenced by FLT3-ITD.20 This discrepancy might be because of differences in treatment protocols or the relative small number of patients with CN-AML in our study.
In summary, we conclude that analysis of the mutational status of FLT3 and NPM1 at diagnosis is important for correct prognostic stratification of patients with pediatric AML and that analysis of the expression level of at least BAALC can add important prognostic information. However, before gene expression levels can be used for prognostic stratification, standardized methods for quantitative measurement and threshold values must be established.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
This work was supported by grants from the Swedish government under the LUA/ALF agreement; the Swedish Childhood Cancer Foundation, the Åke Wiberg Foundation, Stockholm; and Lion's Cancer Foundation, Uppsala, Sweden.
Authorship
Contribution: A.S., M.K., R.H., R.R., G.L., E.F., H.H., L.P., and H.E. contributed to the design and analysis of the study; A.S. and L.P. wrote the manuscript, and all authors agreed on the final version; A.S., M.K., R.H., and H.B.O. performed laboratory-based research; A.S., J.A., and H.E. performed statistical analysis; J.A., E.F., K.J., O.G.J., B.Z., J.P., G.L., and H.H. were involved directly or indirectly in the care of patients and/or sample procurement; G.L. and J.P. established the biobank from which all the samples in this study were retrieved; H.H. is the curator responsible for maintaining and updating the NOPHO AML database; and E.F. maintains the NOPHO cytogenetic database.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Lars Palmqvist, Institute of Biomedicine, Department of Clinical Chemistry and Transfusion Medicine, University of Gothenburg, SE-413 45 Gothenburg, Sweden; e-mail: lars.palmqvist@gu.se; or Hans Ehrencrona, Department of Clinical Genetics, Lund University, SE-221 85, Lund, Sweden; e-mail: hans.ehrencrona@med.lu.se.
References
Author notes
A.S. and M.K. contributed equally to this study.
L.P. and H.E. contributed equally to this study.