To the editor:
Fingolimod (FTY720, 2-amino-2-propane-1,3-diol hydrochloride) is a remarkably efficient immunosuppressive drug that was recently approved as the first oral treatment for multiple sclerosis. Fingolimod prevents lymphocyte egress from lymph nodes by targeting 4 of 5 sphingosine-1-phosphate receptors.1 Two phase 3 trials showed that fingolimod significantly reduced multiple sclerosis disease progression compared with placebo or standard treatment.2,3 However, both trials concluded that longer studies were required to assess possible long-term risks.2,3 Reports from studies in mice and humans strongly support a crucial role of lymphocytes and adaptive immunity in both preventing cancer and fighting established tumors in a process called cancer immunosurveillance.4,5 In transplant recipients, lifelong immunosuppressive treatment is associated with an increased risk of cancer.6
We investigated the effect of fingolimod on cancer immunosurveillance mediated by tumor-specific CD4+ T cells in mouse models for myeloma and B-cell lymphoma. We used severe combined immunodeficient (SCID) mice made transgenic for a T cell receptor (TCR) that recognizes a tumor-specific idiotypic (Id) antigen that is secreted by MOPC315 myeloma and F9 B-cell lymphoma.7,8 The Id-specific TCR-transgenic SCID mice are resistant against subcutaneous challenge with MOPC315 and F9 cells. The protection is Id-specific, CD4+ T cell–mediated and does not require the presence of B cells and CD8+ T cells.7,8 On cancer cell inoculation, Id-specific CD4+ T cells first become activated in the draining lymph nodes where they acquire a T helper type 1 (Th1) phenotype. Id-specific Th1 cells then migrate to the incipient tumor site and secrete IFN-γ, resulting in local activation of macrophages which become tumoricidal and eradicate the cancer cells.9,10
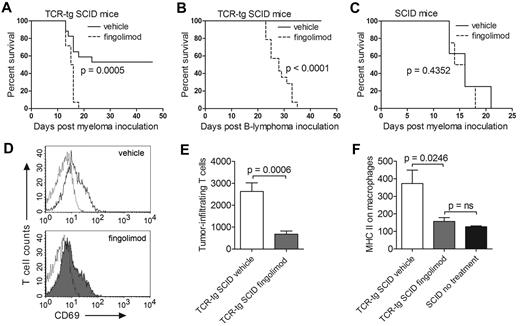
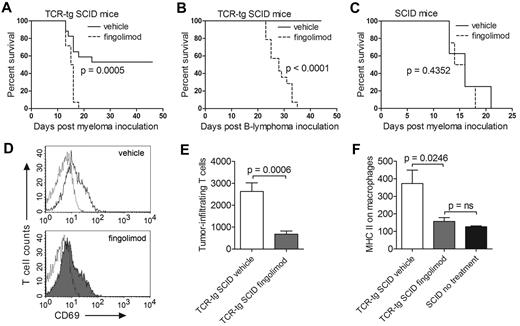
Id-specific TCR-transgenic SCID and control nontransgenic SCID mice were inoculated with cancer cells and treated daily with fingolimod or vehicle only. Fingolimod efficiently blocked rejection of both MOPC315 myeloma and F9 B-cell lymphoma by TCR-transgenic mice (Figure 1A-B). Fingolimod had no effect on the survival of control nontransgenic SCID mice inoculated with MOPC315 (Figure 1C). Fingolimod did not block activation of tumor-specific CD4+ T cells in the draining lymph nodes, as defined by up-regulation of CD69 (Figure 1D). In contrast, fingolimod strongly inhibited migration of tumor-specific CD4+ T cells to the incipient tumor site (Figure 1E). Furthermore, fingolimod prevented Th1-mediated activation of tumor-infiltrating macrophages, as measured by up-regulation of surface major histocompatibility (MHC) class II molecules. In fact, in TCR-transgenic mice treated with fingolimod, tumor-infiltrating macrophages had surface MHC class II levels that were as low as those of macrophages from T cell–deficient SCID mice (Figure 1F). Thus, the data strongly suggest that fingolimod blocks immunosurveillance of B-cell cancers by suppressing migration of tumor-specific Th1 cells from lymph nodes to the incipient tumor site, thereby preventing Th1-mediated activation of tumoricidal macrophages.
Fingolimod blocks antitumor immunity and prevents rejection of myeloma and B-cell lymphoma in TCR-transgenic SCID mice. (A) Id-specific TCR-transgenic (TCR-tg) SCID mice were inoculated subcutaneously with 1.6 × 105 MOPC315 myeloma cells and treated daily with either fingolimod (FTY720, Selleck Chemicals; 2 μg/g bodyweight) or with vehicle only (0.8% DMSO; Sigma-Aldrich) delivered intraperitoneally. Tumor growth was followed by palpation. Mice were euthanized when the tumor reached 10 mm in diameter (n = 14-17). (B) Id-specific TCR-transgenic SCID mice were inoculated subcutaneously with 1.6 × 105 F9 B-lymphoma cells and treated daily with fingolimod or with vehicle only. F9 cells are A20 B-lymphoma cells transfected with Id-containing L-chain from MOPC3157 (n = 14-16). (C) Nontransgenic SCID mice were inoculated subcutaneously with 1.6 × 105 MOPC315 cells and treated daily with fingolimod or with vehicle only (n = 8). (D-F) Id-specific TCR-transgenic and nontransgenic SCID mice were inoculated subcutaneously with 105 MOPC315 myeloma cells embedded in Matrigel.9 TCR-transgenic SCID mice were treated daily with fingolimod (1 μg/g bodyweight) or with vehicle only (n = 8-10). SCID mice were left untreated (n = 2). At day +8, draining lymph nodes and Matrigel plugs were dissected and analyzed by flow cytometry.9 (D) CD69 expression on gated Id-specific CD4+ T cells in draining lymph nodes of representative TCR-transgenic SCID mice treated with vehicle only (top) or fingolimod (bottom). Dotted lines indicate an isotype-matched control antibody. (E) Number of Id-specific T cells per Matrigel plug (mean ± SEM). (F) Expression of the activation marker MHC class II on Matrigel-infiltrating CD11b+ macrophages (geometric mean ± SEM). P values were calculated with the log-rank test (A-C) and the t test (E-F). ns indicates not significant.
Fingolimod blocks antitumor immunity and prevents rejection of myeloma and B-cell lymphoma in TCR-transgenic SCID mice. (A) Id-specific TCR-transgenic (TCR-tg) SCID mice were inoculated subcutaneously with 1.6 × 105 MOPC315 myeloma cells and treated daily with either fingolimod (FTY720, Selleck Chemicals; 2 μg/g bodyweight) or with vehicle only (0.8% DMSO; Sigma-Aldrich) delivered intraperitoneally. Tumor growth was followed by palpation. Mice were euthanized when the tumor reached 10 mm in diameter (n = 14-17). (B) Id-specific TCR-transgenic SCID mice were inoculated subcutaneously with 1.6 × 105 F9 B-lymphoma cells and treated daily with fingolimod or with vehicle only. F9 cells are A20 B-lymphoma cells transfected with Id-containing L-chain from MOPC3157 (n = 14-16). (C) Nontransgenic SCID mice were inoculated subcutaneously with 1.6 × 105 MOPC315 cells and treated daily with fingolimod or with vehicle only (n = 8). (D-F) Id-specific TCR-transgenic and nontransgenic SCID mice were inoculated subcutaneously with 105 MOPC315 myeloma cells embedded in Matrigel.9 TCR-transgenic SCID mice were treated daily with fingolimod (1 μg/g bodyweight) or with vehicle only (n = 8-10). SCID mice were left untreated (n = 2). At day +8, draining lymph nodes and Matrigel plugs were dissected and analyzed by flow cytometry.9 (D) CD69 expression on gated Id-specific CD4+ T cells in draining lymph nodes of representative TCR-transgenic SCID mice treated with vehicle only (top) or fingolimod (bottom). Dotted lines indicate an isotype-matched control antibody. (E) Number of Id-specific T cells per Matrigel plug (mean ± SEM). (F) Expression of the activation marker MHC class II on Matrigel-infiltrating CD11b+ macrophages (geometric mean ± SEM). P values were calculated with the log-rank test (A-C) and the t test (E-F). ns indicates not significant.
In summary, fingolimod is a strong immunosuppressive drug which blocks immunosurveillance of myeloma and B-cell lymphoma by CD4+ T cells in experimental mouse models, resulting in cancer development. Further studies are required to determine the effect of fingolimod on other immune cells involved in cancer immunosurveillance such as CD8+ T cells and NK cells.4 In our experiments, mice were treated with rather high doses of fingolimod (1-2 mg/kg bodyweight/d) to efficiently block sphingosine-1-phosphate receptors. Patients receive much lower doses (0.5-1.25 mg/d) but for longer time periods (years). Therefore, our findings cannot be directly translated to patients. However, the data suggest that long-term treatment with fingolimod may potentially lead to increased risk of cancer in humans.
Authorship
Acknowledgments: The authors thank Egil Røsjø and Trygve Holmøy for useful discussions; Ole Audun Werner Haabeth for technical help; and Inger Øynebråten and Peter O. Hofgaard for critical reading of the manuscript.
This work was supported by grants from Helse Sør-Øst, the Norwegian Cancer Society, Anders Jahre fund, Henrik Homans Minde fund, Legat for fremme av kreftforskningen, Familien Blix fund, S. G. Sønneland foundation, H. G. og Andrine Berg og Hans Gysler Berg fund, and Ella og Kristian Nyerrøds foundation.
Contribution: K.B.L. performed the experiments, collected the data, and prepared the figure; A.C. and B.B. designed the study; K.B.L. wrote the manuscript; A.C. and B.B. contributed in writing the manuscript; and all authors analyzed and discussed the data.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Kristina Berg Lorvik or Alexandre Corthay, Department of Immunology, Oslo University Hospital Rikshospitalet, PO Box 4950 Nydalen, 0424 Oslo, Norway; e-mail: k.b.lorvik@medisin.uio.no or alexandre.corthay@medisin.uio.no.